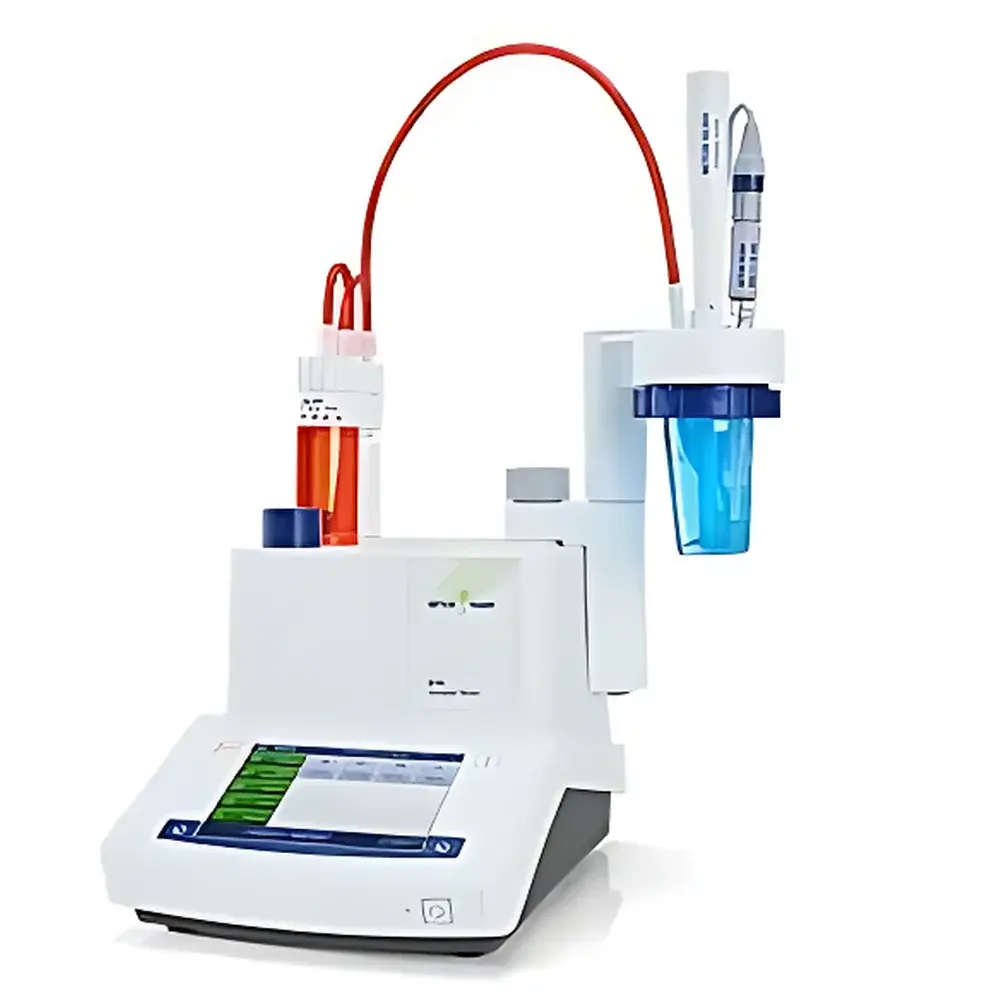
METTLER TOLEDO G10S/G20S Smart Series Automatic Potentiometric Titrator
| Brand | METTLER TOLEDO |
|---|---|
| Origin | Switzerland |
| Model | G10S / G20S |
| Instrument Type | General-purpose Automatic Potentiometric Titrator |
| Measurement Range | ±2000 mV / –26.0 to +40.0 pH |
| Meter Accuracy | 0.01% of reading |
| Meter Resolution | 0.1 mV / 0.001 pH |
| Titration Precision | 0.2 mV / 0.003 pH |
Overview
The METTLER TOLEDO G10S and G20S Smart Series Automatic Potentiometric Titrators are compact, CE-marked benchtop instruments engineered for precision, repeatability, and regulatory compliance in routine titration workflows. Based on potentiometric endpoint detection—measuring potential difference across a reference and indicator electrode as titrant is incrementally added—the system delivers traceable, GLP-aligned results for acid-base, redox, precipitation, and complexometric titrations. Designed specifically for lean laboratory environments, the G10S/G20S platform integrates electrochemical measurement, automated dosing, and intelligent peripheral recognition into a unified architecture. Its core functionality adheres to ISO 8655 (volumetric equipment), ASTM E202 (standard test methods for titrimetric analysis), and supports alignment with USP , EP 2.2.20, and FDA 21 CFR Part 11 when used with LabX software and appropriate audit trail configuration.
