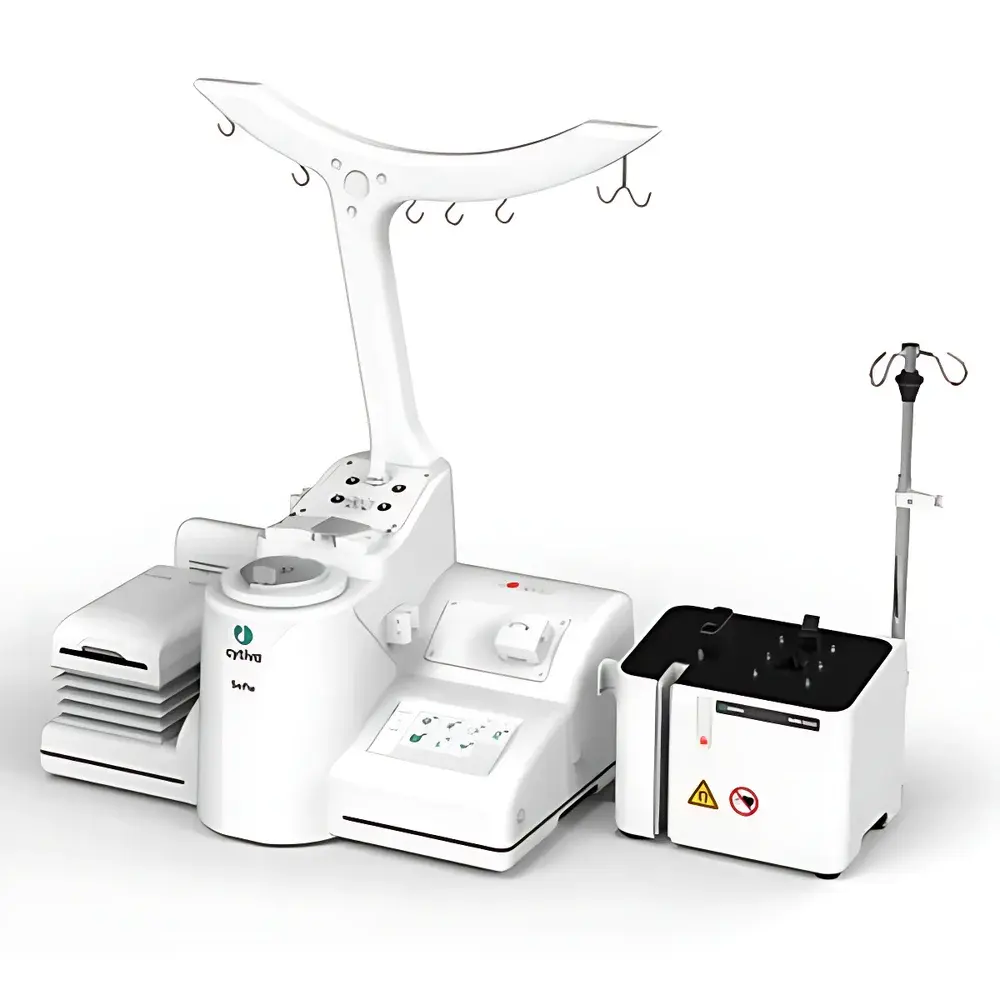
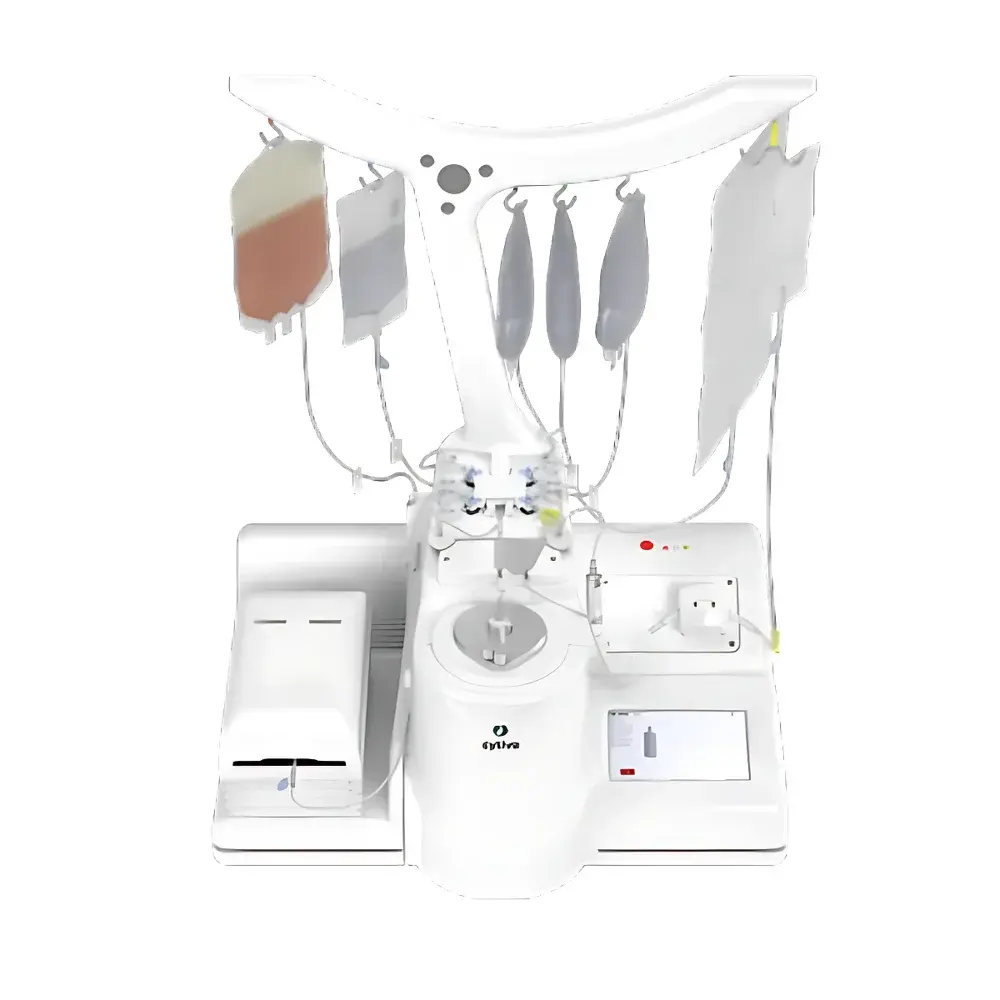
Cytiva Sefia Select™ S-2000 Automated Cell Processing System
| Brand | Cytiva |
|---|---|
| Origin | Switzerland |
| Instrument Model | Sefia S-2000 |
| Type | Fully Automated |
| Regulatory Designation | Import (CE-marked for IVDR/MDR-compliant use in cell therapy manufacturing) |
| Single-Use Kit Compatibility | CT-300.1, CT-400.1 + PB-100.1, CT-200.1, CT-800.1, CT-350.1S |
| Software Integration | Chronicle™ v3.2+ with eSOP/eBMR and 21 CFR Part 11 / EU Annex 11 compliance |
Overview
The Cytiva Sefia Select™ S-2000 Automated Cell Processing System is an integrated, closed-platform instrument engineered for robust, scalable, and regulatory-compliant manufacturing of advanced therapeutic medicinal products (ATMPs), including autologous and allogeneic cell therapies. Based on a dual-module hardware architecture—comprising a processing unit and a control module—the system executes critical unit operations under precisely controlled environmental and fluidic conditions. Its core operational principle relies on programmable peristaltic pumping, pneumatic valve actuation, real-time pressure and temperature sensing, and gravimetric monitoring to ensure process consistency across batch sizes ranging from clinical-scale (1–2 L) to early commercial volumes (up to 10 L). Designed specifically for Good Manufacturing Practice (GMP)-aligned environments, the Sefia Select™ platform supports end-to-end automation of cell isolation, washing, concentration, formulation, and final product dispensing—minimizing manual intervention while maintaining sterility assurance through fully enclosed, single-use fluid paths.
Key Features
- Fully automated, closed-system architecture compliant with ISO 13485 and aligned with ICH Q5A(R2) and Q5D guidance for cell-based therapeutics.
- Dual-hardware configuration enabling parallel or sequential processing of multiple workflows without cross-contamination risk.
