HOGON MT-V6 Color-Enabled Automatic Potentiometric Titrator
| Brand | HOGON |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Direct Manufacturer |
| Instrument Category | Domestic (China-Made) |
| Model | MT-V6 Color Titrator |
| Instrument Type | General-Purpose Potentiometric Titrator |
| Application Scope | Universal |
| pH Measurement Range | 0.000–14.000 |
| pH Resolution | 0.001 |
| Meter Accuracy | ±0.1 mV |
| Meter Resolution | 0.1 mV |
| Burette Precision | 20 mL standard burette / 1 µL |
| Potential Range | ±2000 mV |
| Potential Resolution | 0.1 mV |
Overview
The HOGON MT-V6 Color-Enabled Automatic Potentiometric Titrator is a dual-mode analytical platform engineered for laboratories requiring both electrochemical endpoint detection and optical colorimetric endpoint recognition within a single, unified instrument architecture. It operates on the fundamental principles of potentiometric titration—measuring potential difference across an indicator/reference electrode pair as titrant is incrementally added—and extends this capability with integrated visible-spectrum photometric sensing to detect discrete chromatic transitions (e.g., red-to-blue, yellow-to-purple) associated with complexometric, redox, or acid-base reactions where conventional potentiometric inflection may be shallow or ambiguous. This hybrid detection strategy conforms to ASTM E2087–22 (Standard Guide for Potentiometric Titration) and supports method validation under ISO/IEC 17025:2017 requirements for measurement uncertainty estimation. Designed for trace-to-bulk concentration analysis (ppm to % w/v), the MT-V6 meets core functional demands in regulated environments—including pharmaceutical QC, food safety testing, and environmental compliance labs—where orthogonal endpoint confirmation strengthens data integrity and audit readiness.
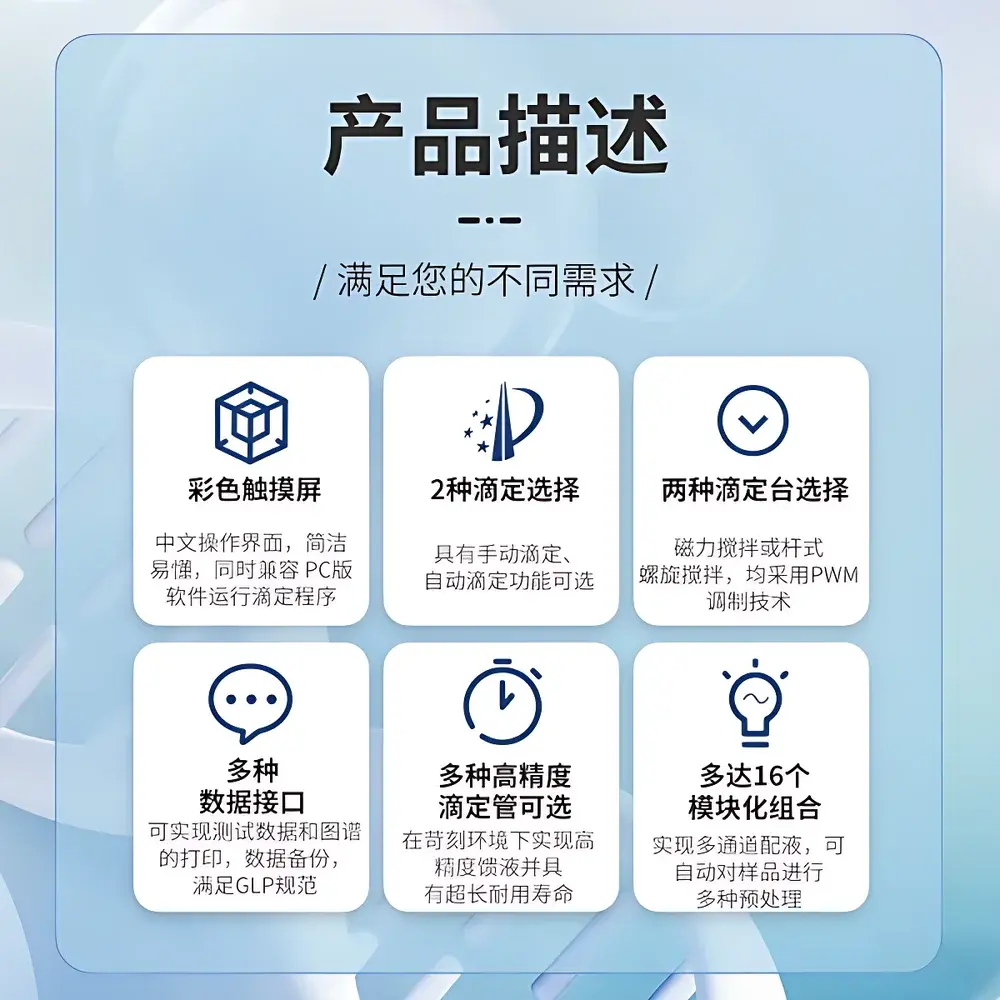
Key Features
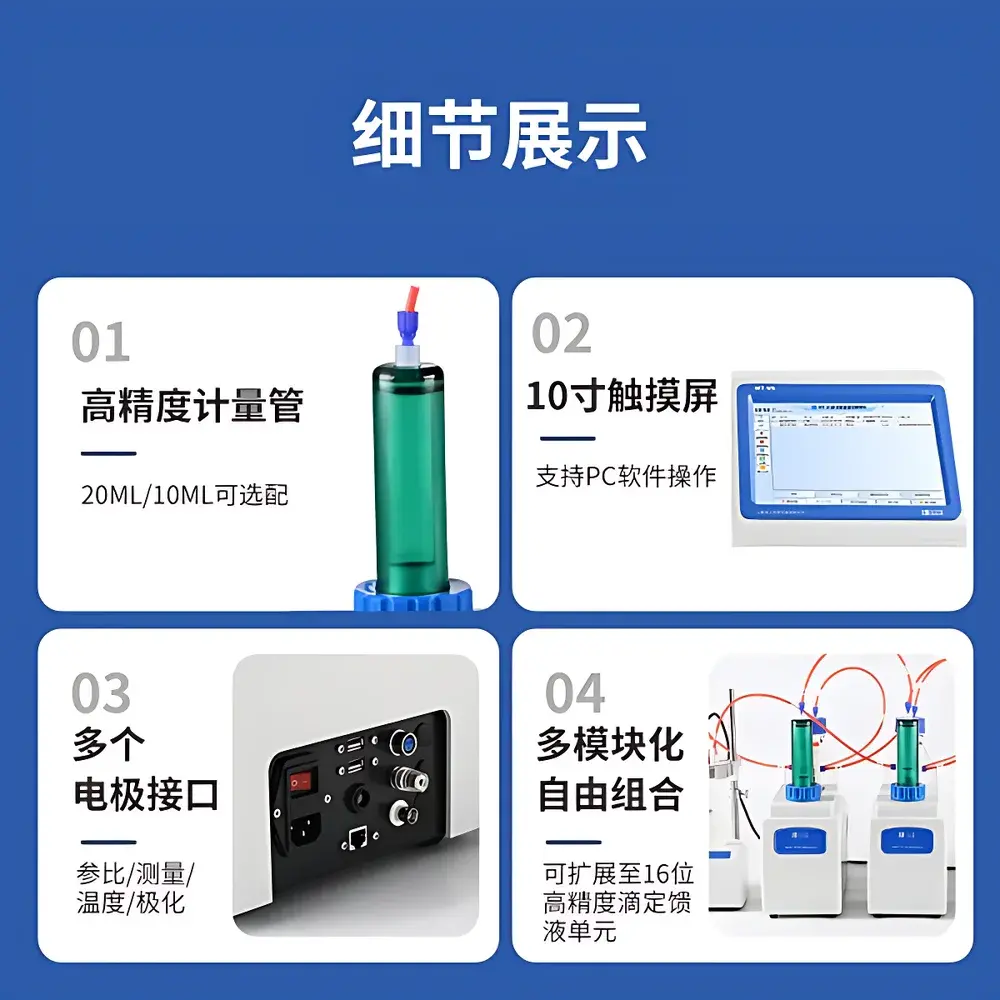
- Dual-Mode Endpoint Detection: Simultaneous real-time monitoring of electrode potential (±2000 mV, 0.1 mV resolution) and optical absorbance/reflectance changes at user-defined wavelengths (400–700 nm), enabling robust endpoint discrimination in EDTA titrations of Cu²⁺/Fe³⁺ or iodometric determinations.
