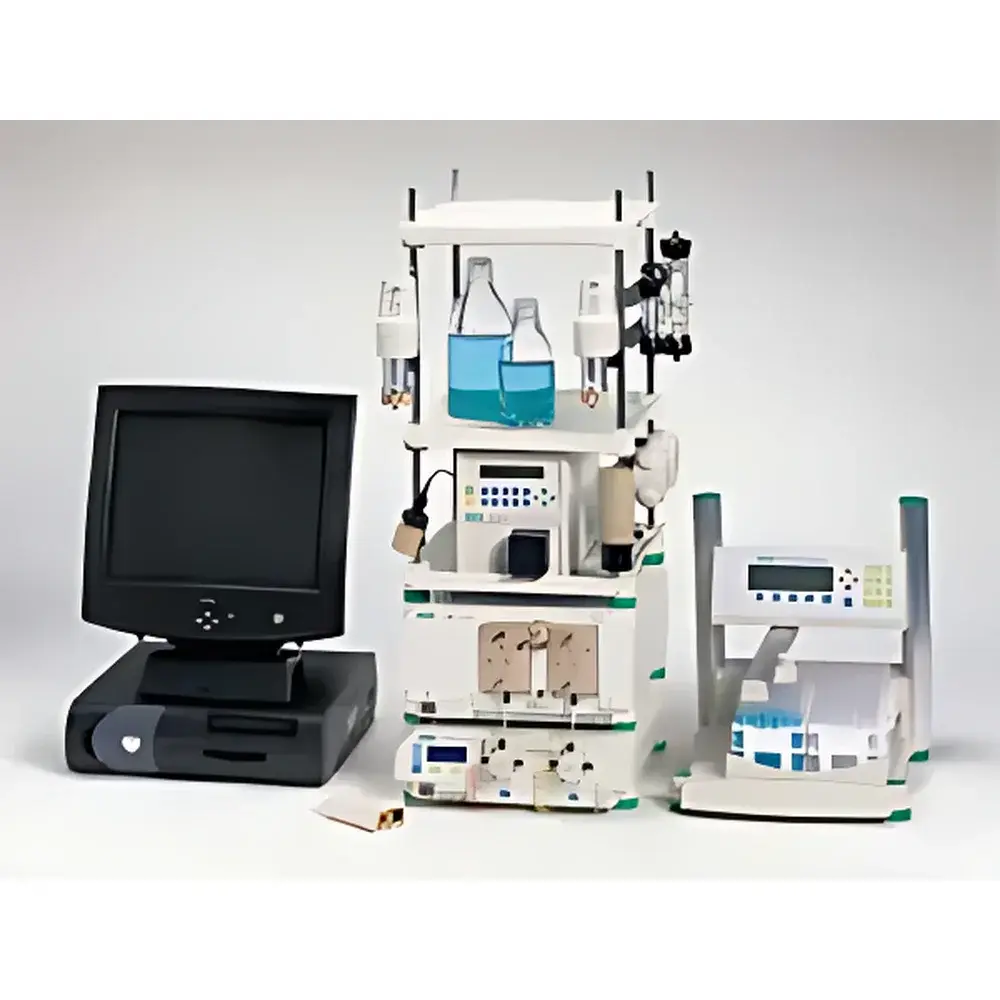
Bio-Rad BioLogic DuoFlow Dual-Flow Medium-to-High-Pressure Protein Analysis and Preparative Chromatography System
| Origin | Imported |
|---|---|
| Manufacturer Type | Authorized Distributor |
| Model | BioLogic DuoFlow System |
| Maximum Pressure | 3500 psi (23 MPa) |
| Flow Rate Range | 0.01–80 mL/min (configurable via pump head selection) |
| UV Detection Wavelengths | Fixed 254/280 nm |
| Conductivity Range | 1–500 mS/cm |
| UV Sensitivity | 0.0001–2.0 OD |
| Fraction Collection Modes | Time-based (0.03–999 min), Drop-based (0–9999 drops), Volume-based, Large-volume (20 positions, up to 500 mL each) |
| Temperature-Controlled Operation | Compatible with chromatography cold cabinets (4–10 °C) |
| System Biocompatibility | Fully bio-inert fluidic path (PEEK, titanium, sapphire, fused silica) |
| Software Platform | BioLogic System Controller v5.x (Windows-based, FDA 21 CFR Part 11 compliant audit trail option) |
Overview
The Bio-Rad BioLogic DuoFlow Dual-Flow Medium-to-High-Pressure Protein Analysis and Preparative Chromatography System is an engineered solution for scalable liquid chromatography applications spanning analytical characterization, process development, pilot-scale purification, and GMP-aligned small-batch production of therapeutic proteins, monoclonal antibodies, viral vectors, and other biologics. Built upon a dual-pump architecture grounded in positive-displacement piston technology, the system operates across two distinct pressure–flow regimes—enabling both high-resolution analytical separations (up to 3500 psi / 23 MPa at 10 mL/min) and robust preparative workflows (up to 1000 psi / 6.6 MPa at 80 mL/min). Its core design adheres to principles of laminar flow control, minimal extra-column dispersion, and thermally stable detection—critical for preserving conformational integrity of labile biomolecules. The system’s fully modular topology allows seamless integration of orthogonal detection modalities (UV-Vis, conductivity, pH, refractive index, fluorescence), automated fraction collection, and auxiliary fluid handling—making it suitable for method scouting, gradient optimization, column screening, and multi-step purification protocols under controlled environmental conditions.
