Kraemer Elektronik UTS 4.3 LAB Automatic Tablet Testing System
| Brand | Kraemer Elektronik |
|---|---|
| Origin | Germany |
| Model | UTS 4.3 LAB |
| Gas Flow Rate | 2300 mL/min |
| Reactor Volume | 3 L |
| Maximum Number of Reactors per System | 12 |
Overview
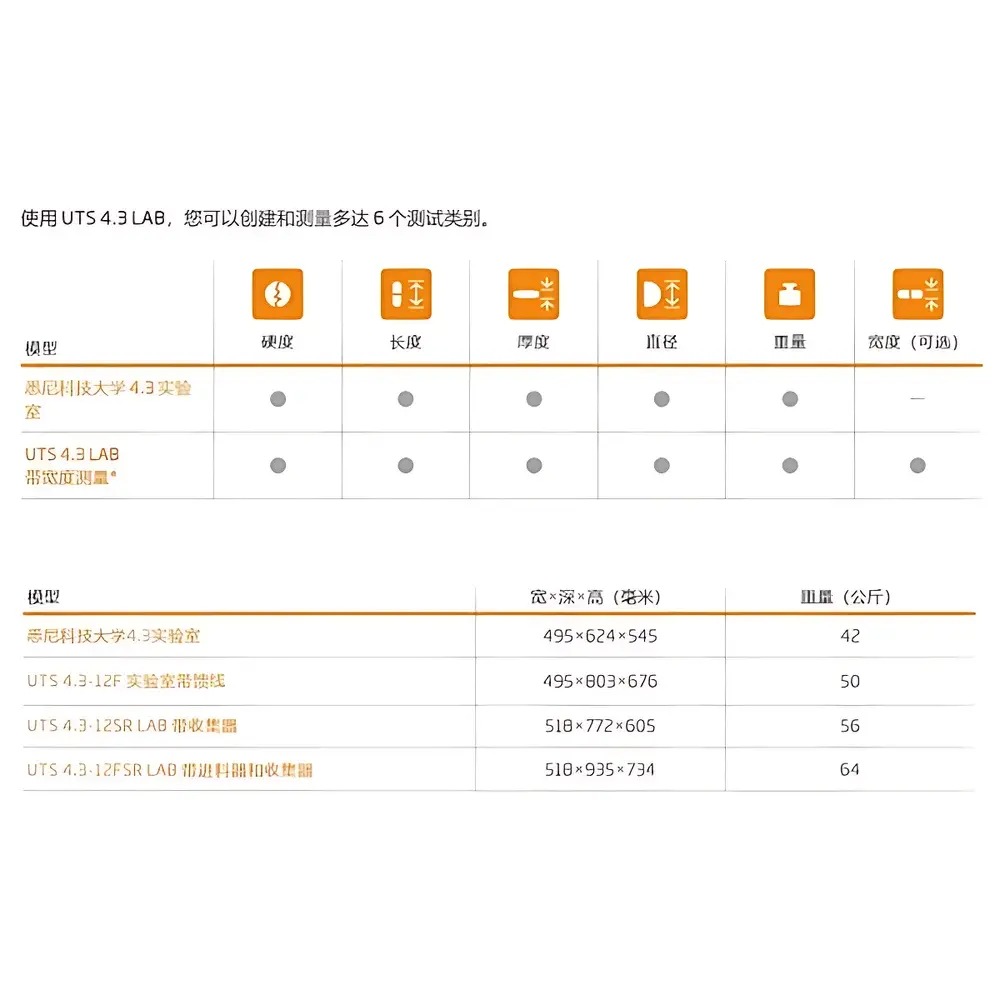
The Kraemer Elektronik UTS 4.3 LAB Automatic Tablet Testing System is a precision-engineered, database-driven platform designed for comprehensive physical and functional evaluation of solid dosage forms in regulated laboratory environments. Built upon the field-proven architecture of the UTS 4.1 series—widely deployed across pharmaceutical R&D, QC laboratories, and formulation development—the UTS 4.3 LAB integrates mechanical testing, dimensional analysis, and automated handling into a single unified workflow. It operates on a validated mechanical principle combining high-resolution load cell measurement (500 N or 1000 N options), programmable tablet positioning, and synchronized multi-station feed/collection logic. Unlike standalone hardness testers or friability analyzers, the UTS 4.3 LAB delivers traceable, repeatable, and audit-ready data across hardness, thickness, diameter, weight variation, and edge geometry—enabling full compliance with ICH Q5A, USP <1217>, EP 2.9.8, and ASTM D6179-20 standards for tablet characterization.
