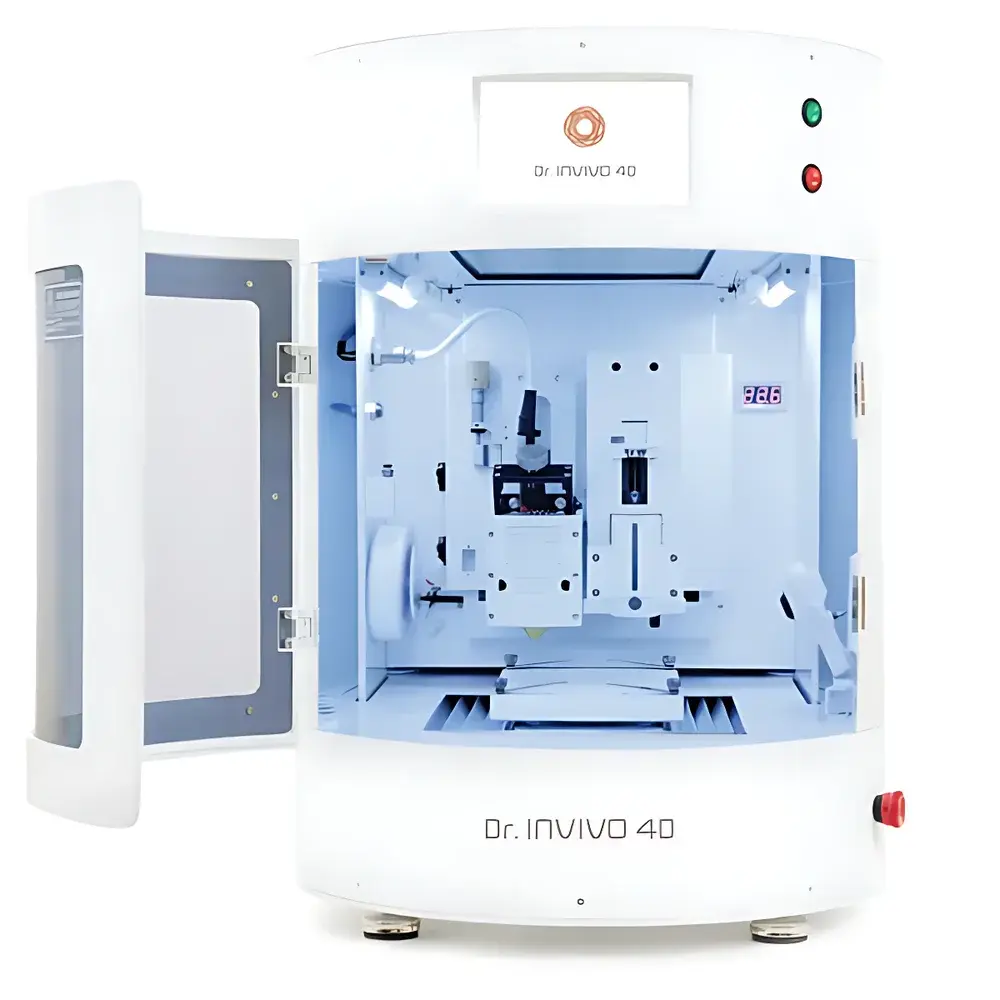
ASI INVIVO 4D High-Resolution Biological 3D Printer
| Brand | ASI |
|---|---|
| Origin | South Korea |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | INVIVO 4D |
| Pricing | Available Upon Request |
Overview
The ASI INVIVO 4D High-Resolution Biological 3D Printer is an advanced biomanufacturing platform engineered for precise, layer-by-layer deposition of living cells and biomaterials under sterile, physiologically relevant conditions. Unlike conventional fused deposition modeling (FDM) or stereolithography (SLA) systems, the INVIVO 4D employs multi-modal extrusion—combining pneumatic, piston-driven, and micro-syringe-based dispensing—with real-time environmental control (temperature, humidity, CO₂) to maintain cell viability during printing. Its core architecture supports true 4D functionality: spatially resolved 3D fabrication coupled with time-dependent structural or functional evolution—enabled by stimuli-responsive bioinks (e.g., thermoresponsive gelatin-methacryloyl, light-crosslinkable hyaluronic acid derivatives). Designed for translational research labs and GMP-adjacent development environments, it bridges high-fidelity tissue architecture with regulatory-compliant process documentation.