- Modular Multi-Channel Architecture: Supports up to 16 independently configurable modules—including sample autosamplers, reagent dispensers, temperature-controlled reaction cells, and auxiliary burettes—allowing parallel processing of heterogeneous sample sets without cross-contamination.
- High-Precision Fluid Handling: Standard 20 mL glass burette with 1 µL dosing resolution; optional 10 mL burette achieves 0.5 µL resolution for micro-volume applications (e.g., high-purity API assays). All burettes feature PTFE-coated pistons and gas-tight seals compliant with ISO 8655-3.
- Intuitive Human-Machine Interface: 10.1-inch capacitive touchscreen with multi-language OS (English, German, Japanese), context-sensitive soft keys, and graphical real-time titration curve overlay (E vs. V, dE/dV vs. V, ΔA vs. V).
- Electrode Agnosticism: Native support for pH, redox, ion-selective (e.g., F⁻, Cl⁻, CN⁻), and noble-metal electrodes (Pt, Ag/AgCl); automatic electrode identification via embedded RFID tags (optional).
Sample Compatibility & Compliance
The MT-V6 accommodates aqueous, alcoholic, and low-viscosity organic media (e.g., acetonitrile/water mixtures) across temperatures from 5 °C to 40 °C (with optional Peltier module). Its color-detection module employs stable white LED illumination and silicon photodiode array detection, calibrated per CIE 1931 XYZ color space standards. The system complies with GLP/GMP documentation requirements: all method parameters, raw sensor outputs, operator IDs, timestamps, and audit trails are immutably logged in encrypted SQLite databases. Data export formats include CSV, PDF (with digital signature), and ASTM E1384-compliant XML for LIMS integration. Instrument qualification packages (IQ/OQ/PQ) align with USP , EP 2.2.46, and FDA 21 CFR Part 11 for electronic records and signatures.
Software & Data Management
HOGON TitrSoft v4.2 firmware provides method development wizards, derivative-based endpoint algorithms (1st/2nd derivative, Gran plot, inflection point search), and customizable pass/fail criteria per sample. Raw data files retain full metadata (burette position, stirring speed, temperature, ambient light intensity). Audit logs record every parameter modification, calibration event, and report generation with user authentication. Export options include direct USB transfer, Ethernet/Wi-Fi network upload, and OPC UA server interface for MES/SCADA systems. All software components undergo annual penetration testing and are validated against IEC 62304 Class B medical device software requirements.
Applications
- Pharmaceuticals: Quantification of active pharmaceutical ingredients (APIs) such as ascorbic acid or chlorhexidine gluconate using cerimetric or iodometric titrations with visual endpoint confirmation per USP .
- Cosmetics: Determination of preservative efficacy (e.g., methylparaben, sodium benzoate) via back-titration with standardized NaOH, where phenolphthalein color fade serves as primary endpoint.
- Environmental Testing: Copper and iron speciation in wastewater per EPA Method 200.7, leveraging color change in bathophenanthroline complexes to resolve overlapping redox potentials.
- Food & Agriculture: Vitamin C content in fruit juices (AOAC 967.21), total acidity in wine (OIV-MA-AS313-01B), and chloride in dairy products using potentiometric–colorimetric dual validation.
FAQ
Does the MT-V6 meet regulatory requirements for GMP-compliant laboratories?
Yes—the system supports 21 CFR Part 11-compliant electronic signatures, role-based access control, and immutable audit trails with time-stamped operator actions.
Can the color detection module be used independently of potentiometric measurement?
No—it operates exclusively in tandem with potentiometric acquisition to provide orthogonal endpoint verification; standalone colorimetry is not supported.
What electrode calibration protocols are supported?
NIST-traceable two-point pH calibration (4.01/7.00/10.01 buffers), redox standardization with quinhydrone or Zn/ZnSO₄, and ISE slope verification per ISO 17025 Annex B.
Is remote operation possible via network connection?
Yes—via secure HTTPS web interface or dedicated client software; remote diagnostics require prior IT security approval and VLAN segmentation.
How is maintenance documented for ISO 17025 accreditation?
All service events (e.g., burette recalibration, optical path cleaning, electrode replacement) are recorded in the internal service log with technician ID, date, and calibration certificate references.