Interscience Scan 50 Manual Colony Counter
| Brand | Interscience |
|---|---|
| Origin | France |
| Model | Scan 50 |
| Power Supply | 100–240 V~, 50–60 Hz |
| Max Power Consumption | 20 W |
| IP Rating | IP20 |
| Dimensions (W × D × H) | 27.3 × 29.5 × 15.1 cm |
| Weight (net) | 4.05 kg |
| Petri Dish Compatibility | Ø 55–90 mm |
| Counting Range | 0–1999 CFU (manual) |
| Illumination | 2nd-generation dark-field indirect LED lighting with 5 intensity levels |
| Touch Surface | Adjustable sensitivity capacitive touch panel |
| Audio Feedback | 9 selectable tones, 5 volume levels, headphone jack |
| Connectivity | USB interface for LIMS/SIL integration, barcode reader support |
| Data Export | CSV (Excel™-compatible), TXT (Word™-compatible) |
| Compliance | ISO 7218, FDA Bacteriological Analytical Manual (BAM) |
| Warranty | 3 years (registration required) |
| Spare Parts Availability | 10 years |
| Standard Accessories | Power cord, USB cable, black marker pen, white background dish, user manual |
Overview
The Interscience Scan 50 Manual Colony Counter is a precision-engineered instrument designed for reliable, ergonomic, and traceable manual colony enumeration in microbiological quality control and research laboratories. Unlike automated imaging systems, the Scan 50 operates on a tactile, human-centered counting paradigm—leveraging optimized optical contrast, responsive haptic feedback, and standardized illumination to support accurate visual identification and recording of colony-forming units (CFUs) across diverse plating methods. Its core measurement principle relies on human visual detection augmented by calibrated dark-field illumination and real-time digital registration, making it especially suitable for laboratories requiring flexibility in media types (e.g., chromogenic, selective, or membrane-based formats) without compromising auditability or regulatory alignment.
Key Features
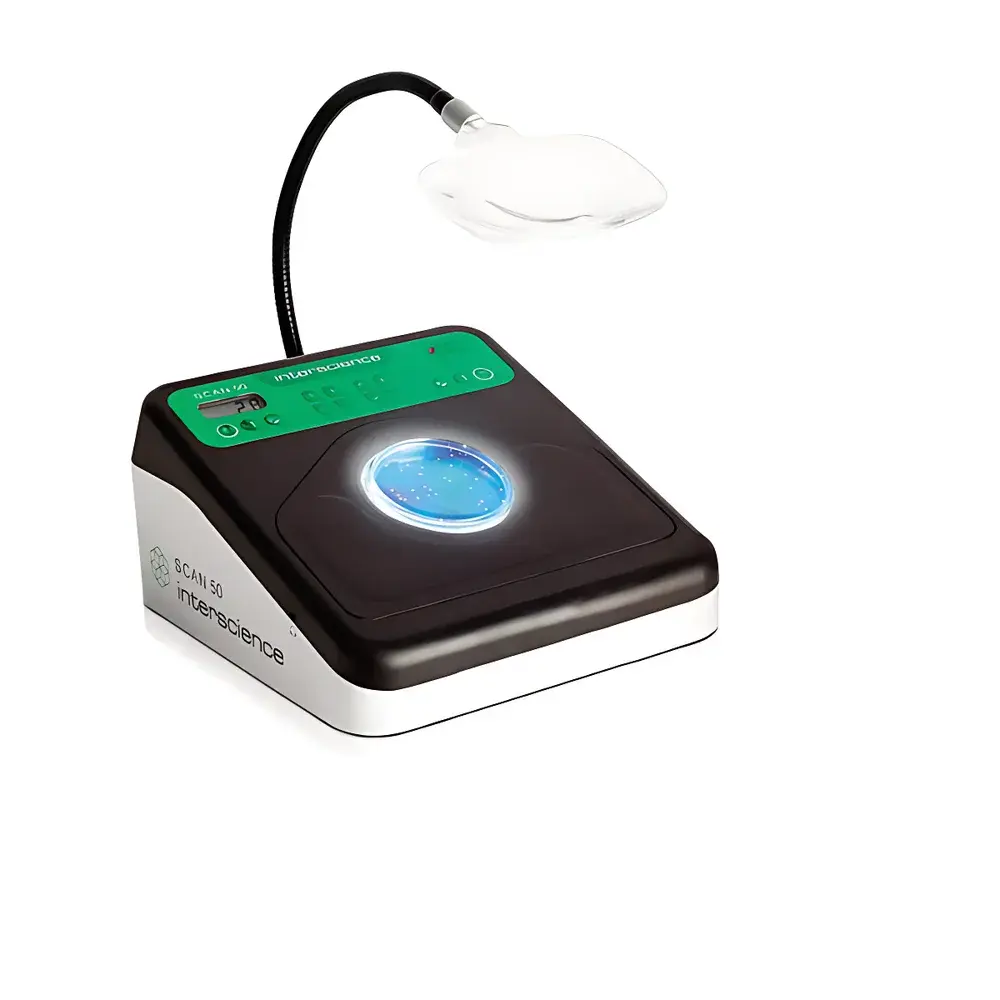
- 2nd-Generation Dark Field Illumination (Dark Field 2): An indirect LED lighting system with five adjustable intensity levels. Light is diffused via reflective surfaces to eliminate glare, shadows, and hotspots—ensuring uniform visibility of colonies across the entire agar surface, including edges and subsurface layers.
