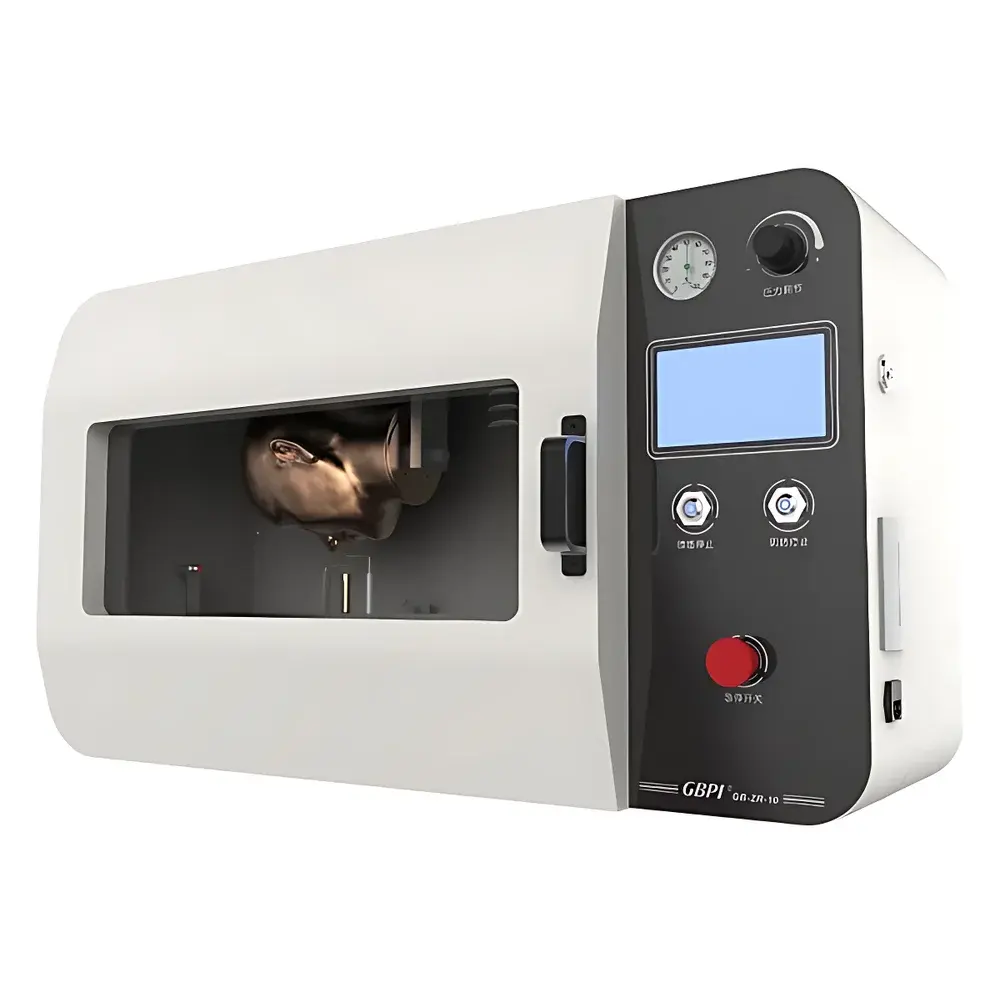
GBPI GB-ZR10A Medical Mask Flame Resistance Tester
| Brand | GBPI |
|---|---|
| Model | GB-ZR10A |
| Origin | Guangdong, China |
| Manufacturer Type | Direct Manufacturer |
| Compliance Standards | GB 19083–2010, YY 0469–2011, GB 2626–2006, GB 19083–2003 |
| Headform Linear Speed | (60 ± 5) mm/s |
| Flame Height | Adjustable to (40 ± 4) mm |
| Flame Tip-to-Headform Distance | (20 ± 2) mm |
| Flame Temperature at 20 mm Above Burner Tip | (800 ± 50) °C |
| Burner Vertical Adjustment Range | 30 mm |
| Headform Wall Thickness | 2–3 mm (flame-resistant stainless steel) |
| Timer Range | 0–9999.9 s, Resolution: ±0.1 s |
| Power Supply | AC 220 V, 50 Hz, 50 W |
| Dimensions (L×W×H) | 970 × 550 × 350 mm |
| Gas Interface | Propane or Liquefied Petroleum Gas (LPG) |
Overview
The GBPI GB-ZR10A Medical Mask Flame Resistance Tester is an engineered test system designed specifically for evaluating the flammability behavior of medical face masks under controlled flame exposure conditions. It operates on the principle of standardized vertical flame impingement, where a conditioned mask specimen is subjected to a defined propane flame at a fixed geometry and velocity—mimicking real-world ignition scenarios such as proximity to surgical lights or electrosurgical units. The instrument implements a precise headform-based mounting configuration, ensuring anatomically relevant orientation and tension during testing. Its core function is to quantify two critical fire response parameters: afterflame time (also known as *continuing combustion time*) and afterglow time (*smoldering time*), both measured with millisecond-level resolution in accordance with national regulatory frameworks governing personal protective equipment (PPE) in healthcare settings.
