FT-ZW108 African Swine Fever Real-Time Fluorescent Quantitative PCR Analyzer
| Brand | Fengtu |
|---|---|
| Origin | Shandong, China |
| Model | FT-ZW108 |
| Channel Configuration | Single-channel (8-well) |
| Temperature Range | 4–99 °C |
| Thermal Accuracy | ±0.1 °C |
| Temperature Uniformity | ≤±0.25 °C |
| Max Heating/Cooling Rate | 5.5 °C/s / 5 °C/s |
| Excitation Wavelength | 460–480 nm |
| Emission Detection Range | 505–525 nm |
| Sensitivity | Single-copy DNA detection |
| Dynamic Range | 10¹–10¹⁰ copies |
| Linearity (R²) | ≥0.999 |
| Inter-well CV | <1% |
| Supported Chemistries | FAM, SYBR Green I |
| Sample Format | 0.2 mL tubes or 8-tube strips |
| Power Supply | 220 V AC, max 550 W |
| Dimensions | 320 × 314 × 206 mm |
| Weight | 4.2 kg |
| Operating System | Embedded Android (dual-language UI) |
| Data Export | CSV, PDF, Excel |
| Connectivity | USB, Wi-Fi |
| Noise Level | ≤60 dB(A) |
Overview
The FT-ZW108 African Swine Fever Real-Time Fluorescent Quantitative PCR Analyzer is a compact, field-deployable molecular diagnostic instrument engineered for nucleic acid amplification and quantification using real-time polymerase chain reaction (qPCR) technology. It operates on the principle of sequence-specific fluorescent probe hybridization (e.g., hydrolysis probes) or intercalating dye-based detection (e.g., SYBR Green I), enabling precise endpoint and kinetic monitoring of amplicon accumulation during thermal cycling. Designed in strict alignment with regulatory requirements for veterinary diagnostics in China—including compliance with Ministry of Agriculture and Rural Affairs (MARA) approval mandates for ASF virus detection kits—the FT-ZW108 supports validated assay protocols for African swine fever virus (ASFV) genomic DNA detection at biosafety level 2 (BSL-2) compatible settings. Its portable form factor (4.2 kg), integrated Android OS, and dual-mode operation—standalone touchscreen control or PC-linked software analysis—make it suitable for both fixed laboratory environments and mobile surveillance units deployed at farms, slaughterhouses, border inspection posts, and regional animal health laboratories.
Key Features
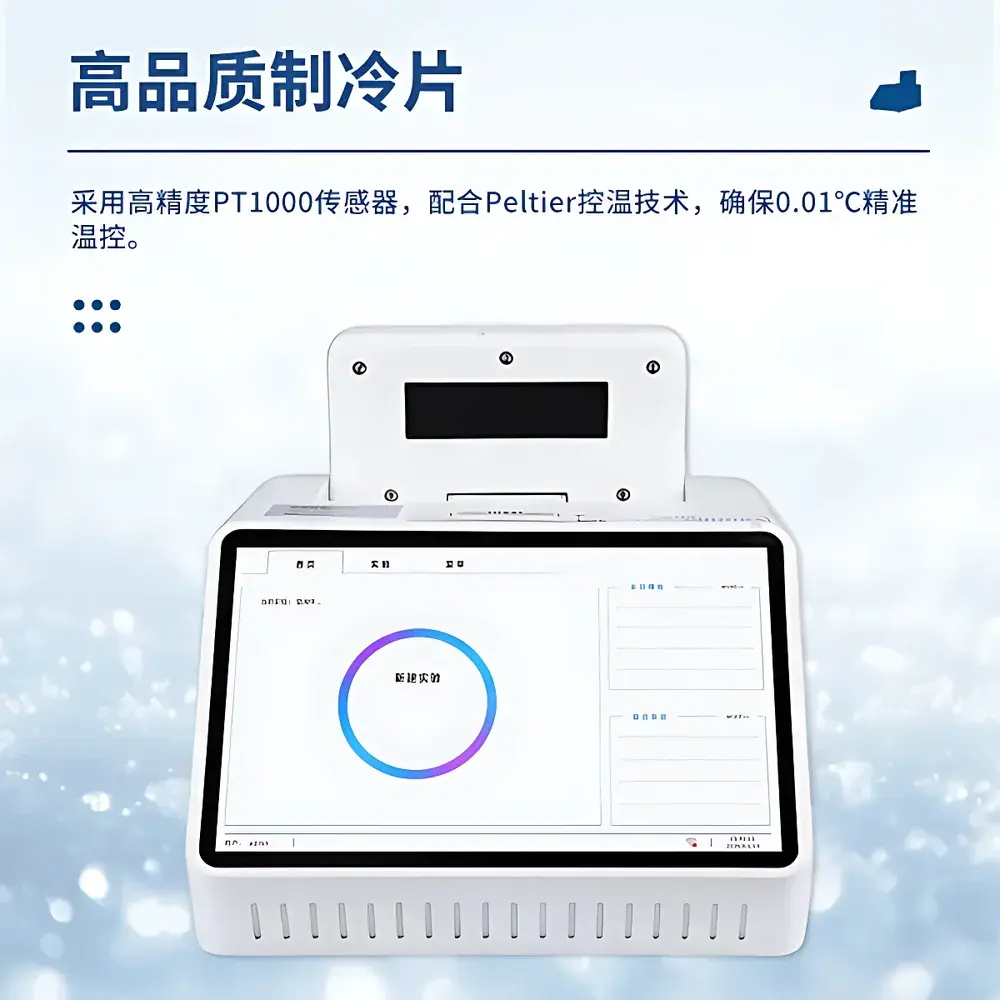
- High-precision thermocycling system utilizing industrial-grade Peltier modules and PT1000 temperature sensors, delivering ±0.1 °C thermal accuracy and ≤±0.25 °C well-to-well uniformity across all 8 reaction positions.
