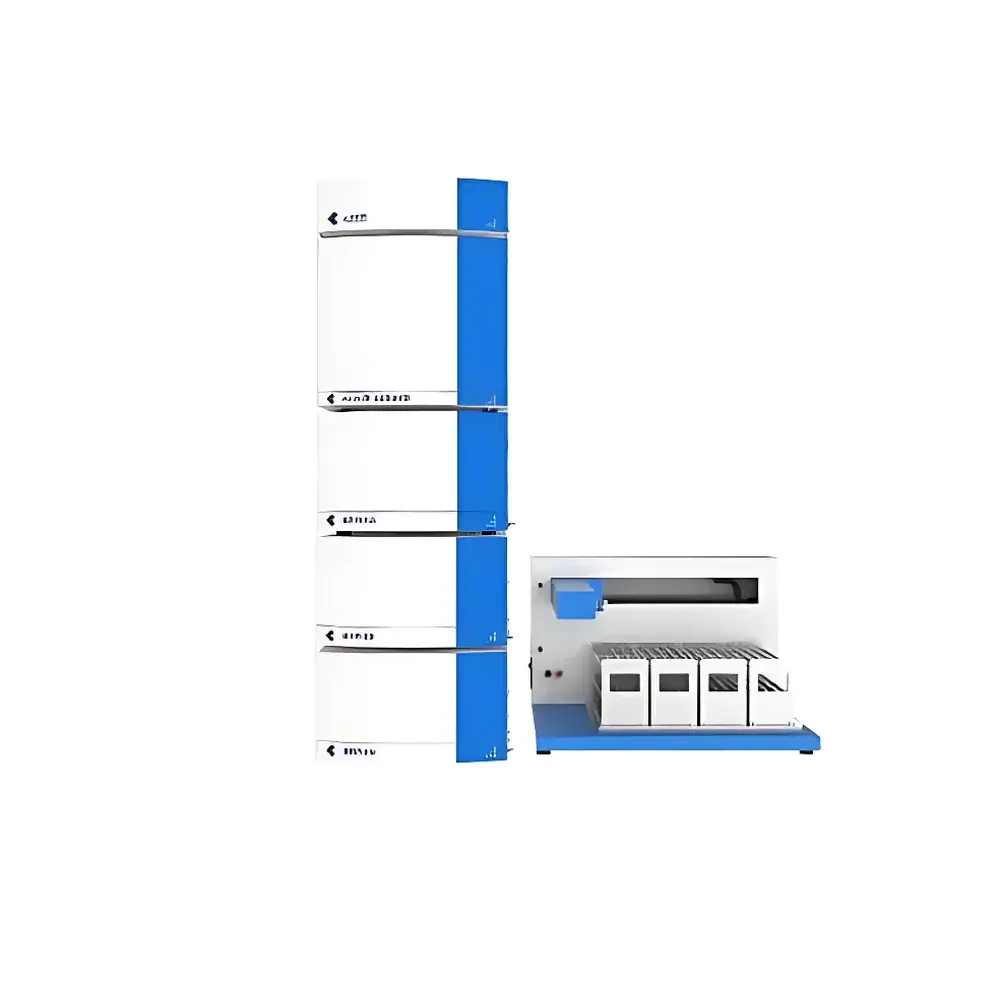
KEZHE PuriMaster-3000A Binary Automated Preparative Liquid Chromatography System
| Brand | KEZHE SHANGHAI |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Direct Manufacturer |
| Product Category | Domestic |
| Model | PuriMaster-3000A |
| Application Level | Industrial-Scale Preparative |
| Instrument Type | Medium-to-Low Pressure Preparative LC |
| Flow Rate Range | 0–250 mL/min (single pump), 0–500 mL/min (dual pump) |
| Flow Accuracy | RSD = 0.5% |
| Flow Precision | RSD = 1.0% |
| Maximum Pressure | 50 bar (standard) |
| Wavelength Range | 190–850 nm (quad-wavelength simultaneous detection) |
| Wavelength Repeatability | ±0.2 nm |
| Baseline Noise | 3 × 10⁻⁵ AU |
| Data Acquisition Rate | 5 Hz |
Overview
The KEZHE PuriMaster-3000A Binary Automated Preparative Liquid Chromatography System is an industrial-grade, medium-pressure preparative HPLC platform engineered for high-throughput isolation and purification of natural products, pharmaceutical intermediates, and synthetic compounds—particularly optimized for complex matrices such as traditional Chinese medicine (TCM) extracts. Built upon validated instrumentation architecture from China’s National Major Scientific Instrument Development Program, the system employs dual high-precision reciprocating piston pumps with active gradient mixing to deliver stable, low-lag binary solvent delivery without requiring external degassing. Its core separation mechanism relies on reversed-phase liquid chromatography (RPLC), with compatibility across standard preparative columns (e.g., C18, Φ10 × 250 mm, 10 µm). The integrated quad-wavelength UV-VIS detector utilizes a deuterium-tungsten lamp source and a 1200-line/mm holographic grating monochromator, enabling concurrent monitoring at four user-selectable wavelengths between 190 nm and 850 nm—critical for tracking co-eluting components with distinct spectral signatures.
