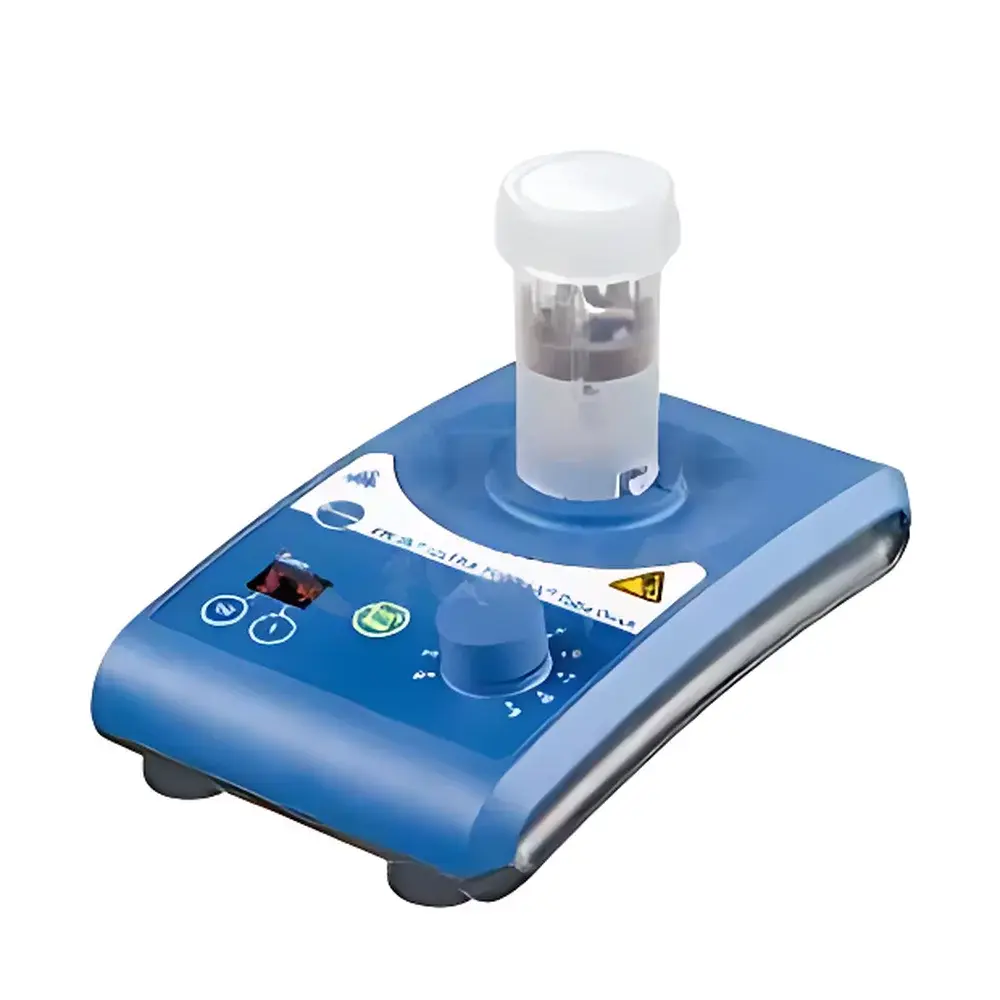
IKA ULTRA-TURRAX® Tube Drive Homogenizer
| Brand | IKA |
|---|---|
| Origin | Germany |
| Model | ULTRA-TURRAX® Tube Drive |
| Instrument Type | Probe-type Rotor-Stator Homogenizer |
| Dimensions (W×D×H) | 100 × 40 × 160 mm |
| Weight | 0.75 kg |
| Processing Volume | 2–15 mL (small tube), 15–50 mL (large tube) |
| Speed Range | 300–6,000 rpm |
| Speed Accuracy | ±10% |
| Max. Viscosity | 5,000 mPa·s |
| Motor Input Power | 20 W |
| Motor Output Power | 17 W |
| Operating Voltage | 100–240 V AC / 24 V DC |
| Current Consumption | 800 mA |
| Noise Level (no dispersing tool) | 50 dB(A) |
| Protection Class | IP20 (DIN EN 60529) |
| Ambient Temperature | 5–40 °C |
| Relative Humidity | ≤80% RH |
| Safety | 24 V low-voltage operation |
| Compliance | CE, RoHS, IEC 61010-1 |
Overview
The IKA ULTRA-TURRAX® Tube Drive is a compact, benchtop homogenizer engineered for high-integrity sample processing in biosafety-sensitive and high-throughput laboratory environments. Unlike conventional rotor-stator homogenizers requiring open-vessel handling or reusable probes, the Tube Drive operates exclusively with sealed, single-use polymer tubes—eliminating aerosol generation, cross-contamination risk, and post-processing cleaning validation. Its core principle relies on controlled mechanical shear via a rotating stator-rotor assembly integrated directly into the tube cap, enabling efficient dispersion, homogenization, emulsification, and cell lysis under fully enclosed conditions. Designed for compliance-critical workflows—including clinical diagnostics, toxicology screening, and regulated pharmaceutical development—the system delivers reproducible energy input across samples through precise speed control (300–6,000 rpm) and programmable time settings. The 24 V DC drive architecture ensures intrinsic electrical safety, while its IP20-rated housing meets IEC 61010-1 requirements for laboratory equipment used in non-hazardous locations.
