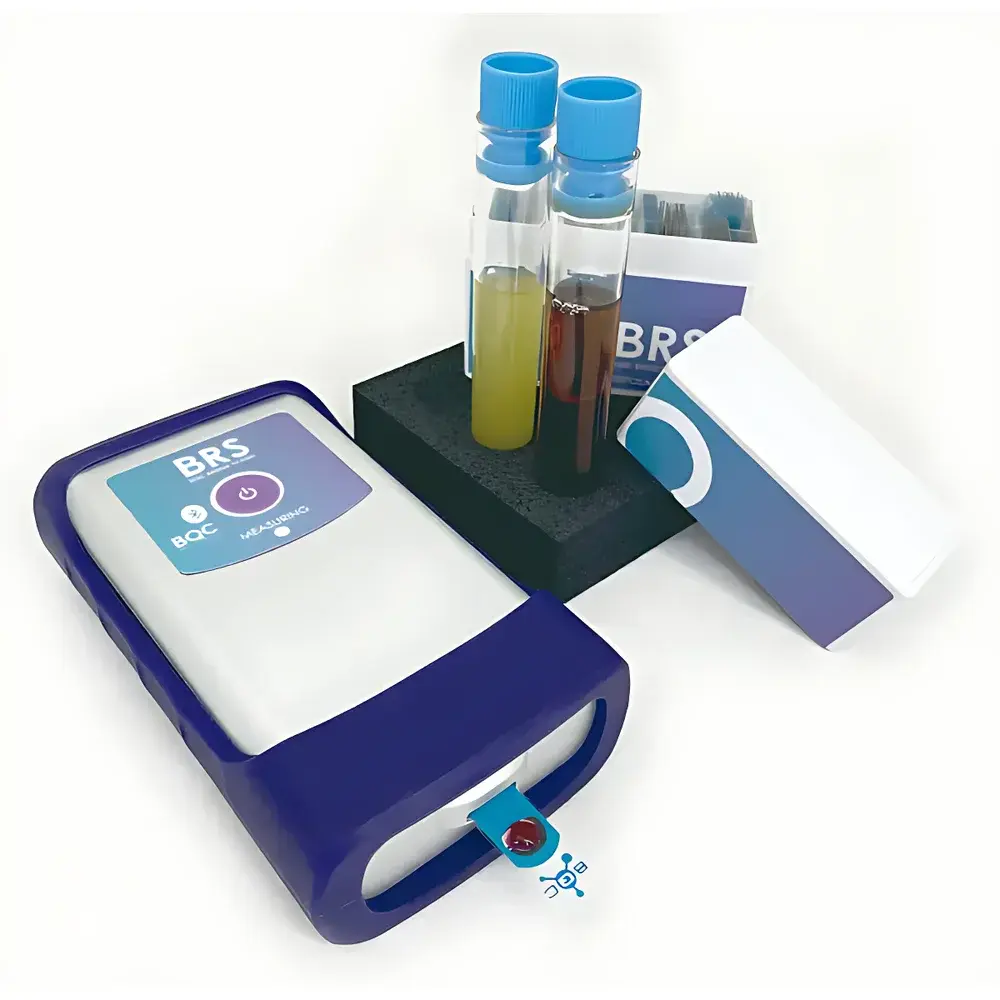
BQC BRS Portable Electrochemical Total Antioxidant Capacity Analyzer
| Brand | BQC |
|---|---|
| Origin | Spain |
| Manufacturer Type | Authorized Distributor |
| Product Category | Imported Instrument |
| Model | BRS |
| Pricing | Available Upon Request |
Overview
The BQC BRS Portable Electrochemical Total Antioxidant Capacity Analyzer is a compact, field-deployable instrument engineered for rapid, label-free quantification of redox-active species in complex liquid matrices. It operates on the principle of controlled-potential voltammetry—specifically linear sweep voltammetry (LSV) and chronoamperometry—to electrochemically oxidize endogenous antioxidants present in biological, food, agricultural, and cosmetic samples. Unlike optical methods (e.g., CUPRAC, ORAC, FRAP), which rely on chromogenic reactions, colorimetric interference, or radical initiation chemistry, the BRS measures the integrated faradaic charge generated during oxidation, directly correlating with total reducing capacity. This electrochemical approach preserves native sample integrity—no organic solvents, no free radical initiators, no derivatization, and no dilution-dependent calibration curves. The device delivers quantitative TAC (Total Antioxidant Capacity) and H2O2 scavenging activity within 60 seconds using only a single 5–10 µL droplet, making it suitable for high-throughput screening and point-of-use analysis in resource-limited or non-laboratory environments.
