LabTech LabPure-10 Circulating Ultrapure Water System
| Brand | LabTech |
|---|---|
| Origin | Beijing, China |
| Manufacturer Type | Direct Manufacturer |
| Country of Origin | Domestic (China) |
| Model | LabTech-10 |
| Pricing | Available Upon Request |
| Pipe Material | PVDF/CPVC |
| Main Loop Velocity | 1.5–3.0 m/s |
| Branch Loop Velocity | 1.0–2.0 m/s |
| Recirculation Ratio | 30–50% of Design Flow Rate |
| Compliance Focus | Low Extractables, Smooth Internal Surface, Air-Tight Integrity |
Overview
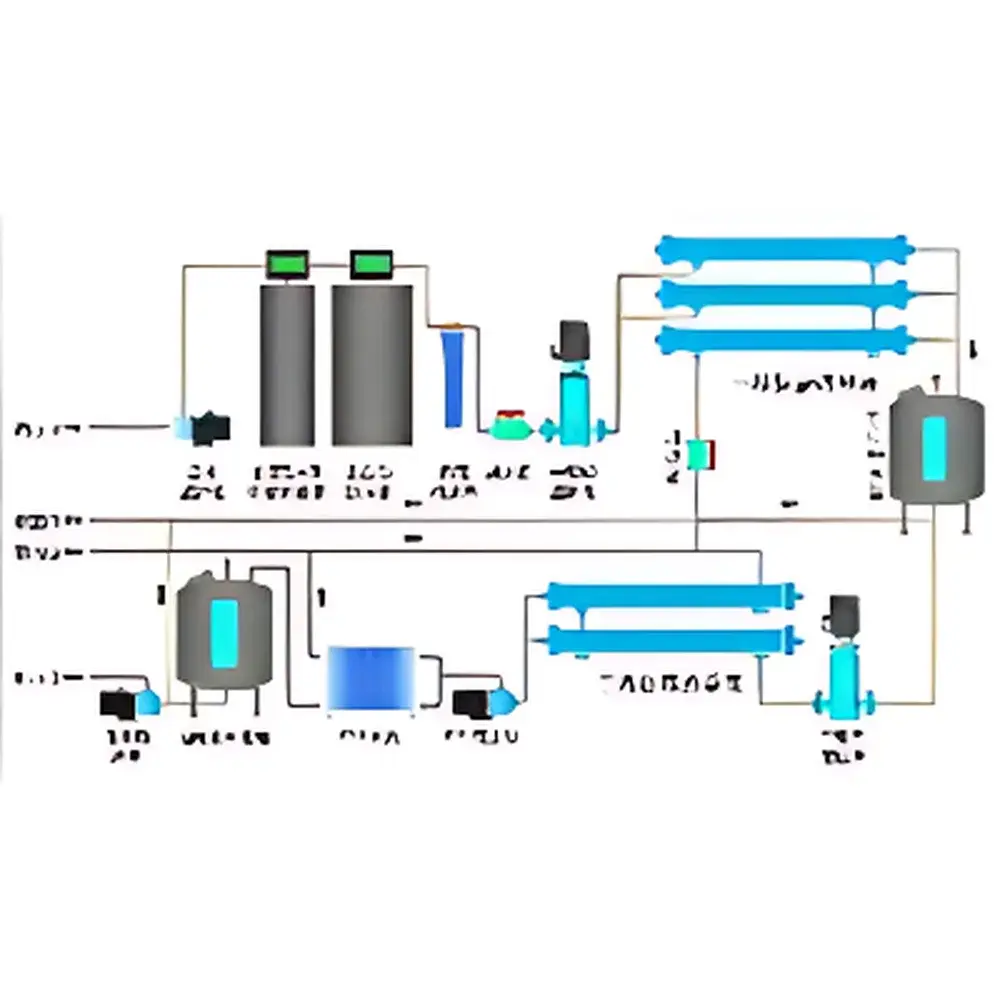
The LabTech LabPure-10 Circulating Ultrapure Water System is an engineered solution for continuous, high-integrity water delivery in analytical laboratories, pharmaceutical QC environments, and research facilities requiring ISO 3696 Grade 1 or ASTM D1193 Type I ultrapure water. Unlike static storage-and-draw systems, the LabPure-10 employs a fully recirculating loop architecture—comprising dedicated supply and return manifolds—to maintain dynamic flow conditions throughout the distribution network. This design eliminates stagnant zones where biofilm formation, particulate settling, or ion leaching may occur. The system operates on the principle of controlled laminar-to-turbulent transition within engineered piping hydraulics, ensuring consistent velocity profiles that suppress microbial adhesion while minimizing shear-induced pipe wall erosion. Its core function is not merely water purification, but sustained water quality preservation across the entire distribution path—from point-of-generation to point-of-use—thereby meeting the stringent physical, chemical, and microbiological stability requirements defined in USP , EP 2.2.42, and ISO 14644-1 Class 5 cleanroom-aligned water handling protocols.