Key Features
- Sealed-tube processing: Eliminates operator exposure to biohazards, volatile compounds, and malodorous analytes.
- Two-tube format support: Interchangeable 2–15 mL and 15–50 mL gamma-irradiated polypropylene tubes—sterile, DNase/RNase-free, and chemically resistant.
- No-clean workflow: Disposable tubes remove need for probe decontamination, reducing turnaround time and validation burden.
- Intuitive interface: Rotary dial for speed and timer setting; audible end-of-process signal confirms completion.
- Low-voltage safety architecture: 24 V DC motor drive minimizes electrical hazard in wet or conductive lab environments.
- Anti-winding design: Prevents entanglement of fibrous or viscous samples around the rotor shaft during high-shear processing.
- Compact footprint (100 × 40 × 160 mm) and lightweight (0.75 kg) enable placement in biosafety cabinets, cold rooms, or mobile labs.
Sample Compatibility & Compliance
The ULTRA-TURRAX® Tube Drive accommodates diverse sample matrices—including soft tissues, blood clots, plant material, bacterial pellets, suspension cultures, and viscous emulsions—without compromising integrity or introducing metallic leachables. All tubes comply with USP Class VI biocompatibility standards and are certified sterile via gamma irradiation (25 kGy). Optional perforated membrane caps allow controlled gas exchange during homogenization of anaerobic or oxygen-sensitive samples. The system supports GLP- and GMP-aligned workflows: its fixed-speed calibration traceability, absence of firmware-based data logging (reducing 21 CFR Part 11 complexity), and mechanical repeatability (±10% speed tolerance) facilitate audit-ready documentation. It conforms to DIN EN 60529 (IP20), CE marking per Directive 2014/30/EU (EMC) and 2014/35/EU (LVD), and RoHS 2011/65/EU.
Software & Data Management
The Tube Drive is a standalone, microprocessor-controlled instrument with no embedded software, network connectivity, or digital data storage. All operational parameters—speed (rpm) and duration (seconds)—are set manually via analog dial and displayed on a fixed-scale LED indicator. This design intentionally avoids electronic record-keeping to simplify regulatory alignment in environments where ALCOA+ principles require manual entry into bound lab notebooks or validated LIMS. For laboratories requiring traceability, external timers or calibrated stopwatch logs may be used in conjunction with standardized SOPs referencing IKA’s factory calibration certificate (included). No firmware updates, cloud services, or proprietary drivers are involved—ensuring long-term operational stability and IT infrastructure independence.
Applications
- Clinical pathology: Homogenization of biopsy specimens, cerebrospinal fluid sediments, and formalin-fixed paraffin-embedded (FFPE) tissue cores prior to nucleic acid extraction.
- Veterinary diagnostics: Rapid lysis of swab samples, fecal suspensions, and organ homogenates for PCR-based pathogen detection.
- Toxicology & forensic science: Uniform dispersion of solid-phase drug residues or environmental contaminants in solvent matrices.
- Pharmaceutical R&D: Preparation of nanoemulsions, liposomal suspensions, and nanoparticle dispersions under inert atmosphere (using membrane-cap tubes).
- Food safety testing: Homogenization of composite food samples (e.g., meat, dairy, produce) for microbiological enumeration per ISO 6887-1.
- Tumor biology: Single-cell suspension generation from murine xenografts without enzymatic digestion artifacts.
FAQ
Is the ULTRA-TURRAX® Tube Drive compatible with standard laboratory centrifuge tubes?
No—only IKA-certified ULTRA-TURRAX® disposable tubes (catalog numbers 0030260000 for 2–15 mL; 0030260001 for 15–50 mL) are mechanically and dimensionally compatible with the drive unit.
Can the system process samples at sub-ambient temperatures?
Yes—provided ambient lab temperature remains within 5–40 °C and relative humidity ≤80%, the device operates reliably in refrigerated rooms or on chilled work surfaces; however, tube material brittleness below 5 °C may increase fracture risk.
Does the 24 V DC power supply meet IEC 61010-1 safety requirements for laboratory use?
Yes—the external power adapter (included) is certified to IEC 62368-1 and provides reinforced insulation between primary and secondary circuits, satisfying the “separated extra-low voltage” (SELV) classification required by IEC 61010-1 Clause 6.6.
How is speed accuracy verified during routine use?
Users may validate rotational speed using a non-contact optical tachometer aligned with the tube cap’s rotation marker; IKA recommends annual verification against the factory calibration certificate using NIST-traceable instrumentation.
Are there any restrictions on solvent compatibility with the disposable tubes?
Tubes are rated for aqueous buffers, alcohols (≤70% v/v), and mild organic solvents (e.g., chloroform, ethyl acetate); strong oxidizers (e.g., concentrated HNO₃), halogenated hydrocarbons (e.g., DCM), and high-concentration phenol solutions are not recommended due to potential stress cracking.