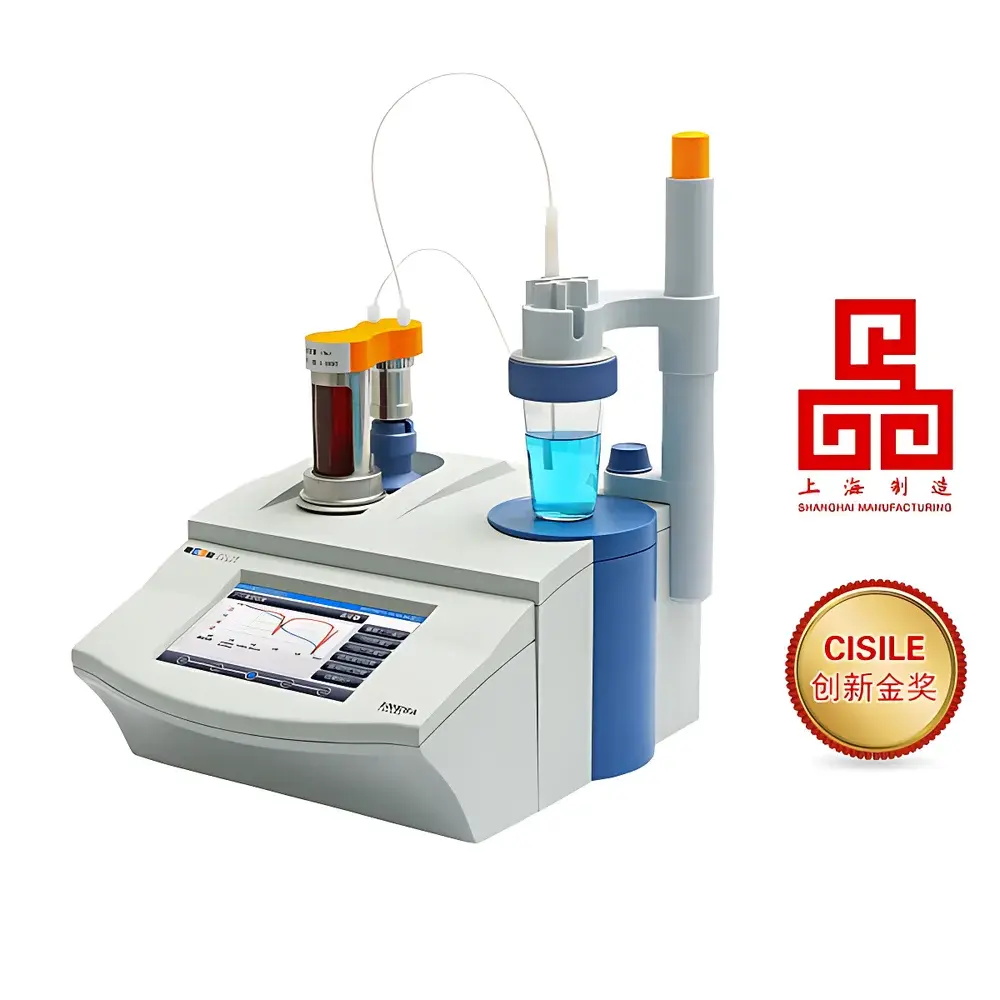
LeiCi ZDJ-5B-T Automatic Potentiometric Titrator
| Brand | LeiCi |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Manufacturer |
| Product Category | Domestic |
| Model | ZDJ-5B-T |
| Instrument Type | General-purpose Potentiometric Titrator |
| Application Scope | Universal |
| Measurement Range | (−2000.00 to 2000.00) mV |
| Meter Accuracy | mV: ±0.03% or ±0.2 mV |
| pH | ±0.002 pH |
| Meter Resolution | 0.01 mV, 0.001 pH |
| Titration Precision | 0.0003 mL |
| Titration Resolution | 1/30000 |
Overview
The LeiCi ZDJ-5B-T Automatic Potentiometric Titrator is a precision-engineered electrochemical analytical instrument designed for high-reproducibility potentiometric and thermometric titrations in quality control, research, and routine laboratory environments. It operates on the fundamental principle of monitoring potential changes (in mV) or temperature shifts (in °C) across a redox, acid–base, precipitation, complexometric, or non-aqueous titration reaction to identify inflection points corresponding to stoichiometric endpoints. Its dual-capability architecture supports both classical potentiometric detection via ion-selective or redox electrodes and thermometric endpoint detection—enabling robust analysis where conventional potentiometric signals are weak or unstable (e.g., in low-conductivity solvents or heterogeneous systems). The instrument complies with core metrological requirements aligned with ISO/IEC 17025 calibration traceability frameworks and supports GLP-compliant data integrity through embedded audit trails, user access controls, and electronic signature readiness.
