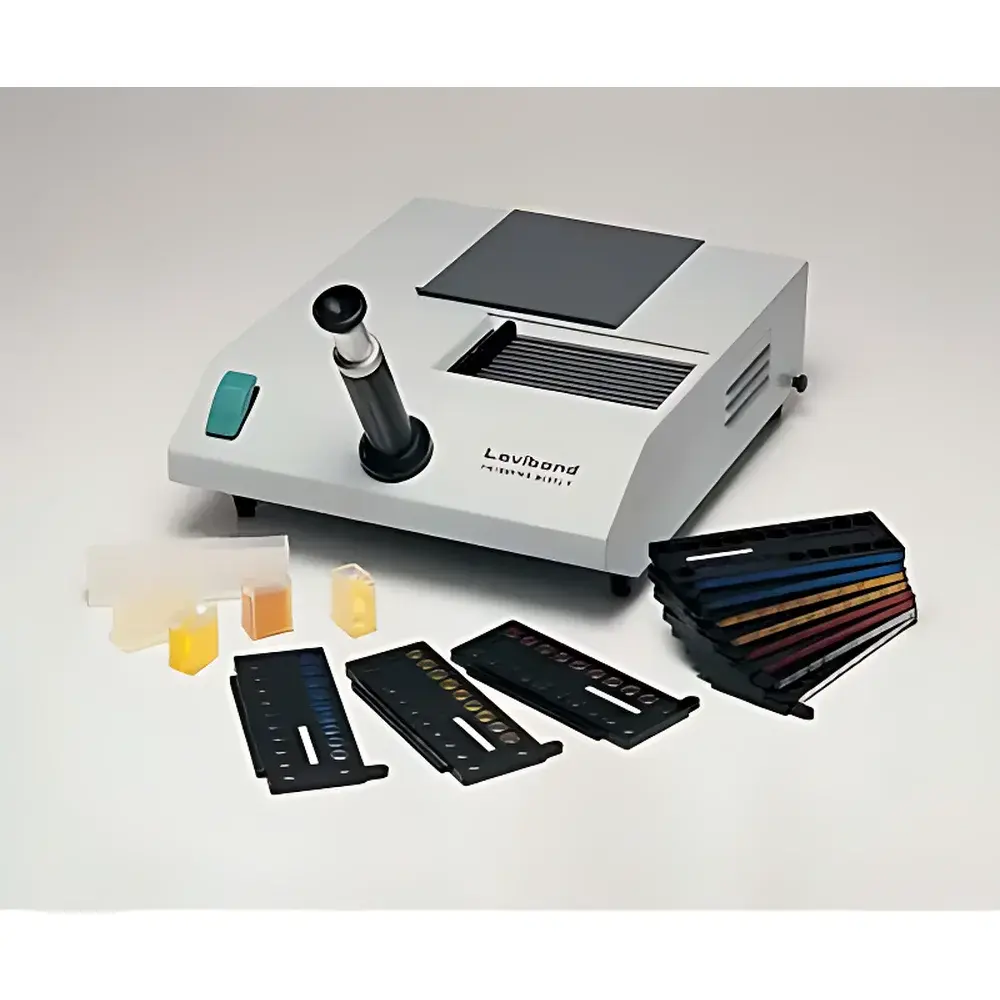
Lovibond Model F Visual Colorimeter
| Brand | Lovibond (Tintometer) |
|---|---|
| Origin | United Kingdom |
| Manufacturer Status | Authorized Distributor |
| Product Origin | Imported |
| Model | Model F |
| Price Range | USD 6,800 – 13,600 (FOB) |
| Color Scale | Lovibond RYBN |
| Red Scale | 0.1–79.9 |
| Yellow Scale | 0.1–79.9 |
| Blue Scale | 0.1–49.9 |
| Neutral Scale | 0.1–3.9 |
| Resolution | 0.1 Lovibond Unit |
| Light Source | Dual 12 V / 10 W Tungsten-Halogen Lamps |
| Illuminant Compliance | CIE Standard Illuminant C |
| Sample Chamber | Removable White Liner with Precision-Registered Cell Notch |
| Power Supply | External Low-Voltage DC Adapter |
Overview
The Lovibond Model F Visual Colorimeter is a precision-engineered optical measurement instrument designed for standardized visual color assessment in industrial quality control and laboratory environments. Based on the internationally recognized Lovibond RYBN color scale—developed over a century of empirical calibration—the Model F implements the classical two-field simultaneous comparison method using physically calibrated glass interference filters. Unlike digital spectrophotometers, this instrument relies on human observer perception under controlled illumination, making it particularly suitable for applications where perceptual color matching aligns with end-user expectations (e.g., edible oils, pharmaceutical syrups, lubricants, and food-grade fats). Its optical architecture conforms to CIE Standard Illuminant C, ensuring consistent chromaticity evaluation across laboratories and regulatory audits. The device operates without software dependency, eliminating calibration drift associated with sensor aging and enabling long-term traceability to NPL-traceable Lovibond reference standards.