Key Features
- Integrated 7-inch industrial-grade touchscreen HMI interface enabling intuitive test setup, real-time parameter monitoring, and one-touch start/stop functionality.
- Anatomically contoured stainless-steel headform (2–3 mm wall thickness) with certified flame resistance, replicating human facial contours to ensure realistic mask drape, strain distribution, and flame interaction dynamics.
- Precision-adjustable burner assembly with 30 mm vertical travel range, facilitating repeatable positioning relative to the mask’s lowest point per GB 19083–2010 Clause 5.10.2.
- Automated burner positioning and ignition sequence synchronized with headform motion, eliminating manual alignment variability and operator-dependent timing errors.
- Dual-channel high-resolution digital timer (0–9999.9 s, ±0.1 s accuracy) independently logging afterflame and afterglow durations; data automatically timestamped and stored in non-volatile memory.
- Embedded K-type thermocouple probe calibrated to measure flame temperature at 20 mm above burner tip, continuously displayed on-screen to verify thermal compliance prior to each test cycle.
- Standardized gas supply interface compatible with commercial propane or LPG cylinders, equipped with integrated pressure regulation and leak-tested delivery lines meeting ISO 4126 safety requirements.
- Self-contained control architecture with embedded logic for sequence validation—e.g., verifying flame stabilization before headform movement initiation—to enforce procedural fidelity across operators and laboratories.
Sample Compatibility & Compliance
The GB-ZR10A accommodates flat-fold, duckbill, and molded cup-style medical masks—including surgical masks, N95 respirators, and KN95 equivalents—without requiring custom fixtures. Specimens are secured directly onto the headform using standardized clamping force protocols aligned with YY 0469–2011 Annex B. The system fully satisfies mandatory testing requirements outlined in GB 19083–2010 (Technical Requirements for Medical Protective Masks), YY 0469–2011 (Surgical Masks), GB 2626–2006 (Respiratory Protective Devices), and GB 19083–2003 (earlier edition). All mechanical tolerances—including headform curvature radius, flame standoff distance, and linear translation speed—are traceably verified against national metrological standards. Routine verification procedures support GLP-compliant documentation for internal quality audits and regulatory submissions.
Software & Data Management
Test data—including timestamps, flame temperature readings, afterflame/afterglow values, operator ID, and environmental notes—is stored locally in CSV format with automatic file naming (e.g., “ZR10A_20240522_142301.csv”). The system supports USB export for integration into laboratory information management systems (LIMS) or statistical process control (SPC) platforms. Audit trails record all parameter modifications, test initiations, and firmware updates in chronological order—enabling full traceability required under ISO/IEC 17025:2017 Clause 7.7 and FDA 21 CFR Part 11-aligned environments when paired with user-access controls. No cloud connectivity or remote access capabilities are implemented, preserving data sovereignty and network security integrity.
Applications
This instrument serves quality assurance laboratories in medical device manufacturing, third-party certification bodies (e.g., CNAS-accredited test labs), and hospital central sterile supply departments conducting incoming PPE verification. It supports R&D validation of novel mask materials—including melt-blown polypropylene composites, flame-retardant-treated nonwovens, and multi-layer laminates—by quantifying comparative flame propagation thresholds. In regulatory contexts, test reports generated by the GB-ZR10A constitute primary evidence for CE marking technical files (under MDR Annex II), NMPA registration dossiers, and export compliance with ASEAN Medical Device Directive (AMDD) requirements. Routine use also fulfills periodic requalification mandates stipulated in GB/T 19001–2016 Clause 8.5.1 for production process stability monitoring.
FAQ
What flame fuel types are supported?
Propane and liquefied petroleum gas (LPG) are validated fuel sources; natural gas is not recommended due to inconsistent stoichiometric combustion profiles.
Is calibration certification included with shipment?
Yes—a factory-issued calibration report traceable to CNAS-accredited reference standards is provided, covering timer accuracy, thermocouple linearity, and linear motion encoder performance.
Can the headform be replaced with alternative anthropomorphic forms?
No—the headform geometry is fixed per GB 19083–2010 Figure 1 specifications; substitution would invalidate compliance with cited standards.
Does the system meet electromagnetic compatibility (EMC) requirements for clinical environments?
It complies with GB/T 18268.1–2010 (IEC 61326-1) Class B emission limits and immunity thresholds appropriate for laboratory and low-interference hospital utility rooms.
How frequently should operational verification be performed?
Daily pre-test verification of flame height, tip temperature, and headform speed is mandated; full calibration is recommended every 12 months or after 500 test cycles, whichever occurs first.