Key Features
- Sub-50 µm feature resolution across X/Y axes, achieved via high-precision stepper motor control and closed-loop feedback positioning.
- Multi-channel independent extrusion system supporting simultaneous deposition of up to six distinct bioinks—including hydrogels, cell-laden pastes, granular scaffolds, and sacrificial materials—without cross-contamination.
- Integrated on-stage incubation chamber maintaining 37 °C ±0.3 °C, 5% CO₂ ±0.2%, and >95% relative humidity throughout print cycles up to 72 hours.
- Modular toolhead interface compatible with thermal, UV, and visible-light crosslinking modules for in situ polymerization and spatial patterning.
- ISO Class 5 (Class 100) cleanroom-rated enclosure with HEPA filtration and laminar airflow, certified per ISO 14644-1.
- Compliance-ready firmware with audit trail logging, user access levels, and electronic signature support aligned with FDA 21 CFR Part 11 requirements.
Sample Compatibility & Compliance
The INVIVO 4D accommodates a broad spectrum of printable biomaterials: natural and synthetic hydrogels (e.g., alginate, collagen I, PEGDA), shear-thinning nanocellulose composites, ceramic-loaded pastes, microsphere suspensions, and thermoplastic filaments with embedded growth factors. All material handling pathways are sterilizable via autoclave (121 °C, 20 min) or ethylene oxide gas. The system meets ISO 13485:2016 design controls for medical device manufacturing support and is validated for use in protocols compliant with ASTM F2792 (Standard Terminology for Additive Manufacturing) and ISO/ASTM 52900 (Additive Manufacturing – General Principles). It supports GLP-aligned experimental workflows and facilitates traceability from raw material lot numbers to printed construct metadata.
Software & Data Management
Controlled via INVIVO Studio—a dedicated Windows-based application built on Qt framework—the platform provides intuitive CAD-to-bioprint workflow integration. Users import STL or 3MF files, apply adaptive slicing with variable layer thickness (10–200 µm), define material-specific extrusion profiles (pressure, speed, dwell time), and assign biological parameters (cell density, viability thresholds). All print jobs generate encrypted JSON logs containing timestamped sensor data (temperature, pressure, position), user actions, and calibration history. Export options include CSV, HDF5, and DICOM-RT for downstream analysis in MATLAB, Python (NumPy/SciPy), or commercial image reconstruction tools. Remote monitoring via secure HTTPS API enables integration into centralized lab information management systems (LIMS).
Applications
- Bioprinting of vascularized 3D tissue constructs for preclinical drug metabolism and toxicity screening (e.g., liver sinusoid models, cardiac microtissues).
- Development of patient-specific osteochondral grafts using dual-material deposition of chondrocyte-laden gelatin methacrylate and calcium phosphate scaffolds.
- Fabrication of organ-on-a-chip devices integrating endothelialized microchannels with parenchymal cell layers for barrier function studies.
- High-throughput fabrication of micropatterned hydrogel arrays for stem cell differentiation assays under controlled mechanical and biochemical cues.
- Educational deployment in graduate-level tissue engineering curricula, supporting hands-on training in bioprocess validation and scaffold design principles.
FAQ
Does the INVIVO 4D support live-cell imaging during printing?
Yes—optional integrated inverted microscope module (Leica DMI series compatible) enables real-time phase-contrast and fluorescence monitoring with motorized Z-stack acquisition synchronized to print head motion.
Can the system be validated for GMP-compliant production?
While classified as a research-use-only (RUO) instrument per regulatory definitions, its hardware architecture, documentation package (IQ/OQ templates included), and Part 11–compliant software enable qualification for early-phase clinical trial material manufacturing under sponsor oversight.
What bioink formulation support is provided?
ASI offers a validated library of >12 proprietary bioinks (including xeno-free, serum-free, and animal-component-free variants), alongside technical collaboration for custom rheological optimization and print parameter mapping.
Is remote service and calibration available?
Yes—cloud-connected diagnostics, over-the-air firmware updates, and scheduled remote calibration verification are supported through ASI’s global service network, with on-site engineering available in EMEA, APAC, and North America regions.