Key Features
- Multi-parameter electrochemical platform: Simultaneous or sequential determination of TAC, H2O2 clearance capacity, and customizable redox endpoints via firmware update
- Micro-volume operation: Requires only 5–10 µL of undiluted, unprocessed sample—ideal for precious clinical or field-collected specimens
- Native-state analysis: No chemical modification, pH adjustment, or matrix masking; compatible with colored, turbid, or particulate-containing samples (e.g., fruit juices, algal extracts, plasma)
- Bluetooth-enabled data acquisition: Real-time synchronization with Android tablets, laptops, or smartphones running the BQC Analytical Suite v3.x
- Integrated disposable screen-printed carbon electrodes (SPCEs): Pre-calibrated, single-use sensor strips with batch-specific electrochemical characterization certificates
- Rugged portable architecture: IP54-rated enclosure, rechargeable Li-ion battery (≥8 h continuous operation), and ambient-temperature stability (15–35 °C)
Sample Compatibility & Compliance
The BRS demonstrates broad compatibility across unprocessed liquid matrices without enzymatic pretreatment or solvent extraction. Validated sample types include human plasma, serum, saliva, urine, cell lysates, tissue homogenates, plant hydroalcoholic extracts, seaweed supernatants, fruit juices, fermented beverages, and cosmetic emulsions. Sample preparation is limited to 1:10 dilution in proprietary BQC Electrolyte Buffer (pH 7.4, ionic strength 0.1 M), ensuring consistent double-layer formation and charge-transfer kinetics. The system adheres to ISO/IEC 17025 principles for method validation, with documented linearity (R² ≥ 0.995 over 0–500 µmol Trolox Equivalents/mL), repeatability (CV < 4.2% intra-day, n = 12), and intermediate precision (CV < 5.8% inter-day, n = 3). While not FDA-cleared as an IVD device, its measurement protocol aligns with ASTM E3152-20 (Standard Guide for Electrochemical Sensor Performance Evaluation) and supports GLP-compliant data recording when used with audit-trail-enabled software configurations.
Software & Data Management
The BQC Analytical Suite (v3.2+) provides full instrument control, real-time voltammogram visualization, automatic peak integration, and TAC quantification against Trolox or ascorbic acid calibration standards. All raw current–potential datasets are stored in open-format .CSV and vendor-neutral .MDF4 files, enabling third-party analysis in MATLAB, Python (SciPy), or OriginLab. Software features include electronic signature support, user role-based access control (admin/operator), 21 CFR Part 11–compliant audit trail (timestamped operator ID, parameter changes, result modifications), and automated report generation in PDF/A-2b format. Data export supports LIMS integration via HL7 or RESTful API endpoints, facilitating traceability in regulated QC/QA workflows.
Applications
- Clinical research: Longitudinal monitoring of systemic redox status in metabolic syndrome, neurodegenerative disorders, and aging cohorts
- Nutraceutical development: Rapid screening of polyphenol-rich botanical extracts, probiotic supernatants, and functional food formulations
- Food quality assurance: Shelf-life prediction based on antioxidant decay kinetics in minimally processed produce and dairy alternatives
- Environmental toxicology: Assessment of oxidative stress biomarkers in marine organism homogenates exposed to heavy metals or microplastics
- Phytochemistry: High-throughput phenolic profiling of breeding lines in crop improvement programs without HPLC infrastructure
FAQ
How does the BRS differentiate between low-molecular-weight and protein-bound antioxidants?
The BRS reports total electroactive reducing capacity under defined scan parameters (−0.2 to +1.2 V vs. Ag/AgCl, 50 mV/s). While it does not chemically resolve individual species, differential pulse voltammetry (DPV) mode—available via optional firmware upgrade—enables semi-quantitative deconvolution of overlapping redox peaks based on formal potentials.
Is electrode fouling observed with viscous or protein-rich samples?
SPCEs exhibit minimal passivation due to hydrophilic surface modification and optimized potential windows; routine cleaning cycles (electrochemical regeneration at −0.8 V for 30 s) restore baseline response for >50 measurements per strip.
Can the BRS be validated for regulatory submission (e.g., EFSA health claim dossiers)?
Yes—method validation packages (including specificity, accuracy, robustness, LOD/LOQ) are available upon request and have been accepted in peer-reviewed submissions supporting EFSA Article 13.5 opinions and USDA Organic certification documentation.