Key Features
- Binary high-pressure gradient pumping system with real-time solvent proportioning; eliminates gradient delay and bubble formation without external degasser
- Automated sample introduction via 42-position autosampler with sealed vial handling, needle wash station, and programmable injection volume (standard 5 mL loop, customizable)
- X-Y robotic fraction collector supporting up to 160 positions for 15 × 150 mm test tubes, enabling high-density, spatially indexed collection
- Quad-channel UV-VIS detector with wavelength self-calibration, lamp life monitoring, and on-the-fly spectral scanning (190–850 nm)
- Modular software architecture supporting time-based, threshold-triggered, peak-area-driven, and manual fraction triggering modes
- Real-time overlay display of chromatogram, fraction collection map, instrument status (valve position, syringe actuation, arm motion), and gradient profile
- Comprehensive data handling including peak integration, purity assessment, retention time alignment, and batch-wise quantitative comparison
- Hardware-level audit trail logging compliant with GLP/GMP documentation requirements
Sample Compatibility & Compliance
The PuriMaster-3000A accommodates a broad range of sample types—including crude botanical extracts, fermentation broths, peptide libraries, and small-molecule reaction mixtures—with loading capacities scalable from milligram to multi-gram per run. Column compatibility extends to standard stainless-steel preparative columns (ID 10–50 mm), polymeric and silica-based stationary phases (C18, C8, phenyl, amino, diol), and custom-packed cartridges. System design adheres to IEC 61010-1 safety standards for laboratory equipment. Software supports 21 CFR Part 11-compliant user authentication, electronic signatures, and immutable audit trails. All UV-VIS performance metrics (wavelength accuracy, baseline stability, noise floor) are traceable to NIST-traceable calibration references and align with ISO 17025 method validation prerequisites for preparative process development.
Software & Data Management
The modular KEZHE PrepStation software provides full control over acquisition, processing, and reporting workflows. It features embedded spectral library matching, dynamic range optimization for absorbance linearity, and automated baseline correction algorithms suitable for overloaded or drifting baselines. Fraction indexing links each collected tube directly to its corresponding chromatographic peak apex, retention window, and integrated area—enabling rapid re-injection or offline analysis. Data export conforms to ASTM E1921 (chromatographic data interchange format) and includes CSV, PDF, and proprietary .kpd binary formats. Role-based access control supports administrator, analyst, and reviewer profiles with granular permission settings. All method files, raw data, processed results, and audit logs are stored in a relational SQLite database with automatic backup scheduling and versioned archiving.
Applications
- Isolation of bioactive constituents from TCM herbs and plant extracts (e.g., flavonoids, alkaloids, saponins, terpenoids)
- Purification of synthetic organic intermediates prior to crystallization or formulation
- Preparative-scale separation of enantiomers using chiral stationary phases
- Removal of residual catalysts or protecting groups in API synthesis workflows
- Generation of reference standards for QC laboratories under ISO/IEC 17025 accreditation
- Method scouting and scale-up studies bridging analytical to pilot-scale purification
FAQ
What is the maximum column backpressure the system can sustain?
The standard configuration supports up to 50 bar (725 psi); optional high-pressure upgrade kits extend operational limits to 30 MPa (4350 psi) with reinforced fluidic path components.
Can the system be integrated with mass spectrometry for online fraction characterization?
Yes—via optional flow-split interfaces and compatible ESI/LC-MS trigger modules; the 5 Hz acquisition rate ensures sufficient data density for MS synchronization.
Does the software support IQ/OQ/PQ documentation templates?
Yes—preconfigured qualification protocols aligned with ASTM E2500 and EU Annex 15 are included, covering pump linearity, detector response, fraction collector positional accuracy, and gradient composition verification.
Is remote monitoring and operation supported?
The system supports secure TLS-encrypted remote access via browser-based interface, enabling real-time status checks, method upload, and emergency pause/resume functions without local workstation dependency.
What validation documentation is provided with shipment?
Each unit ships with Factory Acceptance Test (FAT) report, Certificate of Conformance, UV-VIS photometric calibration certificate, and pump flow accuracy verification data—all signed and dated by KEZHE’s Quality Assurance department.