- Optimized optical architecture featuring a single high-intensity blue LED excitation source (460–480 nm) and a matched photodiode detector (505–525 nm), eliminating need for optical calibration and ensuring long-term signal stability over >10,000 cycles.
- Robust mechanical design with corrosion-resistant aluminum sample block, fully sealed thermal chamber, and condensation-resistant housing to prevent moisture ingress into critical electronics—extending mean time between failures (MTBF) and reducing maintenance frequency.
- Embedded Android-based operating system with intuitive graphical interface, supporting real-time curve visualization, automatic baseline/threshold determination, and multi-experiment concurrent data review without interrupting active runs.
- Comprehensive data management suite enabling export to CSV, PDF, and Excel formats; direct printing via USB thermal, Bluetooth, or A4 printers; and secure wireless transfer to local servers or cloud repositories compliant with internal IT governance policies.
Sample Compatibility & Compliance
The FT-ZW108 accepts standard 0.2 mL single tubes and 8-tube strips, accommodating reaction volumes from 15 to 100 μL. It is fully compatible with MARA-approved ASFV detection kits—including fluorescent qPCR reagent sets based on FAM-labeled probes—and supports open-platform usage with third-party master mixes and primers conforming to ISO/IEC 17025 method validation guidelines. All assays performed on this platform are traceable to certified reference materials and meet analytical performance criteria defined in GB/T 38928–2020 (Chinese national standard for ASFV nucleic acid detection). While not pre-certified under EU IVDR or USDA APHIS frameworks, its technical specifications align with core functional requirements outlined in OIE Terrestrial Manual Chapter 2.8.12 for ASF diagnosis and support GLP-aligned documentation practices including audit trails, user access logs, and electronic signature readiness per FDA 21 CFR Part 11 principles.
Software & Data Management
The onboard software provides full-cycle experimental control—from protocol setup and run initiation to post-acquisition analysis—with features including TouchDown cycling, HRM (High-Resolution Melting) capability, and group-wise comparative quantification across batches. Data integrity safeguards include automatic timestamping, fault logging with root-cause diagnostics, and interruption-resume functionality. The system supports dual-language UI (English/Chinese), customizable report templates, and batch-level metadata tagging for sample tracking. Exported datasets retain full experimental context: thermal profiles, fluorescence kinetics, Ct values, efficiency calculations, and amplification curves—structured for downstream integration with LIMS or ELN platforms. No proprietary file locking is applied; raw fluorescence intensity matrices (.csv) and processed results (.pdf) remain fully accessible and interoperable.
Applications
The FT-ZW108 serves as a primary tool for rapid, on-site confirmation of ASFV infection in porcine tissue, blood, serum, oral fluids, and environmental swabs. Its validated performance extends to differential detection of other economically significant swine pathogens—including Classical Swine Fever Virus (CSFV), Porcine Reproductive and Respiratory Syndrome Virus (PRRSV), Pseudorabies Virus (PRV), and Porcine Circovirus Type 2 (PCV2)—as well as avian influenza virus (AIV) in poultry samples. Regulatory end users—including provincial animal husbandry bureaus, customs inspection agencies, and food safety authorities—leverage its portability and MARA-compliant assay compatibility for outbreak response, routine surveillance, and pre-slaughter screening. In research contexts, it supports assay development, limit-of-detection studies, and inter-laboratory reproducibility assessments under controlled environmental conditions (18–35 °C ambient, ≤85% RH).
FAQ
Is the FT-ZW108 approved by MARA for official ASF testing?
Yes—the instrument is intended for use exclusively with MARA-registered ASFV qPCR kits, fulfilling the mandatory equipment requirement stipulated in Notice No. 272 (2020) of the Ministry of Agriculture and Rural Affairs.
Can the FT-ZW108 perform multiplex detection?
No—it is configured as a single-channel analyzer optimized for FAM/SYBR Green I detection; multiplexing requires instrumentation with ≥2 optical channels and corresponding filter sets.
What is the recommended calibration interval?
Thermal and optical calibration is factory-performed and stable for ≥3 years under normal operating conditions; verification checks using NIST-traceable temperature standards are advised annually.
Does the system support remote diagnostics or firmware updates?
Yes—Wi-Fi connectivity enables secure OTA (over-the-air) firmware upgrades and remote technical support sessions initiated via encrypted session tokens.
Are consumables such as reaction tubes or seals proprietary?
No—all components adhere to ANSI/SBS microplate standards; standard 0.2 mL PCR tubes and 8-well strips from major global suppliers are fully compatible.