Key Features
- Intuitive, database-backed touchscreen interface with modular software architecture—no external PC or third-party applications required
- 12-position feeder and 12-position collector stations enabling unattended batch processing of up to 144 tablets per cycle
- Oval Centering System (OZB) integrated as standard for reliable orientation and measurement of elliptical, oblong, and irregularly shaped tablets
- High-accuracy load cell options: 500 N (standard) or 1000 N (for high-hardness formulations or coated extended-release tablets)
- FDA 21 CFR Part 11-compliant software package including electronic signatures, role-based user management, full audit trail, and batch-level reporting
- Dual communication interfaces: USB 2.0 for local data export and Gigabit Ethernet (LAN) for network integration with LIMS, MES, or centralized QA databases
- Robust stainless-steel frame with IP54-rated enclosure suitable for ISO Class 7/8 cleanroom-adjacent lab environments
Sample Compatibility & Compliance
The UTS 4.3 LAB accommodates tablets ranging from 4 mm to 25 mm in diameter (or longest dimension), including round, oval, square, rectangular, capsule-shaped, and bi-convex geometries. Its adaptive gripper mechanism and OZB subsystem ensure consistent centering and contact alignment—even for low-aspect-ratio or highly asymmetric tablets. All hardware and firmware comply with CE marking requirements under the EU Machinery Directive 2006/42/EC and EMC Directive 2014/30/EU. The system supports IQ/OQ documentation packages aligned with GMP Annex 15 and is preconfigured for validation against USP <1217> (tablet hardness), <905> (uniformity of dosage units), and <1118> (instrument qualification). Optional TTS11 license enables direct CSV/LIMS export and full traceability of raw sensor data, calibration logs, and operator actions.
Software & Data Management
The embedded UTS TouchOS v4.3 software provides real-time visualization of force-displacement curves, statistical process control (SPC) charts, and histogram-based distribution analysis. Data is stored in an internal SQL Server Express database with automatic daily backups and configurable retention policies. Each test record includes timestamped metadata: operator ID, tablet ID (via optional barcode reader integration), environmental conditions (if linked to external温湿度 sensors), and instrument calibration status. Audit trails meet ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available) and support FDA inspection readiness. Software modules—including advanced SPC analytics, custom report templates (PDF/Excel), and remote diagnostics—are licensed separately and installed via secure encrypted update packages.
Applications
- Pre-formulation screening of excipient impact on tablet mechanical integrity
- QC release testing for hardness, thickness, and diameter uniformity per batch
- Stability study monitoring of tablet friability and disintegration kinetics over time
- Process validation of direct compression, wet granulation, and roller compaction lines
- Biodegradation testing support (when coupled with gas evolution monitoring modules for CO₂ quantification in OECD 301/310-compliant assays)
- Regulatory submission dossier preparation with fully traceable, Part 11–compliant datasets
FAQ
Is the UTS 4.3 LAB suitable for GLP-compliant laboratories?
Yes—the system’s audit trail, electronic signature capability, and documented IQ/OQ protocols fulfill core GLP requirements per OECD Principles of Good Laboratory Practice.
Can the system be integrated into an existing LIMS environment?
Yes—via the optional TTS11 license, which provides standardized CSV export, HL7-compatible metadata tagging, and direct SQL query access to the internal database.
Does the OZB system require manual adjustment for different tablet shapes?
No—the OZB uses motorized, vision-assisted alignment and automatically adapts its clamping profile based on preloaded shape templates or real-time edge detection.
What calibration standards are supported?
NIST-traceable stainless-steel reference tablets (hardness: 5–200 kp) and certified gauge blocks (thickness: 1–10 mm) are recommended; calibration certificates are generated in accordance with ISO/IEC 17025.
Is remote software support available?
Yes—Kraemer Elektronik offers secure remote maintenance via TLS-encrypted VNC sessions, subject to customer network policy approval and firewall configuration.