- Integrated sensor suite—including conductivity, pH, pressure, temperature, and weight transducers—for real-time process monitoring and adaptive parameter adjustment.
- Modular single-use kits (CT-series) validated for specific applications: PremierCell (density gradient mononuclear cell enrichment), MagnetSelect (magnetic bead-based T-cell isolation), S-Wash (cell wash/concentration up to 1.2 L), FlexCell (harvest/formulation up to 10 L across ≤3 final bags), and ReadySelect (final formulation and cryopreservation of up to 4 doses).
- Pre-configured, user-editable application software modules with configurable hold times, flow rates, centrifugation equivalents (via sedimentation algorithms), and alarm thresholds.
- Touchscreen HMI with contextual workflow guidance, step-by-step visual prompts, and audit-trail-enabled operator actions.
Sample Compatibility & Compliance
The Sefia Select™ S-2000 accommodates fresh or cryopreserved leukapheresis products, peripheral blood mononuclear cells (PBMCs), and expanded T-cell populations within defined viability (>70%) and volume (0.5–12 L) ranges. All contact surfaces are constructed from USP Class VI-certified polymers; all single-use kits undergo gamma irradiation (25–35 kGy) and are certified endotoxin-free (<0.25 EU/mL). The system meets requirements for sterile barrier integrity per ISO 11607-1 and supports validation protocols per ASTM E2897 (sterility testing of closed systems) and ISO 14644-1 Class 5 environments when operated inside isolators or RABS. It is CE-marked under MDR 2017/745 and IVDR 2017/746 for in vitro diagnostic use in cell therapy process monitoring and release testing support.
Software & Data Management
Chronicle™ software serves as the centralized digital backbone for the Sefia Select™ platform. It provides electronic standard operating procedure (eSOP) authoring, version-controlled deployment, and execution logging with full 21 CFR Part 11 and EU Annex 11 compliance—including electronic signatures, audit trails, role-based access control, and immutable data archiving. Each run generates a complete electronic batch manufacturing record (eBMR) containing time-stamped instrument telemetry, operator inputs, kit lot traceability, and deviation flags. Real-time dashboards display key performance indicators (KPIs) such as yield recovery, volume loss, and buffer consumption. Alarm notifications are delivered via SMS and email with configurable escalation paths. Chronicle™ also enables integration with MES and ERP systems via HL7/FHIR-compatible APIs for supply chain synchronization and quality event management.
Applications
- Automated isolation of CD3+/CD4+/CD8+ T cells from leukapheresis material using magnetic nanoparticle labeling and column-free separation.
- Scalable washing and concentration of CAR-T or TCR-engineered cells prior to cryopreservation or infusion.
- Formulation of final drug product into multiple cryobags or infusion bags with precise cell density, volume, and excipient concentration control.
- Process development and tech transfer support, including comparative studies between manual and automated workflows per ISCT guidelines.
- GMP production of investigational new drug (IND) and marketing authorization application (MAA) batches under FDA, EMA, and PMDA oversight.
FAQ
What regulatory standards does the Sefia Select™ S-2000 support?
It is designed to meet GMP requirements per 21 CFR Parts 210/211, EU GMP Annex 1, and ISO 13485:2016. Chronicle™ software ensures compliance with 21 CFR Part 11 and EU Annex 11 for electronic records and signatures.
Can the system be validated for commercial manufacturing?
Yes—Cytiva provides qualification documentation packages (IQ/OQ/PQ), URS templates, and protocol support for site-specific validation aligned with PDA Technical Report No. 57 and ISCT Best Practices.
Are single-use kits supplied sterile and ready-to-use?
All CT-series kits are gamma-irradiated, individually sealed, and supplied with CoA, CoC, and bioburden/endotoxin test reports.
How is process traceability maintained across multiple instruments?
Chronicle™ enables centralized parameter management, synchronized firmware updates, and aggregated analytics across distributed Sefia Select™ installations via secure cloud or on-premise deployment.
Does the system support remote monitoring and troubleshooting?
Yes—through Chronicle™’s secure web interface, authorized users can view live status, historical runs, and diagnostic logs; Cytiva Field Service Engineers may initiate remote diagnostics with customer consent.