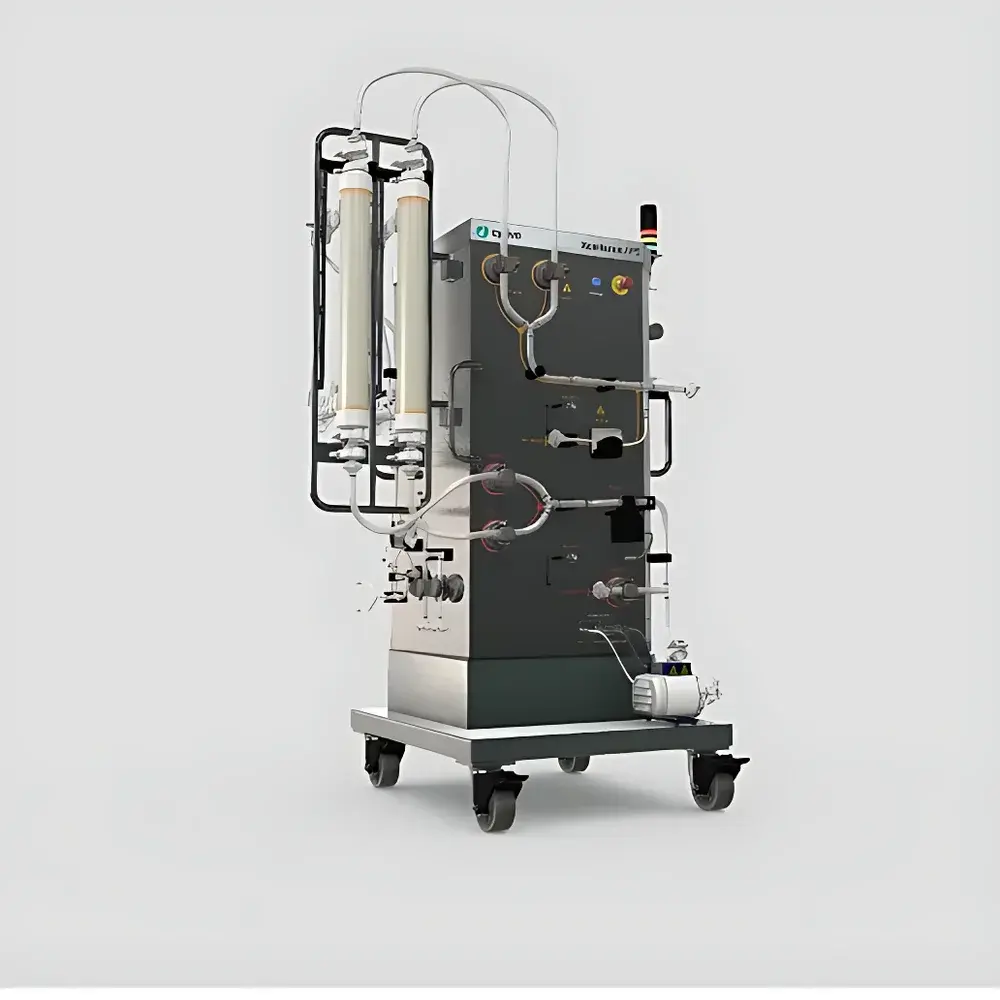
Cytiva Xcellerex APS Automated Perfusion System
| Brand | Cytiva |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | Xcellerex APS |
| Pricing | Available Upon Request |
Overview
The Cytiva Xcellerex APS Automated Perfusion System is an integrated, programmable perfusion platform engineered for high-reproducibility mammalian cell culture operations under cGMP-compliant conditions. Based on tangential flow filtration (TFF) and automated diaphragm pump control, the APS delivers precise, continuous medium exchange while retaining viable cells within the bioreactor—enabling extended culture durations, elevated viable cell densities (>20 × 10⁶ cells/mL), and consistent product titer accumulation over weeks. Designed specifically to interface with the Xcellerex XDR single-use bioreactor series, the APS eliminates manual intervention in filter switching, pump priming, and perfusion rate modulation—reducing operator-dependent variability and supporting robust process transfer from lab-scale development (e.g., 3 L–50 L XDR) to clinical manufacturing (up to 200 L). Its architecture supports both fed-batch intensification and true continuous perfusion modes, making it suitable for early-phase clinical material production, process characterization studies, and platform-based mAb or recombinant protein manufacturing.