Key Features
- One Click® intuitive interface: Customizable home-screen shortcuts enable rapid initiation of standardized titration methods without navigation layers—reducing operator training time and minimizing procedural deviation.
- Plug-and-Play electrode & burette recognition: Integrated RFID chips in METTLER TOLEDO-certified electrodes and titration units auto-configure calibration history, serial number, and expiration data upon connection—eliminating manual entry errors and ensuring full traceability.
- Compact modular design: Footprint under 300 × 400 mm accommodates integration with Rondolino autosamplers (up to 9 positions) or standalone operation; optimized for limited bench space without compromising mechanical stability or thermal drift control.
- Integrated solvent manager with LevelSens™: Fully automated reagent dispensing, waste aspiration, and bottle-level monitoring prevent overflow, reduce exposure risk, and maintain consistent delivery pressure across titrant batches.
- Pre-titration electrode diagnostics: Built-in impedance and offset verification ensures electrode readiness prior to each analysis—enforcing method compliance and reducing false endpoints caused by aging or fouled sensors.
Sample Compatibility & Compliance
The G10S/G20S supports aqueous and non-aqueous matrices—including acetic anhydride, glacial acetic acid, and ethanol-based systems—via compatible electrode families (e.g., DG115, InLab® Micro, and pH/Redox combination sensors). All hardware components meet IEC 61010-1 safety standards for laboratory electrical equipment. When operated with validated methods and connected peripherals, the system satisfies GLP documentation requirements for instrument qualification (IQ/OQ/PQ), including electronic signatures, user access controls, and secure data storage. The G20S variant extends compliance readiness through native integration with LabX Titration Express, enabling full 21 CFR Part 11–compliant audit trails, role-based permissions, and immutable result archiving.
Software & Data Management
G10S operates via embedded firmware with local method storage and real-time result display. G20S adds full LabX Titration Express connectivity—a Windows-based application that centralizes method development, instrument control, and data governance. LabX provides configurable report templates (PDF/CSV), cross-method search filters, version-controlled method libraries, and automatic backup to network drives or SQL databases. All raw sensor signals, dosing logs, and metadata (user ID, timestamp, environmental conditions) are retained with cryptographic integrity. Audit trail generation includes event type, initiator, timestamp, and pre-/post-change values—fully aligned with ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available).
Applications
- Pharmaceutical QC: Assay of active pharmaceutical ingredients (APIs), residual chloride in injectables, and Karl Fischer water content via coulometric or volumetric titration modes.
- Food & Beverage: Total acidity (TA), free SO₂ in wine, salt content in dairy products, and peroxide value in edible oils.
- Chemical Manufacturing: Neutralization number in lubricants, saponification value in fatty acids, and catalyst residue quantification in polymer synthesis.
- Environmental Testing: Alkalinity and hardness profiling in wastewater, cyanide determination in industrial effluents, and fluoride analysis in drinking water per EPA Method 300.0.
- Academic Research: Teaching labs benefit from guided method wizards, error simulation tools, and export-ready datasets for statistical validation (e.g., recovery studies, ruggedness testing).
FAQ
What distinguishes the G10S from the G20S model?
The G10S is a standalone unit with embedded touchscreen control and local data storage; the G20S adds Ethernet/Wi-Fi connectivity, LabX Titration Express compatibility, and full 21 CFR Part 11 support for regulated environments.
Can the system perform thermometric or photometric titrations?
No—this is a dedicated potentiometric titrator. Thermometric and photometric endpoints require separate METTLER TOLEDO platforms (e.g., T50/T70 or DP50 series).
Is calibration verification traceable to NIST standards?
Yes—electrode calibration certificates include traceability statements to national metrology institutes; certified reference materials (e.g., NIST SRM 186c pH buffers) are supported in all calibration routines.
How is method security enforced during multi-user operation?
LabX implements hierarchical user roles (Operator, Supervisor, Administrator), encrypted password policies, and mandatory electronic signatures for method modifications or result approvals.
What maintenance intervals are recommended for optimal performance?
Daily: Electrode rinsing and storage solution check; quarterly: Burette piston lubrication and seal inspection; annually: Full IQ/OQ verification using METTLER TOLEDO’s certified service kits and documented protocols.