Key Features
- Integrated dual-channel titration system: one built-in burette (standard 10 mL) plus optional external burette module (10 mL or 20 mL), enabling rapid reagent switching without manual disassembly;
- Valve-burette monolithic design with chemically inert fluid path materials—resistant to concentrated acids, perchloric acid, strong alkalis, and organic solvents (e.g., acetonitrile, glacial acetic acid), certified for non-aqueous titrations per ASTM D664 and USP <771>;
- 7-inch capacitive color touchscreen with guided workflow navigation, real-time titration curve plotting (mV vs. volume, dE/dV vs. volume, or ΔT vs. volume), and simultaneous display of method parameters, raw signal, and calculated results;
- Multi-mode titration engine supporting dynamic titration (adaptive addition), preset endpoint titration, constant-dose titration, blank correction, manual stepwise addition, and user-defined custom protocols;
- Comprehensive method management: up to 100 stored titration methods with editable parameters, 10 customizable quick-access shortcuts, formula-based result calculation (e.g., % purity, mg KOH/g, mmol/L), and post-run recalculation with modified equivalence point criteria;
- Built-in temperature titration module (0.000–60.000 °C range, ±0.3 °C accuracy at 0–60 °C) with automatic temperature compensation (−5.0 to 105.0 °C) and 0.001 °C repeatability for thermometric endpoint validation;
- Full compliance with data integrity standards: 200 GLP-compliant result records with timestamped metadata (operator ID, method version, electrode serial, ambient temperature/humidity), statistical reporting (mean, SD, RSD, confidence intervals), and export-ready CSV/PDF output.
Sample Compatibility & Compliance
The ZDJ-5B-T accommodates diverse sample matrices including aqueous solutions, alcoholic extracts, petroleum distillates, pharmaceutical suspensions, food emulsions, and polymer dissolution media. Its corrosion-resistant fluidic components meet ISO 8573-1 Class 3 purity requirements for reagent delivery. The instrument supports electrode configurations compliant with IUPAC recommendations for potentiometric measurements and is validated for use with glass pH electrodes, silver/silver chloride reference electrodes, platinum redox electrodes, and thermistors meeting DIN EN 60751 Class A tolerances. All firmware and software operations adhere to FDA 21 CFR Part 11 principles—including electronic signatures, role-based access control (administrator/operator/auditor), and immutable audit logs—making it suitable for regulated environments under GMP, ISO 9001, and pharmacopeial testing (USP, EP, JP).
Software & Data Management
Controlled via REX Titrator Software (included), the ZDJ-5B-T enables bidirectional RS-232 and USB communication with Windows-based PCs. The software provides method development wizards, real-time remote monitoring, automated report generation (with configurable templates), and U disk plug-and-play data export without driver installation. All measurement data—including raw sensor readings, derivative curves, equivalence point detection logic, and operator annotations—are stored in encrypted binary format with SHA-256 checksum verification. Data backups support network drive mapping and scheduled auto-export to centralized LIMS servers. Firmware updates are performed via signed package files to ensure integrity and version traceability.
Applications
- Acid–base titrations: total acidity in wines (AOAC 942.15), free fatty acids in oils (ISO 660), alkalinity in wastewater (APHA 2320 B);
- Oxidation–reduction titrations: iron content in ores (ASTM E1087), ascorbic acid in beverages (AOAC 967.21), permanganate demand in water;
- Complexometric titrations: calcium/magnesium hardness (ISO 6059), EDTA-based metal ion quantification in plating baths;
- Precipitation titrations: chloride in brines or pharmaceutical excipients (USP <511>);
- Non-aqueous titrations: assay of weak bases (e.g., amines, alkaloids) in acetic anhydride/glacial acetic acid per USP <771>;
- Thermometric titrations: determination of carboxylic acid content in polymers where pH electrodes exhibit drift or sluggish response.
FAQ
Does the ZDJ-5B-T support regulatory compliance for pharmaceutical QC labs?
Yes—it implements full 21 CFR Part 11 functionality including electronic signatures, audit trail review, and user privilege segregation, and meets data retention requirements for USP Analytical Instrument Qualification.
Can the instrument perform simultaneous potentiometric and thermometric titration on the same sample?
No—the modules operate independently; however, sequential analysis using both endpoints (e.g., pH followed by ΔT confirmation) is fully supported within a single method script.
What electrode types are natively compatible with the ZDJ-5B-T?
Standard BNC-input pH, redox, ion-selective, and thermistor probes; no proprietary connectors required.
Is method transfer between instruments possible?
Yes—method files (.rex) are portable across all LeiCi REX-series titrators and retain full parameter fidelity, including custom formulas and curve-fitting settings.
How is滴定精度 (0.0003 mL) verified during calibration?
Precision is confirmed gravimetrically per ISO 8655-3 using certified weights and Class A volumetric glassware, with documented uncertainty budgets traceable to NIM (National Institute of Metrology, China).