- Capacitive Touch Surface with Adjustable Sensitivity: Enables direct, intuitive colony marking on the Petri dish lid or base. Each touch triggers simultaneous haptic pulse, audible tone, and visual LED confirmation—reducing cognitive load and minimizing miscounting.
- Ergonomic Workstation Design: Low-profile chassis with optional angled magnifier stand (Cat. No. 435 001) supports neutral wrist posture and reduces musculoskeletal strain during prolonged use. Smooth, non-porous glass and removable side supports facilitate rapid disinfection between samples.
- Multi-Method Compatibility: Validated for pour plate, spread plate, spiral plating, loop dilution, and membrane filtration techniques. Supports chromogenic media, Petrifilm™, Compact Dry™, MC-Media Pads™, EasyPlate™, and filter membranes—without modification or calibration drift.
- Regulatory-Ready Data Handling: Integrated USB port enables direct export of count results—including sample ID, timestamp, operator code, and colony count—to CSV (Excel™-compatible) or plain-text (Word™-compatible) files. Optional barcode scanner integration ensures unambiguous sample traceability.
Sample Compatibility & Compliance
The Scan 50 accommodates standard Petri dishes ranging from Ø 55 mm to Ø 90 mm, covering common formats used in food, pharmaceutical, environmental, and clinical microbiology testing. It meets critical performance benchmarks outlined in ISO 7218:2018 (“Microbiology of the food chain — General requirements and guidance for microbiological examinations”) and aligns with methodology validation criteria in the U.S. FDA Bacteriological Analytical Manual (BAM). While not an automated image analyzer, its manual counting workflow—when performed under defined SOPs—supports GLP-compliant documentation and satisfies internal QA/QC requirements for method verification and personnel competency assessment. The device’s mechanical and electrical design conforms to IEC 61010-1 safety standards for laboratory equipment.
Software & Data Management
The Scan 50 operates as a standalone hardware counter with no embedded firmware or proprietary software—eliminating licensing fees, version dependencies, or cybersecurity vulnerabilities associated with networked platforms. All data management occurs at the host PC level via standard USB HID protocol. Count records are exported as flat-file outputs compatible with LIMS, ELN, or enterprise spreadsheet environments. Timestamps, operator identifiers (entered manually or via barcode), and sample metadata are preserved in each export, enabling full audit trail reconstruction. For laboratories operating under FDA 21 CFR Part 11 requirements, the system supports complementary electronic signature workflows when integrated with validated third-party data capture applications—though the Scan 50 itself does not perform electronic signing or audit log generation.
Applications
- Food and beverage microbiological testing (total viable count, coliforms, yeasts/molds)
- Pharmaceutical environmental monitoring (ISO 14644-1 classified areas)
- Clinical microbiology labs performing routine culture enumeration
- Water and wastewater analysis per APHA/EPA protocols
- Academic and industrial R&D labs requiring flexible, low-cost colony quantification
- Training environments where manual counting proficiency must be assessed and documented
FAQ
Is the Scan 50 compatible with chromogenic or selective media?
Yes—it is specifically engineered for high-contrast visualization on chromogenic agar, MacConkey, XLD, and other differential media due to its tunable dark-field illumination.
Can the device connect to a laboratory information management system (LIMS)?
Yes, via standard USB connection; exported CSV or TXT files can be ingested directly into most LIMS platforms without middleware.
Does the Scan 50 require calibration or annual certification?
No—its operation is based on tactile input and fixed optical geometry. However, periodic verification using NIST-traceable reference plates is recommended per ISO/IEC 17025 guidelines.
Is the magnifier included in the base configuration?
No—the angled magnifier (Cat. No. 435 001) is an optional accessory designed to enhance ergonomics and reduce eye fatigue during extended use.
What is the expected service life and spare parts availability?
Interscience guarantees spare parts availability for ten years following product discontinuation, and the unit carries a three-year warranty upon online registration.