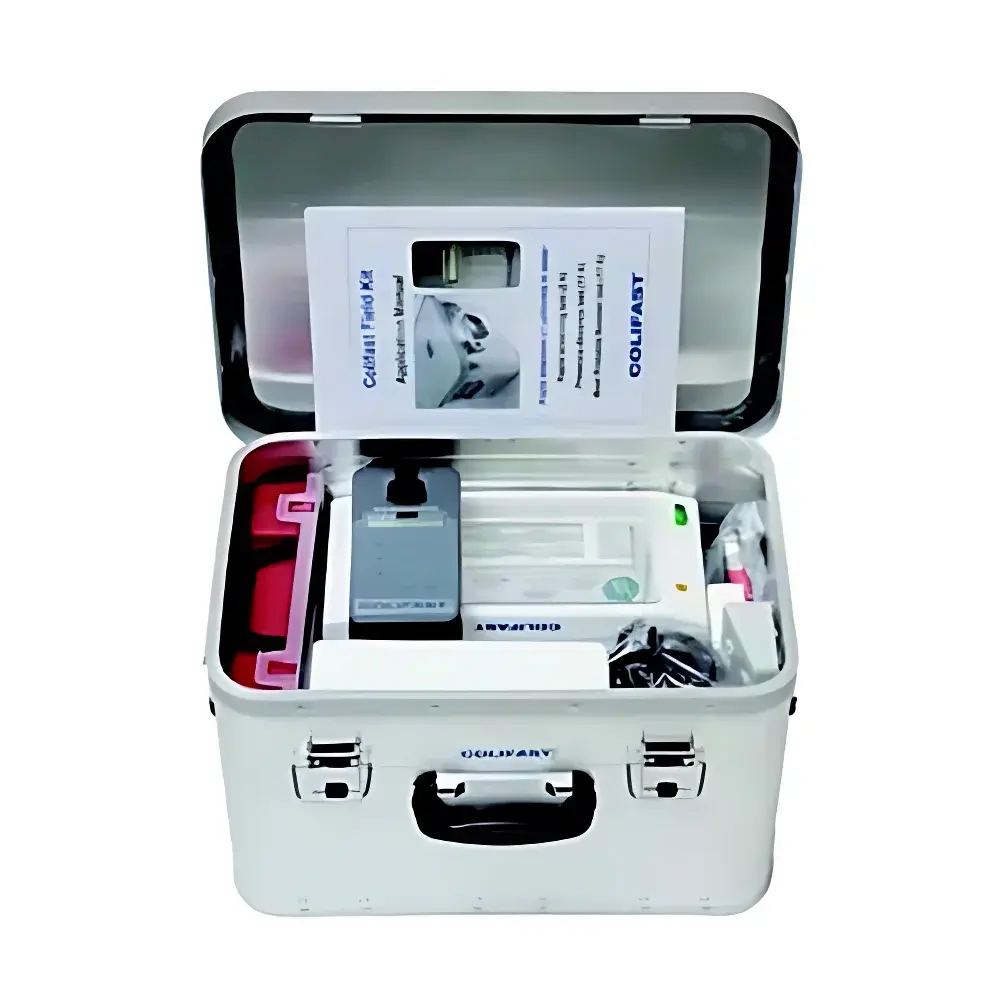
BBE Colifast Field Kit Portable E. coli and Total Coliform Detection System
| Brand | BBE Moldaenke |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Regional Classification | Domestic (China) |
| Model | Colifast Field Kit |
| Pricing | Available Upon Request |
Overview
The BBE Colifast Field Kit is a field-deployable, enzymatic fluorescence-based detection system engineered for rapid, on-site quantification and presence/absence testing of total coliforms, thermotolerant coliforms (including Escherichia coli), and specifically E. coli in water samples. It operates on the principle of enzyme-substrate fluorogenic reaction: coliform bacteria—particularly those expressing β-D-galactosidase (total coliforms) and β-D-glucuronidase (E. coli)—hydrolyze chromogenic/fluorogenic substrates embedded in proprietary selective growth media. This enzymatic cleavage releases a fluorescent compound whose intensity correlates directly with viable bacterial concentration. The system integrates incubation, fluorescence excitation at 365 nm, and photometric detection within a compact, battery-compatible micro-detection unit. Unlike membrane filtration or standard MPN methods, Colifast eliminates the need for colony isolation, plate reading, or subjective interpretation—delivering objective, instrument-based results traceable to ISO 9308-1 and EN ISO 9308-2 for routine water quality monitoring.
