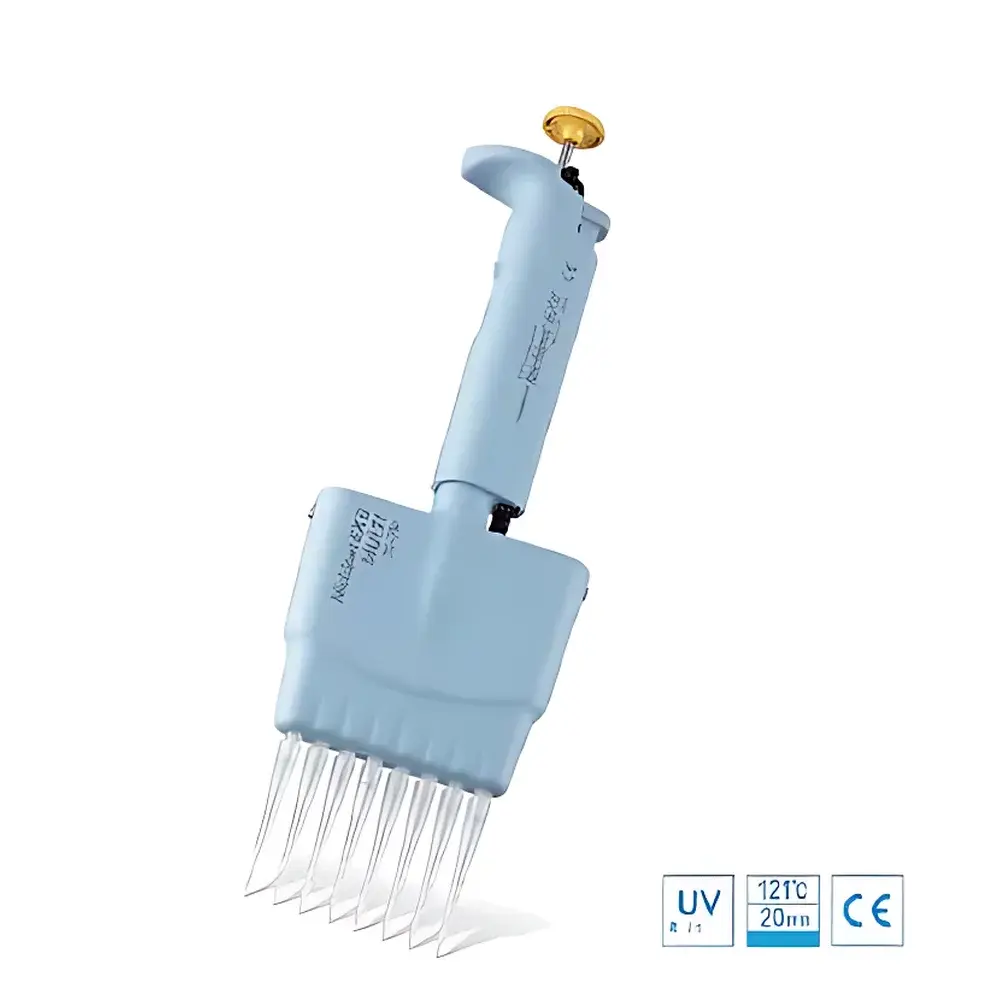
Nichiryo Nichipet EX II MULTI Digital Adjustable Multi-Channel Pipette
| Brand | Nichiryo |
|---|---|
| Origin | Japan |
| Model | EX II MULTI |
| Pipette Type | Manual Multi-Channel Pipette |
| Channels | 8 or 12 |
| Volume Range Options | 0.5–10 µL (V), 5–100 µL (S), 20–200 µL (L), 30–300 µL (K) |
| Minimum Increment | 0.01 µL (V), 0.1 µL (S), 0.2 µL (L), 1 µL (K) |
| Sterilization | Full-unit autoclavable at 121°C for 20 min |
| Tip Compatibility | BMT2-UT/UTWR/UTWRB, FUT/FUTRB, BMT2-SG/SGR/SGRB, FSE/FSERB, FAG/FAGRB, BMT2-K/KR/KRB |
| Material | UV-resistant polymer housing |
| Weight Reduction | ~30% vs. previous generation |
| Ergonomic Design | Rotatable 360° plunger knob, reduced plunger force, serviceable modular construction |
Overview
The Nichiryo Nichipet EX II MULTI is a precision-engineered manual multi-channel pipette designed for high-throughput, reproducible liquid handling in molecular biology, clinical diagnostics, and pharmaceutical quality control laboratories. Built upon Nichiryo’s decades of Japanese engineering heritage, the EX II MULTI employs a robust air-displacement mechanism with calibrated piston seals and low-friction internal pathways to ensure consistent volumetric accuracy across all channels—critical for applications such as ELISA plate filling, PCR setup, cell culture reagent dispensing, and serial dilution workflows. Unlike electronic pipettes, its fully mechanical operation eliminates battery dependency and electromagnetic interference concerns, making it ideal for use inside biosafety cabinets, cold rooms, and ISO-certified cleanrooms where instrument reliability and regulatory traceability are paramount.