Key Features
- Dual-pump architecture with interchangeable F10 (10 mL/min, 3500 psi) and F40 (40 mL/min, 1000 psi) pump heads—enabling single-system adaptability across analytical and preparative workflows without hardware replacement.
- Fully bio-inert fluidic path constructed from PEEK, titanium, sapphire, and fused silica components—validated for compatibility with aggressive solvents (e.g., 8 M urea, 6 M guanidine HCl), low-pH eluents, and oxidizing agents used in protein denaturation or cleaning-in-place (CIP) procedures.
- Integrated dual-wavelength UV detector (254 nm & 280 nm) with optional wavelength expansion to 214 nm (peptide bond), 365 nm (tryptophan), and visible-range filters (405–546 nm); upgrade path to real-time, four-channel simultaneous spectral acquisition.
- Conductivity detector (1–500 mS/cm range) with temperature compensation and digital signal filtering—essential for monitoring buffer exchange, ion-exchange elution profiles, and desalting steps.
- BioFrac fraction collector featuring XY-axis positioning, programmable overlap collection, 20 large-volume positions (up to 500 mL each), motorized vertical arm movement, and integrated ice-bath tray—designed to maintain sample stability during extended collection cycles.
- Expandable valve manifold architecture supporting up to 12-port switching valves—enabling automated buffer selection, sample loading, column backflushing, multi-column serial/parallel operation, and pH-adjusted in-line mixing via optional Maximizer module.
Sample Compatibility & Compliance
The DuoFlow system accommodates a broad spectrum of sample matrices—including clarified cell lysates, ammonium sulfate precipitates, dialyzed supernatants, and filtered monoclonal antibody harvests—across column dimensions ranging from analytical (4.6 × 150 mm) to preparative (26 × 300 mm). Its pressure and flow flexibility supports common chromatographic modes: reversed-phase (RP-HPLC), size-exclusion (SEC), ion-exchange (IEX), hydrophobic interaction (HIC), and affinity chromatography (e.g., Protein A, Ni-NTA). All wetted materials comply with USP Class VI biocompatibility standards and ISO 10993-5 cytotoxicity testing. When configured with electronic signature, audit trail, and user access controls, the system meets GLP and GMP requirements per FDA 21 CFR Part 11 and EU Annex 11 for regulated environments. Routine operation aligns with ASTM E2500-13 (good practice for verification of pharmaceutical equipment) and ISO/IEC 17025 traceability frameworks.
Software & Data Management
BioLogic System Controller v5.x provides a unified Windows-based interface for instrument control, method development, real-time data visualization, and post-run analysis. The software supports hierarchical project management (user → project → method → run), parameter-driven gradient programming, and time-resolved event logging (valve actuation, pump start/stop, detector zeroing). Chromatograms are annotated with retention time markers, peak integration boundaries, and baseline correction points; overlay comparisons between runs support method transfer validation. Raw data files (.bdf) are stored in vendor-neutral formats compatible with third-party analysis tools (e.g., Chromeleon, Empower, MATLAB). Optional 21 CFR Part 11 compliance package includes role-based authentication, electronic signatures, immutable audit trails, and encrypted database backups—facilitating regulatory submissions and internal quality audits.
Applications
- Process development of recombinant proteins and bispecific antibodies—including capture, intermediate purification, and polishing steps under varying pH and conductivity conditions.
- Stability-indicating assays for assessing aggregation (SEC), deamidation (IEX), oxidation (RP-HPLC), and fragmentation (CE-SDS surrogate).
- Buffer formulation studies using in-line pH/conductivity blending to map elution windows and optimize salt gradients.
- Multi-column chromatography (MCC) and periodic counter-current chromatography (PCC) implementation via synchronized valve sequencing and fraction redirection logic.
- Preparative isolation of post-translational modification variants (e.g., glycoforms, phosphorylated isoforms) using shallow gradient IEX or HILIC methods.
- Method qualification per ICH Q2(R2) guidelines—including precision, accuracy, linearity, LOD/LOQ, and robustness assessment across multiple operators and instruments.
FAQ
Can the DuoFlow system be validated for use in GMP manufacturing environments?
Yes—when equipped with the 21 CFR Part 11 compliance option and operated under documented SOPs, the system supports IQ/OQ/PQ execution and is routinely deployed in clinical-stage bioprocess labs for release testing and intermediate purification.
Is remote monitoring or network integration supported?
The controller supports TCP/IP communication and OPC UA server configuration, enabling integration into plant-wide SCADA systems and centralized data lakes via secure industrial Ethernet protocols.
What column hardware interfaces are natively supported?
Standard 1/4″-28 UNF and 10-32 threaded fittings accommodate most commercial columns; optional Swagelok® or VICI Valco™ adapters enable direct connection to custom-packed or monolithic devices.
How is system suitability verified prior to critical runs?
Built-in system suitability tests include pressure decay checks, flow accuracy verification (gravimetrically calibrated), UV lamp intensity profiling, and conductivity cell constant validation—all reportable within the audit trail.
Does Bio-Rad provide application support for novel purification strategies?
Yes—Bio-Rad’s Application Science team offers method development consulting, column selection guidance, and protocol optimization services aligned with USP , ICH Q5, and EMA CHMP/BWP/466/97 regulatory expectations.