Azenta Biobanking Solutions
| Brand | Azenta (formerly Brooks Life Sciences) |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Product Category | Imported Biobanking Infrastructure Systems |
| Temperature Ranges | RT to –20°C, –80°C, –150°C (Vario), –196°C (Cryo) |
| Automation Level | Fully Automated Sample Storage & Retrieval |
| Compliance | Designed for GLP/GMP-aligned biorepository operations, 21 CFR Part 11–ready software architecture |
| Dimensions (BioStore II –80°C, Quad-height) | 12.77 m × 2.78 m × 3.04 m |
| Dimensions (BioStore III Cryo –196°C, Ultra-high capacity) | 18.17 m × 4.78 m × 3.24 m |
| LN₂ Consumption (BioStore III Cryo, static) | ~9.5–12.5 L/day |
| Temperature Hold Time (fail-safe) | ≥21 days at –190°C after power loss |
Overview
Azenta Biobanking Solutions represent a vertically integrated, end-to-end infrastructure platform engineered for high-integrity, scalable, and audit-ready biological sample stewardship. Built upon the legacy of Brooks Life Sciences and now unified under Azenta Life Sciences, these systems implement cryogenic automation grounded in validated thermal physics and robotic precision engineering. The core architecture leverages liquid nitrogen (LN₂) vapor-phase storage at –196°C (BioStore III Cryo), mechanically stabilized ultra-low temperature (ULT) environments at –80°C (BioStore II), and controlled–20°C or ambient–temperature automated repositories (SampleStore II), all governed by a centralized informatics layer. Unlike modular add-ons, Azenta’s approach embeds traceability, thermal stability, and operational resilience into the physical and digital design—ensuring sample integrity across decades of longitudinal studies, clinical trial biobanks, and translational research cohorts.
Key Features
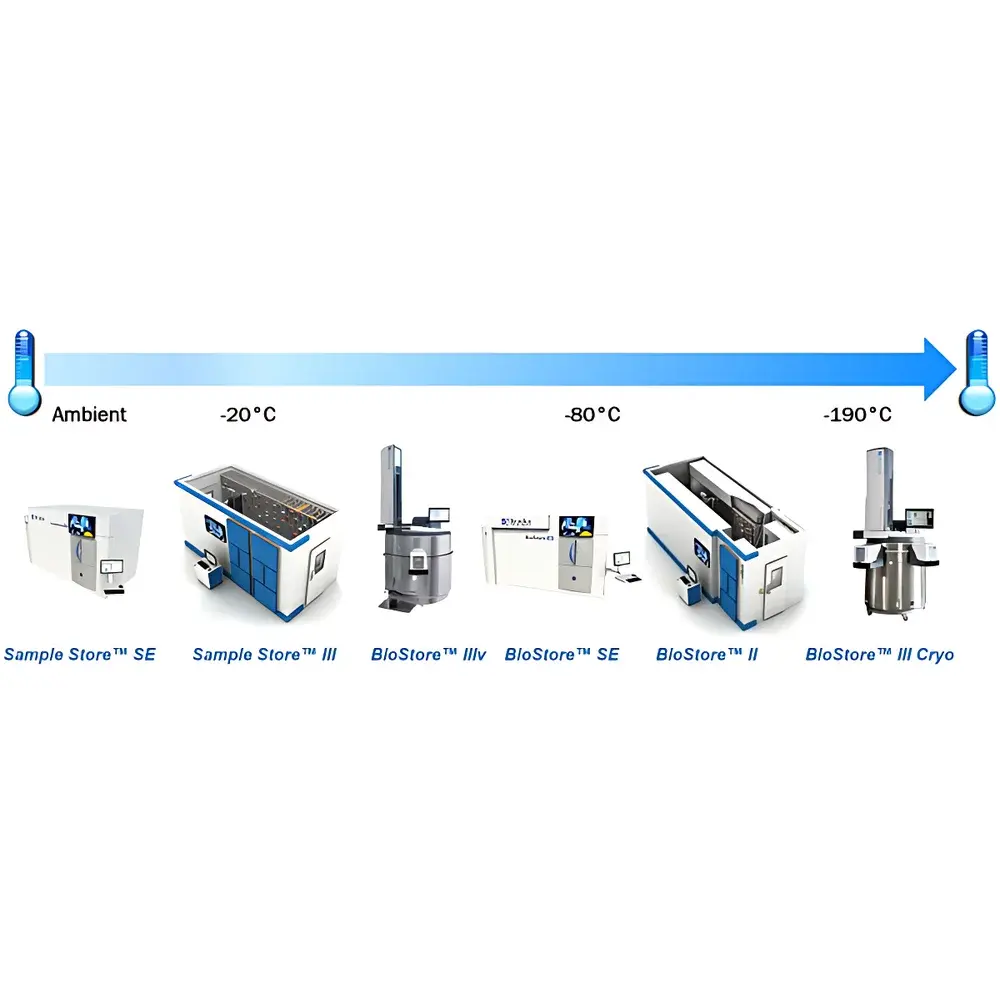
- Multi-tiered temperature automation: Seamless integration across RT/–20°C (SampleStore II), –80°C (BioStore II), and cryogenic –196°C (BioStore III Cryo) or variable-range –20°C to –150°C (BioStore III Vario) platforms—each with independent environmental monitoring and failover logic.
