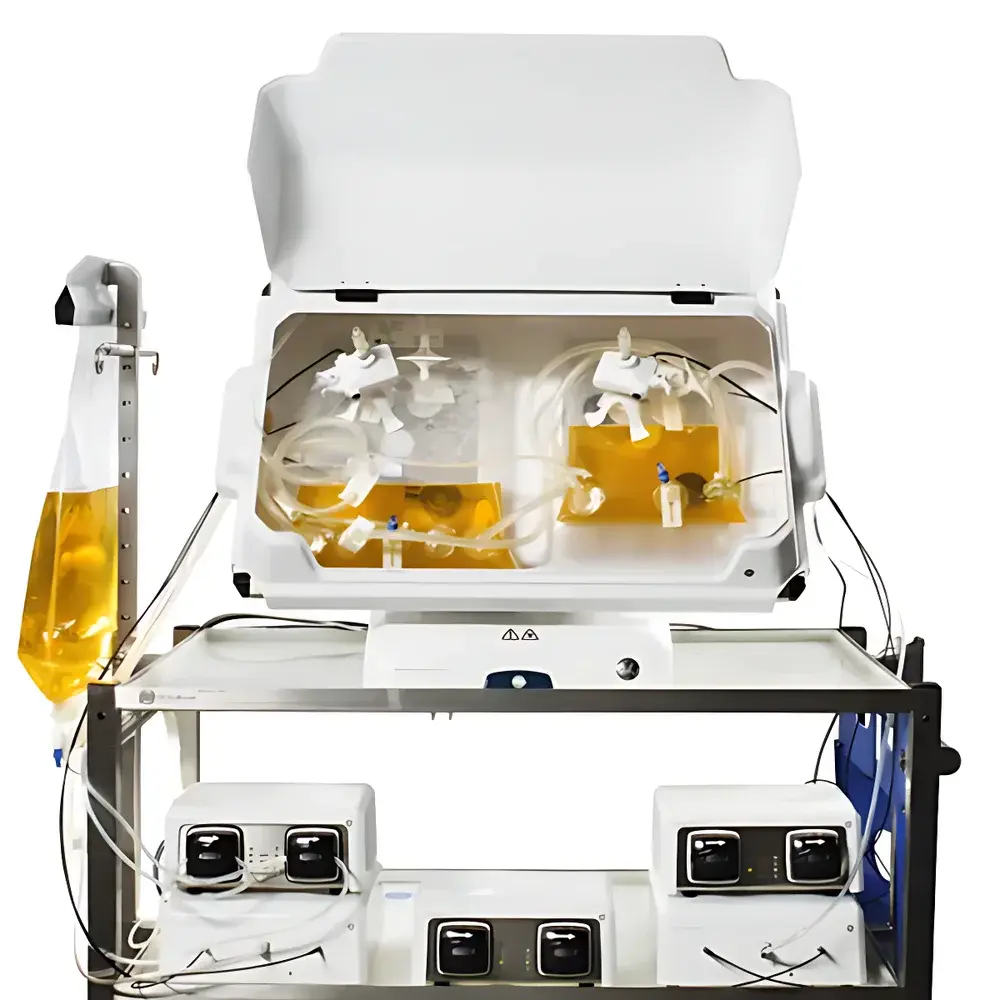
Cytiva ReadyToProcess WAVE 25 Rocking Bioreactor
| Brand | Cytiva |
|---|---|
| Origin | USA |
| Model | ReadyToProcess WAVE 25 |
| Maximum Working Volume | 25 L |
| Drive Mechanism | Motorized Rocking Platform with Adjustable Speed, Angle, and Motion Profile |
| Temperature Control | Integrated Heated Tray with Dual Independent PID Loops (Dual-Mode Operation) |
| Weight Measurement | Built-in Load Cells with Auto-Calibration Capability |
| Gas Transfer | Passive O₂/CO₂ Exchange via Sterile Vent Filters (Heated Exhaust Filter Included) |
| Data Storage | Dual Mirrored SSDs + UNICORN Database Integration |
| Communication Protocol | OPC UA Compliant |
| Compliance | Designed for GMP Environment |
Overview
The Cytiva ReadyToProcess WAVE 25 Rocking Bioreactor is an engineered platform for scalable, single-use mammalian and microbial cell culture under controlled bioprocess conditions. Based on the well-established wave-mixing principle—where gentle, oscillatory motion induces laminar flow and interfacial gas exchange across the liquid–gas boundary—the system enables robust suspension and adherent cell growth in disposable Cellbag™ bioreactor bags without mechanical agitation or sparging. Its rocking mechanism generates predictable hydrodynamic shear profiles, minimizing cellular stress while maintaining homogenous nutrient distribution and efficient CO₂ removal. Designed for process development through clinical manufacturing, the WAVE 25 supports volumes from 1 L to 25 L in a compact footprint, facilitating seamless technology transfer from bench-scale to pilot production. The system operates within ISO Class 5–7 cleanroom-compatible environments and integrates into end-to-end single-use workflows compliant with ICH Q5D, Q5A(R2), and USP guidance for cell-based therapeutics.