Key Features
- Full-loop recirculation architecture with dual-path (supply + return) stainless steel or PVDF-lined manifolds to eliminate dead legs and ensure zero-stagnation hydraulic design
- Optimized flow velocity control: main loop maintained at 1.5–3.0 m/s; branch lines regulated to 1.0–2.0 m/s—within ASME BPE-recommended ranges for ultrapure water systems to inhibit biofilm accumulation without inducing excessive pressure drop
- Low-extractable PVDF/CPVC piping system selected per ASTM F2136 and ISO 15877 standards, featuring ultra-smooth internal surface finish (Ra ≤ 0.4 µm), minimal ionizable impurity content, and certified low TOC leachability (< 1 ppb after 72-h extraction test)
- Air-tight jointing methodology using orbital fusion welding (PVDF) or solvent-cemented CPVC joints with leak-tested integrity verification—preventing ambient air ingress that could elevate dissolved oxygen, CO₂, and airborne particulates
- Modular scalability: designed for integration with upstream purification units (e.g., reverse osmosis, electrodeionization, UV oxidation, 0.1 µm final filtration) and compatible with optional real-time resistivity (≥18.2 MΩ·cm @ 25°C), TOC (<5 ppb), and total viable count (TVC < 0.1 CFU/mL) monitoring modules
Sample Compatibility & Compliance
The LabPure-10 is validated for use with sensitive analytical instrumentation including HPLC, LC-MS, ICP-MS, atomic absorption spectrometry, and cell culture media preparation. Its distribution performance aligns with regulatory expectations for GMP-compliant water systems under FDA 21 CFR Part 211 and EU Annex 1, particularly regarding prevention of microbial proliferation and chemical contamination. All wetted materials comply with USP Class VI biocompatibility testing, and the piping system meets ISO 15877 Category S5 (hot water service) and ASTM D1784 cell classification for CPVC. Documentation packages support IQ/OQ/PQ execution and include material traceability records, weld logs, pressure test reports, and extractables profiling data per USP .
Software & Data Management
While the LabPure-10 is a hardware-engineered distribution platform (not a digitally native instrument), it supports integration into facility-wide Building Management Systems (BMS) via 4–20 mA analog outputs or Modbus RTU communication for flow rate, pressure differential, and temperature monitoring. Optional digital add-ons include time-stamped audit trails for maintenance events, automated log generation for weekly microbiological sampling points, and electronic record retention compliant with ALCOA+ principles. When paired with LabTech’s optional LabMonitor Suite, users gain configurable alerts for velocity deviation (>±10%), pressure loss (>15% from baseline), or loop residence time exceedance—enabling proactive intervention before water quality drift occurs.
Applications
- HPLC and UHPLC mobile phase preparation where sub-ppb ionic contamination directly impacts baseline noise and column lifetime
- ICP-MS sample introduction systems requiring ultra-low background metals and negligible carbon contribution
- Pharmaceutical stability studies and dissolution testing governed by USP analytical instrument qualification guidelines
- Genomics labs performing next-generation sequencing library prep, where nuclease-free, DNase/RNase-free water integrity is non-negotiable
- Calibration laboratories maintaining ISO/IEC 17025 accreditation, where water purity must be demonstrably traceable to NIST SRM-certified reference materials
FAQ
What is the recommended recirculation ratio for optimal microbial control?
A minimum recirculation flow of 30% of peak design demand is required; 50% is recommended for facilities operating under continuous 24/7 usage or those supporting critical GMP applications.
Why is PVDF preferred over stainless steel in certain LabPure-10 configurations?
PVDF offers superior resistance to chloride-induced stress corrosion cracking, lower metallic leachables, and inherently smoother electrostatic surface properties—critical when supplying water to trace-metal-sensitive instruments such as ICP-MS.
Can the LabPure-10 be retrofitted into an existing lab water infrastructure?
Yes—modular manifold design allows phased integration; however, full compliance requires replacement of legacy piping segments downstream of the last purification unit to ensure system-wide velocity and material consistency.
Does the system include validation documentation for GxP environments?
Standard delivery includes material certifications and pressure test records; full IQ/OQ documentation packages are available as a configurable service option aligned with Annex 15 and ASTM E2500-13.