METTLER TOLEDO Excellence Series Melting Point Apparatus MP55/MP70/MP80/MP90
| Brand | METTLER TOLEDO |
|---|---|
| Origin | Switzerland |
| Model | MP55 / MP70 / MP80 / MP90 |
| Temperature Range | Ambient to 400 °C |
| Heating Rate | 0.1–20 °C/min |
| Temperature Accuracy | ±0.2 °C |
| Temperature Resolution | 0.1 °C |
| Capillary Diameter Compatibility | <1.8 mm |
| Sample Capacity | Up to 6 samples simultaneously |
| Compliance | GLP/GMP-ready, ISO/IEC 17025-aligned calibration traceability |
Overview
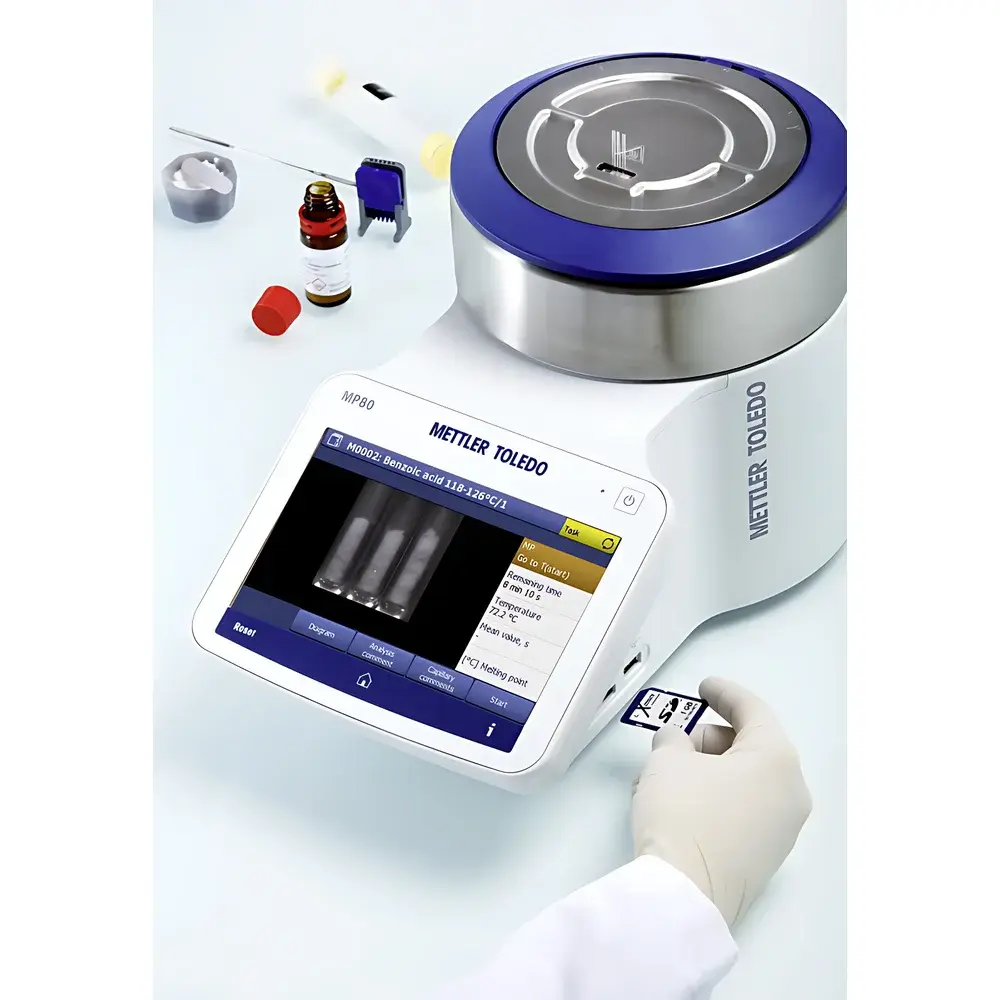
The METTLER TOLEDO Excellence Series Melting Point Apparatus (MP55, MP70, MP80, MP90) is a fully automated, video-based thermal analysis system engineered for precise, reproducible determination of melting point, boiling point, cloud point, and slip melting point. Operating on the principle of controlled heating combined with real-time optical monitoring of sample phase transitions, the instrument captures high-resolution video of capillary tube samples while simultaneously recording light transmission intensity curves. This dual-mode detection—visual confirmation plus photometric quantification—ensures unambiguous endpoint identification, eliminating subjectivity inherent in manual observation. Designed for routine quality control laboratories in pharmaceuticals, chemicals, polymers, and food science, the system delivers regulatory-compliant data acquisition without requiring operator intervention beyond initial sample loading and method selection.