Key Features
- Motorized rocking platform with fully adjustable parameters: speed (rpm), tilt angle (°), and acceleration profile—enabling optimization of mixing intensity and mass transfer for diverse cell lines including CHO, HEK293, hybridomas, and insect cells.
- Integrated temperature control via heated aluminum tray with embedded platinum resistance thermometers (Pt100); dual independent PID loops support simultaneous but separate thermal regulation of two Cellbag™ units in dual-culture mode.
- High-precision load cells calibrated to ±0.5% full scale, enabling real-time media weight tracking for feed strategies, perfusion pump calibration, and automated pH/titer compensation routines.
- Ergonomic tilting capability (up to 15°) for simplified sampling, harvest, and bag installation—reducing operator intervention and contamination risk during critical process steps.
- Modular tray design accommodating three bag sizes: 5 L, 10 L, and 25 L; all trays include spring-loaded latching mechanisms and light-blocking lids to protect photosensitive media components and extend optical sensor lifetime.
- Dual mirrored solid-state drives (SSDs) ensure redundant, tamper-resistant storage of all operational logs, setpoints, alarms, and event timestamps—meeting ALCOA+ data integrity requirements.
Sample Compatibility & Compliance
The WAVE 25 is validated for use with Cytiva’s ReadyToProcess Cellbag™ family (including standard, high-density, and perfusion configurations) and compatible with common single-use sensors (e.g., PreSens, Hamilton, Sartorius). It supports integration with external pH, DO, and glucose monitoring systems via analog/digital I/O. From a regulatory perspective, the hardware architecture conforms to IEC 61000-6-2/6-4 electromagnetic compatibility standards and carries CE marking per Machinery Directive 2006/42/EC. When operated with UNICORN 7.0 or later and configured with electronic signatures, audit trails, and role-based access control, the system meets FDA 21 CFR Part 11 and EU Annex 11 expectations for computerized system validation (CSV) in GMP environments. Documentation packages—including DQ/IQ/OQ protocols, risk assessments (FMEA), and change control records—are available upon request.
Software & Data Management
Control and monitoring are managed through Cytiva’s UNICORN software suite, which provides a validated, configurable interface for defining recipes, scheduling events, and visualizing real-time trends. All process data—including rocking parameters, temperature gradients, weight differentials, and alarm histories—are written to encrypted, timestamped files stored locally on the dual SSD array. Post-run, data exports seamlessly to the centralized UNICORN Database Server for long-term archival, cross-batch comparison, and statistical process control (SPC) analysis. OPC UA server functionality allows bidirectional communication with MES, SCADA, and LIMS platforms, supporting Industry 4.0-aligned digital twin implementations. Optional OptiRun service plans include remote diagnostics, predictive maintenance alerts, and lifecycle firmware updates aligned with current regulatory expectations.
Applications
- Monoclonal antibody (mAb) and recombinant protein production in suspension-adapted CHO cells.
- Virus propagation for vaccine manufacturing (e.g., adenovirus, AAV, lentivirus) using serum-free or chemically defined media such as HyClone VaccineXpress™.
- Stem cell expansion and differentiation studies requiring low-shear, physiologically relevant microenvironments.
- Process characterization and scale-down modeling for upstream bioprocess development and comparability studies.
- Perfusion-based continuous culture with integrated pump modules (e.g., ReadyToProcess Pump 25) and inline analytics.
- Support of ADCF (Animal-Derived Component-Free) and fully chemically defined processes compliant with regulatory filing requirements.
FAQ
What is the maximum working volume supported by the ReadyToProcess WAVE 25?
The system supports a maximum working volume of 25 L in its largest Cellbag™ configuration, with validated performance across the full range from 1 L to 25 L.
Does the WAVE 25 support perfusion culture?
Yes—when paired with the ReadyToProcess Pump 25 and compatible tubing sets, the system enables continuous media exchange with automatic weight-based feedback control for stable perfusion rates.
Can temperature be independently controlled for two bags operating simultaneously?
Yes—dual-mode operation allows separate setpoint definition and closed-loop regulation for each bag using dedicated PID controllers and thermal sensors.
Is the system compliant with 21 CFR Part 11?
Full compliance is achieved when deployed with UNICORN software configured with electronic signatures, audit trail logging, and user access management per validated SOPs.
How is gas exchange managed without sparging?
Oxygen transfer and CO₂ removal occur passively across the expanded liquid surface area generated by wave motion, augmented by heated sterile exhaust filters to prevent condensation-induced backpressure.