Key Features
- Ergonomic Optimization: A 30% weight reduction versus the prior-generation EX I series significantly decreases operator fatigue during prolonged multi-well plate handling; the redesigned plunger mechanism reduces required actuation force by up to 40%, minimizing repetitive strain risk.
- Full Autoclavability: The entire pipette—including shaft, tip cone, and upper housing—is certified for steam sterilization at 121°C for 20 minutes (ISO 15195-compliant validation protocol), enabling strict adherence to GLP/GMP environmental control requirements without disassembly.
- Digital Volume Setting & Locking: Large, tactile rotary dials provide precise digital volume adjustment with visual LED confirmation (on compatible models); an integrated volume lock prevents accidental changes during transport or benchtop use.
- UV-Stable Construction: Housing components are fabricated from proprietary UV-resistant thermoplastics, maintaining structural integrity and dimensional stability after continuous exposure to germicidal UVC (254 nm) lamps—essential for biosafety cabinet-integrated workflows.
- Modular Service Architecture: Field-replaceable seals, O-rings, and spring assemblies allow rapid maintenance using standard calibration tools; no specialized technician support is required for routine recalibration or seal replacement.
- Multi-Range Flexibility: Four interchangeable volume ranges (V: 0.5–10 µL, S: 5–100 µL, L: 20–200 µL, K: 30–300 µL) accommodate diverse assay needs—from low-volume nucleic acid transfers to high-volume buffer dispensing—without cross-contamination risk between ranges.
Sample Compatibility & Compliance
The EX II MULTI is validated for use with a broad spectrum of sterile, low-retention, and filter tip formats—including BMT2-series universal-fit tips (UT/UTWR/UTWRB), FUT/FUTRB filtered tips, SGR/SGRB low-binding variants, and FAG/FAGRB wide-bore tips for viscous or foaming samples. All tip interfaces conform to ISO 8655-2:2022 dimensional tolerances and undergo batch-specific leak testing per ISO 8655-5. The pipette meets JIS T 9201:2020 (Japanese Industrial Standard for pipettes) and supports compliance with FDA 21 CFR Part 11 when used with validated electronic logbooks for calibration records. Its autoclave certification aligns with ISO 17664 for reprocessing of reusable laboratory devices.
Software & Data Management
As a manually operated instrument, the EX II MULTI does not incorporate embedded firmware or wireless connectivity. However, it integrates seamlessly into regulated environments via documented calibration workflows: each unit ships with a factory-issued calibration certificate traceable to NMIJ/AIST (National Metrology Institute of Japan), and optional calibration verification kits (NICH-VER-01) enable in-house performance checks per ISO 8655-6. Calibration logs—including date, operator ID, reference standard used, and deviation values—are maintained in paper-based or LIMS-linked electronic formats compliant with ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available).
Applications
- High-density microplate preparation for qPCR, digital PCR, and next-generation sequencing library construction
- Reproducible dispensing of enzyme cocktails, antibodies, and cell culture media in 96- and 384-well formats
- Standard curve generation and QC sample dilution in clinical chemistry and immunoassay validation studies
- Cell-based assay setup requiring uniform seeding density across replicates
- Regulated release testing under USP , , and EU GMP Annex 1 environmental monitoring protocols
FAQ
Is the EX II MULTI compatible with third-party tips?
Yes—when tip dimensions comply with ISO 8655-2:2022 specifications, including outer diameter, taper angle, and sealing surface geometry.
Can volume settings be locked during operation?
Yes—the integrated mechanical volume lock prevents inadvertent adjustment during pipetting or storage.
What is the recommended recalibration interval?
Nichiryo recommends verification every 3 months for daily use, or prior to critical assays, following ISO 8655-6 guidelines.
Does the pipette require special cleaning agents?
No—routine decontamination with 70% ethanol or isopropanol is sufficient; avoid chlorine-based or strong oxidizing solutions.
Is there a service program for international users?
Yes—authorized Nichiryo service centers in North America, EMEA, and APAC provide certified recalibration, seal replacement, and metrological verification with NIST-traceable documentation.