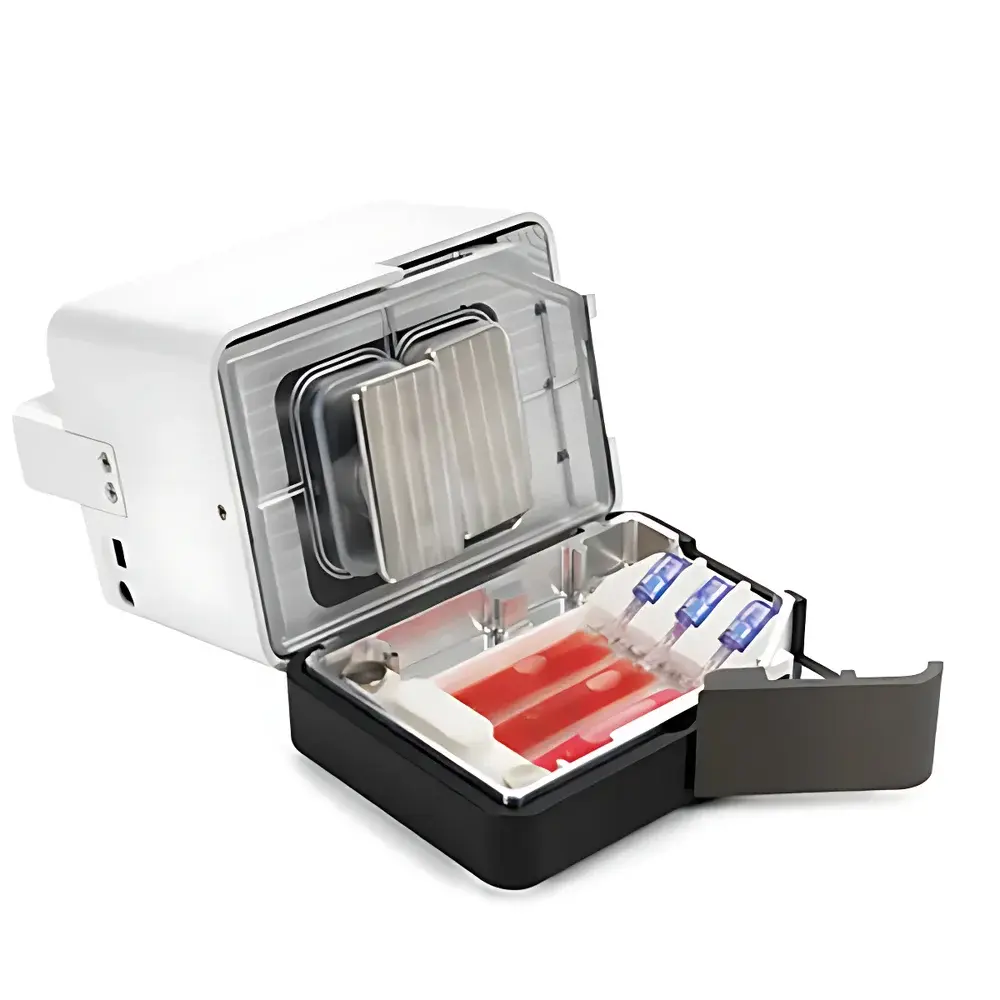
Cytiva VIA Extractor™ Temperature-Controlled High-Performance Tissue Processor
| Brand | Cytiva |
|---|---|
| Origin | United Kingdom |
| Manufacturer Type | Authorized Distributor |
| Product Origin | Imported |
| Model | Omics Bundle |
| Separation Method | Automated |
| Cell Viability (%) | ≥90% |
| Compatibility | Requires Integration with VIA Freeze™ Uno Controlled-Rate Freezer |
| Intended Use | Research Use Only (RUO), Not for Diagnostic Purposes |
Overview
The Cytiva VIA Extractor™ Temperature-Controlled High-Performance Tissue Processor is an engineered solution for reproducible, gentle, and scalable dissociation of solid tissues—including fresh or cryopreserved tumor biopsies, lymphoid organs, and other mammalian tissues—into high-viability single-cell suspensions. Operating on the principle of controlled mechanical agitation combined with programmable thermal regulation, the system enables precise modulation of shear stress, temperature, and incubation time to preserve native cellular integrity, membrane functionality, and intracellular signaling states. Unlike conventional manual enzymatic digestion or aggressive mechanical homogenization—which often induce transcriptional artifacts, surface marker loss, or suboptimal yield—the VIA Extractor™ implements a closed, semi-automated workflow that maintains physiological relevance while delivering consistent input material for downstream single-cell omics applications. Its design aligns with GLP-aligned laboratory practices and supports traceability requirements common in translational research environments.