Key Features
- Field-ready portable architecture: All components—including micro-detection unit, temperature-controlled incubator (37 °C or 44 °C selectable), calibrated reagents, and consumables—are housed in a rugged, transportable case compliant with IP54 environmental protection standards.
- Dual-mode detection capability: Supports both quantitative Most Probable Number (MPN) estimation (detection limit: 1 CFU/100 mL) and qualitative presence/absence testing (1 CFU/100 mL sensitivity) within 6–15 hours, depending on target organism and sample matrix.
- Chemically selective media formulation: Growth medium contains nutrient sources, enzymatic substrates (e.g., MUG for E. coli), and selective inhibitors (e.g., bile salts, sodium lauryl sulfate) to suppress non-coliform flora while supporting robust target organism growth.
- Integrated chlorine neutralization: Includes 10% sodium thiosulfate solution for immediate deactivation of residual disinfectants—critical for accurate enumeration in chlorinated distribution systems or post-treatment effluents.
- No laboratory infrastructure required: Operates independently of microbiological hoods, autoclaves, or trained microbiologists; validated for use by field technicians following GLP-aligned procedural documentation.
- Traceable calibration protocol: Supplied with Cal 48 reference solution and blank control vials enabling daily instrument verification per ISO/IEC 17025 calibration traceability requirements.
Sample Compatibility & Compliance
The Colifast Field Kit is validated for use with raw surface water, treated drinking water, recreational bathing waters (fresh and marine), wastewater effluents, and process water from food/beverage production lines. Sample volumes are standardized at 10 mL per test vial, compatible with both grab and filtered sampling protocols. The assay complies with method performance criteria outlined in ISO 9308-1 (total coliforms), ISO 9308-2 (thermotolerant coliforms), and US EPA Method 1604 for E. coli. All reagents meet EU REACH and CLP regulatory classifications. Documentation supports audit readiness under WHO Guidelines for Drinking-water Quality, EU Bathing Water Directive (2006/7/EC), and FDA 21 CFR Part 11-compliant data integrity frameworks when paired with optional digital reporting modules.
Software & Data Management
While the base Colifast Field Kit operates as a standalone hardware system, its fluorescence readouts are digitally recorded via the integrated micro-detection unit’s internal memory (up to 500 test records). Optional USB export enables transfer to BBE’s Colifast Connect desktop software (Windows-compatible), which provides automated MPN table lookup, trend charting across sampling campaigns, PDF report generation (including operator ID, timestamp, location GPS metadata), and audit trail logging aligned with GLP/GMP data integrity principles. All software modules support electronic signature functionality and comply with ALCOA+ (Attributable, Legible, Contemporaneous, Original, Accurate) data governance standards.
Applications
- River and watershed fecal pollution source tracking via longitudinal sampling and spatial mapping of coliform hotspots.
- Regulatory compliance monitoring at inland and coastal bathing sites—supporting real-time decisions on public access advisories.
- In-process verification in municipal drinking water treatment plants, including pre- and post-filtration, disinfection efficacy assessment, and distribution system integrity checks.
- Emergency response deployment during natural disasters or infrastructure failures—enabling rapid verification of point-of-use water safety prior to distribution.
- Industrial hygiene validation in pharmaceutical manufacturing, dairy processing, and beverage bottling facilities where water contact surfaces require routine microbial release testing.
FAQ
What is the minimum detection level for E. coli using this kit?
The system achieves a confirmed detection limit of 1 CFU per 100 mL for E. coli in presence/absence mode, and down to 1–10 CFU/100 mL in MPN mode depending on incubation duration and sample turbidity.
Can the kit be used with seawater or high-salinity samples?
Yes—the selective media formulation has been empirically validated for marine and brackish matrices; no dilution or salinity adjustment is required for samples up to 35 g/L TDS.
Is instrument calibration required before each use?
Daily verification using supplied Cal 48 solution and blank vial is recommended; full recalibration is performed annually or after sensor replacement, per ISO/IEC 17025 maintenance schedules.
How does the system handle samples containing residual chlorine?
Each kit includes a dedicated 10% sodium thiosulfate dropper bottle for immediate neutralization of free chlorine and chloramines prior to inoculation—ensuring viability of target organisms.
Are consumables supplied with batch-specific certificates of analysis?
Yes—each lot of growth medium vials and Cal 48 solution is accompanied by CoA documentation specifying expiry date, sterility validation (ISO 11737-1), and fluorometric performance verification against NIST-traceable standards.