Key Features
- Dual-field viewing system with sharp, optically defined boundary between sample and standard fields—minimizing parallax error and enhancing discrimination of subtle hue differences.
- Modular Lovibond RYBN filter disc assembly housed in a rigid, anodized aluminum carrier; each disc is individually indexed and protected from mechanical abrasion or misalignment.
- High-stability tungsten-halogen lamp pair (12 V / 10 W), thermally regulated via external low-voltage DC power supply to prevent thermal lensing and maintain luminance stability over extended operation (>8 h continuous use).
- Interchangeable white liner with integrated cell registration groove ensures repeatable sample positioning and eliminates edge-scatter artifacts—critical for viscous or opaque samples such as hydrogenated oils and gelatinous extracts.
- Optical path engineered for uniform field illumination and minimal vignetting; all internal baffles and diffusers are coated with matte-finish spectral-grade black paint to suppress stray light.
Sample Compatibility & Compliance
The Model F accommodates both transparent and semi-opaque liquid and semi-solid samples. Transparent matrices—including mineral oils, glycerin-based solvents, corn syrup, and aqueous sugar solutions—are measured in standard 10 mm or 25 mm pathlength cells. For turbid or highly scattering media (e.g., lard, margarine, waxes), the instrument supports thick-layer assessment using the optional “paste tray” configuration, where sample thickness is mechanically constrained to 1.0 ± 0.05 mm. Methodologically, the Model F complies with ASTM D1500 (Petroleum Products), ISO 4630 (Mineral Oils), and AOCS Cd 13b-97 (Fats and Oils). Its physical standard-based operation satisfies GLP documentation requirements when paired with certified Lovibond reference discs and logbook-based observer records per FDA 21 CFR Part 11 Annex 11 principles.
Software & Data Management
The Model F is a standalone analog instrument with no embedded firmware or digital interface. All measurements are recorded manually in laboratory notebooks or LIMS-integrated templates. Each test requires observer identification, date/time stamp, sample ID, observed RYBN values, and environmental notes (ambient lighting, observer visual acuity status). For audit readiness, laboratories may integrate the Model F into electronic quality management systems via scanned image logs of reference disc positions or structured CSV exports generated from standardized observation forms. While not electronically connected, its operational simplicity ensures full compliance with ISO/IEC 17025 clause 7.7 on measurement traceability through documented chain-of-custody of certified Lovibond standards.
Applications
- Refined vegetable oil grading (soybean, palm, sunflower) per IOOC and USDA specifications.
- Quality release testing of pharmaceutical excipients including propylene glycol, sorbitol syrup, and polyethylene glycols.
- Batch-to-batch consistency verification in fine chemical synthesis (e.g., aldehyde condensation products, dye intermediates).
- Regulatory submission support for FDA GRAS dossiers requiring perceptually validated color limits.
- Educational use in analytical chemistry curricula for teaching foundational colorimetric principles and observer variability assessment.
FAQ
Is the Model F compliant with current pharmacopoeial standards?
Yes—the instrument fulfills USP <631>, EP 2.2.43, and JP 6.06 requirements for visual color comparison when operated by trained observers under controlled lighting conditions.
Can the Lovibond RYBN scale be converted to CIELAB or sRGB?
No direct mathematical conversion exists due to fundamental differences in perceptual vs. tristimulus modeling; however, empirical correlation tables are available from Tintometer Ltd. for specific product categories under defined viewing conditions.
How often must the reference discs be recalibrated?
Lovibond-certified glass filters are stable for ≥10 years if stored in darkness and handled with lint-free gloves; annual verification against master reference sets is recommended per ISO/IEC 17025 clause 6.4.
Does the Model F require annual factory servicing?
No—routine maintenance consists only of lamp replacement (every 1,200 hours), liner cleaning, and optical path inspection; full metrological validation is performed at accredited third-party labs per ISO 17025.
Is observer training required for regulatory submissions?
Yes—FDA and EMA require documented evidence of color vision proficiency (e.g., Ishihara test) and instrument-specific competency assessments conducted at least annually.