Key Features
- Automated, software-coordinated filter switching between primary and backup TFF cassettes—ensuring uninterrupted perfusion during maintenance or fouling events
- Integrated peristaltic and diaphragm pump modules with real-time flow rate feedback control (±2% accuracy across 0.1–5 L/h range)
- Native communication via OPC UA and Modbus TCP protocols for seamless integration into DeltaV, DCS, or MES environments
- Pre-validated control logic compliant with ISA-88 and ISA-95 batch execution standards
- Touchscreen HMI with audit-trail-enabled user access levels (Operator, Technician, Administrator) meeting FDA 21 CFR Part 11 requirements
- Modular design allowing scalability from benchtop (XDR-3/XDR-10) to clinical-scale (XDR-50/XDR-200) bioreactors without hardware reconfiguration
Sample Compatibility & Compliance
The APS is validated for use with standard Xcellerex TFF cassettes (0.5 m² and 1.0 m² surface area, 0.2 µm polyethersulfone membranes) and compatible with common CHO, HEK293, and CAP-T cell lines cultured in chemically defined, animal-component-free media. All wetted materials comply with USP Class VI biological safety testing and meet ISO 10993-5 cytotoxicity requirements. The system supports installation qualification (IQ), operational qualification (OQ), and performance qualification (PQ) documentation packages aligned with ICH Q5D, Q5A(R2), and Annex 1 (EU GMP) expectations. Full traceability of all critical process parameters—including perfusion rate, harvest volume, cell viability (via integrated inline capacitance probe option), and pressure differentials—is retained for GLP/GMP audits.
Software & Data Management
Control and monitoring are executed through the embedded Xcellerex Control Software v4.x, which provides recipe-driven operation, dynamic setpoint adjustment, and automated log generation. All process data—including timestamps, operator actions, alarm events, and parameter deviations—are stored locally in encrypted SQLite databases and synchronized to networked file servers or cloud-based LIMS via configurable SFTP or HTTPS endpoints. Electronic signatures, change history tracking, and configurable alarm escalation (email/SMS) are enabled by default. Raw data exports adhere to ASTM E2500-13 guidelines and support CSV, PDF, and ASAM-ODS formats for regulatory submission readiness.
Applications
- Process development and optimization of perfusion-based mAb production in early-phase clinical supply (Phase I/II)
- Long-term stability assessment of high-expressing clones under continuous culture conditions
- Generation of high-titer, low-aggregate drug substance for formulation screening
- Comparative evaluation against alternating tangential flow (ATF) systems in side-by-side bioreactor runs
- Scale-down modeling for commercial-scale perfusion processes using XDR-3 and XDR-10 platforms
- Support of Quality-by-Design (QbD) initiatives through Design of Experiments (DoE)-driven perfusion parameter mapping
FAQ
Can the APS be used independently of the Xcellerex XDR bioreactor?
Yes—the APS includes standalone control firmware and can be interfaced with third-party bioreactors via analog/digital I/O or industrial Ethernet, though full automation (e.g., automatic bioreactor weight-based feed initiation) requires XDR-native integration.
Does the system support remote monitoring and troubleshooting?
Yes—remote desktop access (with customer-defined firewall policies) and secure VNC-based diagnostics are available through Cytiva’s Global Service Network; all connections comply with ISO/IEC 27001-certified cybersecurity protocols.
What validation support is provided for GMP manufacturing?
Cytiva supplies a comprehensive validation toolkit including URS templates, FAT/SAT protocols, IQ/OQ/PQ test scripts, and raw data summary reports—all aligned with ISPE Baseline Guide Volume 5 (Biotechnology) and PIC/S PI 007-6.