- Fail-safe thermal retention: BioStore III Cryo systems maintain ≤ –190°C for ≥21 days post-power failure, verified per ICH M7 and ISO 20387 Annex A requirements for biobank continuity planning.
- Modular, SBS-compliant storage: Supports standardized cryoboxes (e.g., FluidX 2D-barcoded tubes), blood bags, PCR plates, and custom cassettes—enabling mixed-format inventory within a single robotic footprint.
- Full-chain auditability: Every sample movement is timestamped, user-authenticated, and logged with source location, destination slot, and environmental history—meeting FDA 21 CFR Part 11 electronic record and signature requirements.
- Zero-unattended-thaw assurance: Intelligent pathing algorithms prevent incidental warming of non-target samples during retrieval; no manual intervention required for routine access or emergency recovery.
Sample Compatibility & Compliance
Azenta biobanking systems accommodate primary human biospecimens (whole blood, PBMCs, tissue sections, FFPE blocks), nucleic acids (DNA/RNA), cell lines, and viral vectors—across ISO 20387:2018–certified biobank workflows. All hardware and software components are validated per ISO/IEC 17025 principles for measurement traceability and undergo IQ/OQ/PQ protocols aligned with CAP, CLIA, and EU IVDR Annex II expectations. The FluidX 2D tube ecosystem—including scanners, tube openers, and laser-marking stations—is pre-qualified for ISO/IEC 17025–compliant barcode readability down to 0.25 mm² feature size. Cryo storage modules comply with ASTM F2649 (Standard Practice for Cryopreservation of Cells) and support long-term viability validation per USP sterility and cryopreservation guidance.
Software & Data Management
The integrated Biobank Informatics Platform (BIP) serves as the central orchestration engine—supporting role-based access control (RBAC), real-time inventory dashboards, automated QC flagging (e.g., temperature excursions >±0.5°C), and HL7/FHIR-compliant data exchange with LIMS, EMR, and genomic analysis pipelines. Audit trails include immutable event logs with SHA-256 hashing, configurable retention policies (≥10 years), and exportable reports for regulatory submissions (FDA eCTD, EMA CTD). Software validation documentation—including URS, FS, DS, and test scripts—is provided as part of the system delivery package, supporting GxP inspection readiness.
Applications
- Population-scale biobanks requiring >1 million sample capacity with <0.001% misplacement rate
- Phase I–III clinical trial specimen repositories with chain-of-custody enforcement and protocol-driven retrieval rules
- Academic core facilities managing multi-omics sample cohorts (genomics, proteomics, metabolomics) under shared-access governance models
- Cell and gene therapy (CGT) manufacturing sites storing master/working cell banks under cGMP Annex 1–aligned cold chain controls
- National health initiatives deploying federated biobank networks with synchronized metadata standards (e.g., MIABIS 2.0)
FAQ
What temperature uniformity is maintained across the BioStore III Cryo chamber?
Temperature variation remains within ±0.8°C across all storage zones under steady-state conditions, verified via NIST-traceable thermocouple mapping per ISO/IEC 17025.
Can existing legacy samples be migrated into an automated Azenta system without integrity compromise?
Yes—validated migration protocols include pre-transfer viability assays, controlled-rate re-freezing (where applicable), and dual-verification logging (pre- and post-load inventory reconciliation).
Is remote monitoring and alarm notification supported?
All systems integrate with enterprise SNMP and Syslog infrastructure; SMS/email alerts trigger on predefined thresholds (e.g., LN₂ level 30 sec, temp deviation >±1.0°C).
How is software validation handled for regulated environments?
Azenta provides full GAMP 5–aligned validation documentation, including risk assessments (ICH Q9), test scripts, and evidence packages suitable for FDA, EMA, and PMDA review.
Are consumables such as FluidX tubes supplied under ISO 13485-certified manufacturing?
Yes—FluidX 2D-coded tubes, racks, and sealing films are manufactured under ISO 13485:2016 and certified free of detectable DNase/RNase, human DNA, and endotoxin (<0.01 EU/mL).