DHS C1/C4 Air-Bath Cell Thawing Instrument
| Brand | DHS |
|---|---|
| Origin | Tianjin, China |
| Manufacturer Type | Authorized Distributor |
| Instrument Category | Dry-Phase Thawing System |
| Model | C1 (Single-Position) / C4 (Four-Position) |
| Temperature Range | 35–40 °C |
| Temperature Control Accuracy | ±0.5 °C |
| Compatible Vials | 2 mL Internal Thread, External Thread, and CBS-Standard Cryovials |
| Regulatory Design Basis | Engineered for GLP/GMP-aligned workflows |
Overview
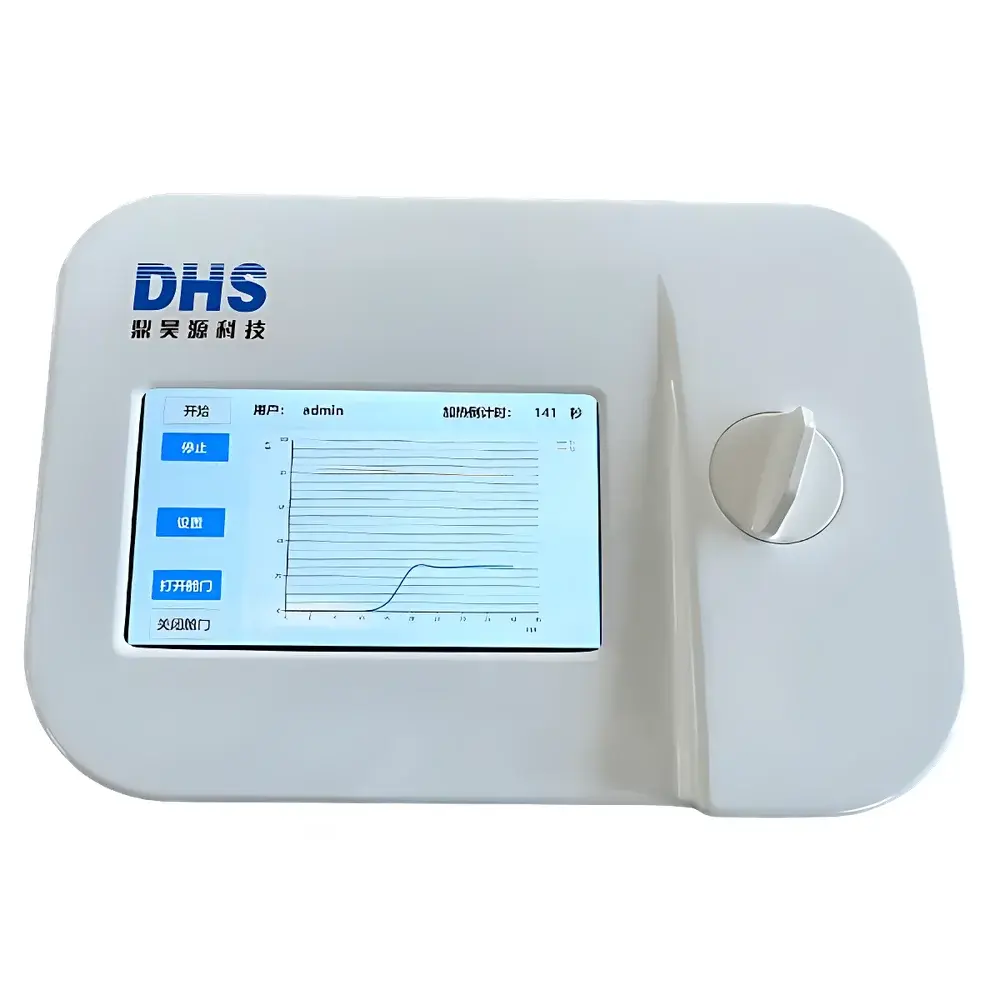
The DHS C1 and C4 Air-Bath Cell Thawing Instruments are precision-engineered dry-phase thawing systems designed to replace conventional water-bath methods in cell therapy manufacturing, biopharmaceutical development, and academic research laboratories. Unlike immersion-based thawing—where uncontrolled thermal gradients, microbial contamination risks, and operator-dependent variability compromise process consistency—the C1/C4 platforms employ regulated convective air heating combined with real-time thermal monitoring to deliver reproducible, traceable, and contamination-free cell recovery. The core principle relies on controlled convective heat transfer within a calibrated air chamber, synchronized with dual-sensor thermal feedback: one probe monitors the heating block temperature, while a second high-response thermistor embedded in the vial holder surface tracks the actual thermal profile at the cryovial wall interface. This dual-curve acquisition enables precise detection of the phase-transition inflection point (solid-to-liquid transition), allowing algorithmic termination of the thaw cycle at optimal biological viability thresholds—typically aligned with the critical −10 °C to 0 °C range where ice recrystallization damage is minimized.