Key Features
- Gentle, temperature-regulated tissue dissociation optimized for viability retention (≥90%) and minimal perturbation of intracellular biochemistry.
- Closed-system processing using sterile, multi-compartment Omics Sample Bags—reducing cross-contamination risk and enabling parallel processing of up to three samples per run.
- Modular integration with the VIA Freeze™ Uno Controlled-Rate Freezer, leveraging Stirling-cycle refrigeration technology to eliminate liquid nitrogen dependency and ensure precise thermal profiles during both pre-dissociation cooling and real-time process control.
- Standardized, operator-independent protocol execution—minimizing inter-sample variability and enhancing experimental reproducibility across batches and users.
- Configurable processing parameters (temperature, duration, agitation intensity) tailored to tissue type, density, and size via intuitive interface controls.
- Omics Bundle configuration includes the VIA Extractor™ processor, VIA Freeze™ Uno freezer, Omics Clamp for secure bag mounting, and Omics Sample Bags with integrated enzyme delivery ports.
Sample Compatibility & Compliance
The VIA Extractor™ accommodates human and murine solid tissues—including but not limited to tumor resections, spleen, thymus, lung, and liver—with demonstrated performance across FFPE-adjacent fresh-frozen specimens and enzymatically sensitive samples. It is compatible with standard dissociation reagents (e.g., collagenase IV, DNase I, Accutase®) delivered via Omics Injection Syringes through luer-lock–equipped sample bag ports. The system complies with ISO 13485–aligned manufacturing standards for research instrumentation and supports audit-ready documentation workflows. While not cleared for clinical diagnostics, it meets essential requirements for RUO use under FDA 21 CFR Part 11–compatible data management when paired with validated software modules. All consumables are certified endotoxin-free and sterile-filtered, fulfilling ISO 11137 sterility assurance level (SAL) 10⁻⁶ specifications.
Software & Data Management
The VIA Extractor™ operates under firmware-controlled logic synchronized with the VIA Freeze™ Uno’s embedded thermal management system. Process logs—including start/stop timestamps, setpoint temperatures, actual chamber temperatures, agitation cycles, and user ID—are automatically recorded and exportable in CSV format for integration into LIMS or ELN platforms. Audit trail functionality captures all parameter modifications, ensuring alignment with GLP/GMP documentation expectations for preclinical single-cell assay development. No proprietary cloud service is required; local storage and USB-based data retrieval maintain full data sovereignty—a critical consideration for institutions handling sensitive biological datasets.
Applications
- Preparation of high-fidelity single-cell suspensions for scRNA-seq, scATAC-seq, and spatial transcriptomics library construction.
- Generation of viable immune cell isolates from tumor microenvironments for flow cytometry, CyTOF, and functional assays.
- Standardized dissociation workflows in academic core facilities supporting multi-investigator omics consortia.
- Translational biomarker discovery requiring preservation of phospho-epitopes and transient signaling states.
- GMP-adjacent process development for autologous cell therapy manufacturing where consistency and viability are regulatory endpoints.
FAQ
Is the VIA Extractor™ suitable for frozen tissue samples?
Yes—it is validated for use with cryopreserved tissues thawed under controlled conditions prior to loading into the Omics Sample Bag.
Does the system require liquid nitrogen?
No—the integrated VIA Freeze™ Uno utilizes closed-loop Stirling-cycle refrigeration, eliminating reliance on cryogenic gases.
Can I use third-party dissociation enzymes?
Yes, provided they are compatible with the specified pH, osmolarity, and temperature ranges outlined in the Omics Reagent Compatibility Guide.
What is the minimum tissue mass recommended per run?
Optimal performance begins at 25 mg; maximum capacity per bag is 200 mg, depending on tissue density and fibrosis level.
Is remote monitoring supported?
Real-time status indicators are available locally via front-panel display; remote access requires integration with facility-wide IoT infrastructure and is not natively enabled.