Key Features
- One-Click™ automated measurement workflow: Initiate full thermal ramp, image capture, endpoint detection, and result reporting with a single touchscreen command.
- Integrated high-definition video camera and LED illumination system enabling real-time visualization and post-run playback of sample melting behavior.
- Optical intensity curve analysis: Quantitative photometric tracking of light transmission through the capillary provides objective, algorithm-driven melting onset and clear-point determination.
- Multi-sample capability: Simultaneous processing of up to six samples per run with independent temperature programming and individual result logging.
- Full-color capacitive touchscreen interface with intuitive icon-based navigation, multilingual support (English, German, French, Chinese, Japanese), and context-sensitive help.
- Robust thermal architecture featuring platinum resistance thermometer (PRT) sensing, PID-controlled heating block, and active cooling for rapid cycle times between runs.
- Comprehensive security and audit trail: User access levels, electronic signatures, and time-stamped event logs meet FDA 21 CFR Part 11 and EU Annex 11 requirements.
Sample Compatibility & Compliance
The system accommodates standard thin-walled glass capillaries (outer diameter <1.8 mm), compatible with both loose powder and pre-packed reference standards. It supports common pharmacopoeial methods including USP <741>, Ph. Eur. 2.2.17, and JP 2.60. All instruments are supplied with factory calibration certificates traceable to NIST and PTB standards. Built-in calibration verification using certified reference substances (e.g., indium, caffeine, phenacetin) allows users to perform periodic performance checks in accordance with ISO/IEC 17025 clause 6.5. Calibration data, verification results, and method parameters are stored internally with immutable timestamps. The hardware and firmware architecture comply with IEC 61010-1 safety standards and electromagnetic compatibility (EMC) directives (EN 61326-1).
Software & Data Management
The embedded operating system includes method templates preconfigured for USP, EP, and JP monographs, as well as customizable user-defined protocols. Measurement data—including raw intensity curves, annotated video clips (AVI/MPEG-4), temperature profiles, and calculated endpoints—is saved in a structured internal database. Export options include CSV, PDF (with embedded signature fields), and XML formats suitable for LIMS integration. Data integrity safeguards include write-protected storage, automatic backup to USB drives, and optional network synchronization with METTLER TOLEDO LabX® software for centralized instrument management, statistical process control (SPC), and cross-laboratory trend analysis. All exported reports contain full metadata: operator ID, instrument serial number, calibration status, environmental conditions, and digital signatures.
Applications
- Pharmaceutical QC: Identity confirmation and purity assessment of active pharmaceutical ingredients (APIs) and excipients per ICH Q5 and Q6 guidelines.
- Chemical manufacturing: Verification of batch-to-batch consistency and detection of polymorphic forms or eutectic impurities.
- Academic research: Characterization of novel organic compounds, metal-organic frameworks (MOFs), and supramolecular assemblies.
- Food & agriculture: Determination of fat melting profiles (e.g., cocoa butter equivalents) and adulteration screening via melting range deviation.
- Polymers & materials science: Thermal stability evaluation of thermoplastics, waxes, and phase-change materials (PCMs).
FAQ
What regulatory standards does the MP series support?
The MP55/MP70/MP80/MP90 meets GLP and GMP documentation requirements, supports 21 CFR Part 11 compliance when used with LabX, and enables full audit trail generation for ISO 9001 and ISO/IEC 17025 accreditation.
Can I verify calibration without external equipment?
Yes—the system includes built-in calibration verification using certified reference substances; results are automatically logged with pass/fail criteria and uncertainty estimates.
Is remote monitoring or control possible?
When connected to LabX software via Ethernet or Wi-Fi, users can monitor live measurements, receive email alerts upon completion, and manage methods from any networked workstation.
How is sample cross-contamination prevented?
Each capillary is thermally isolated within its own dedicated heating channel; the system performs automatic thermal equilibration before each run and enforces mandatory cooldown periods between measurements.
What video file format is generated during analysis?
Recordings are saved in AVI format with H.264 encoding at 30 fps, synchronized frame-by-frame with temperature and light intensity data.