Key Features
- Dual-channel real-time temperature logging: simultaneous visualization of heating block temperature and cryovial wall temperature curves on an integrated high-resolution LCD display
- Programmable thaw profiles with user-defined setpoints across the 35–40 °C operational range, compliant with ISO 13485 and ASTM F2657-21 guidance for cryopreserved cell product handling
- ±0.5 °C temperature control accuracy verified per IEC 61000-4-3 electromagnetic compatibility and thermal stability testing protocols
- Ergonomic, compact footprint design suitable for biosafety cabinets (BSCs), cleanroom Grade A/B environments, and GMP manufacturing suites
- Non-invasive, dry-phase operation eliminates waterborne endotoxin exposure, condensation-related cross-contamination, and vial labeling degradation
- Modular configuration: C1 supports single 2 mL cryovial processing; C4 accommodates four independent 2 mL vials with individual thermal monitoring per position
Sample Compatibility & Compliance
The C1/C4 instruments accept all industry-standard 2 mL cryogenic vials—including internal-thread (e.g., Nunc™, Corning®), external-thread (e.g., Greiner Bio-One™), and CBS-style formats—without adapters or mechanical modification. Thermal uniformity validation confirms ≤±0.8 °C spatial deviation across the entire vial contact zone under steady-state conditions. Designed for alignment with regulatory expectations in cell and gene therapy (CGT) manufacturing, the system supports audit-ready documentation: timestamped thermal logs, operator ID tagging, and configurable electronic signatures compatible with FDA 21 CFR Part 11 requirements when integrated with validated LIMS or ELN platforms. All firmware and calibration records adhere to ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available).
Software & Data Management
While the base C1/C4 units operate via embedded microcontroller logic with local display and manual start/stop controls, optional USB-C data export enables CSV-formatted thermal datasets for offline analysis in MATLAB®, Python (NumPy/Pandas), or JMP®. For enterprise deployment, the instrument’s RS-485 interface supports integration into centralized SCADA or MES systems using Modbus RTU protocol. Audit trail functionality includes automatic capture of calibration events, error codes, ambient temperature fluctuations, and user-initiated parameter changes—each with cryptographic timestamping and immutable storage. Data integrity safeguards meet ICH GCP and EU Annex 11 criteria for electronic record retention in clinical-grade cell processing facilities.
Applications
- GMP-compliant thawing of CAR-T, NK, TIL, and MSC products prior to infusion or expansion
- QC release testing of cryopreserved primary cells in contract development and manufacturing organizations (CDMOs)
- Reproducible thawing for high-content screening (HCS) assays requiring batch-to-batch viability consistency
- Standardized recovery of reference cell lines (e.g., ATCC®, ECACC®) in regulatory submission packages
- Process characterization studies supporting Q5A(R2) comparability assessments during cell line or formulation changes
FAQ
Does the C1/C4 require water or consumables for operation?
No—this is a dry-phase, air-convection system. No water, oil, or disposable heating elements are required.
Can the instrument be calibrated in-house?
Yes—users may perform routine verification using NIST-traceable Class A PT100 probes; full calibration requires DHS-certified service engineers and documented calibration certificates.
Is the system compatible with automated liquid handlers?
The C4 model features programmable relay outputs and TTL-level trigger signals for synchronization with robotic arms or track-based automation systems.
What regulatory standards does the C1/C4 support?
It supports alignment with ISO 13485:2016, ASTM F2657-21, USP <1043>, and ICH Q5A(R2); full compliance depends on site-specific validation protocols and SOP implementation.
How is data security ensured during electronic record transfer?
USB exports use encrypted file headers; network-enabled variants implement TLS 1.2 encryption and role-based access control (RBAC) for remote data retrieval.