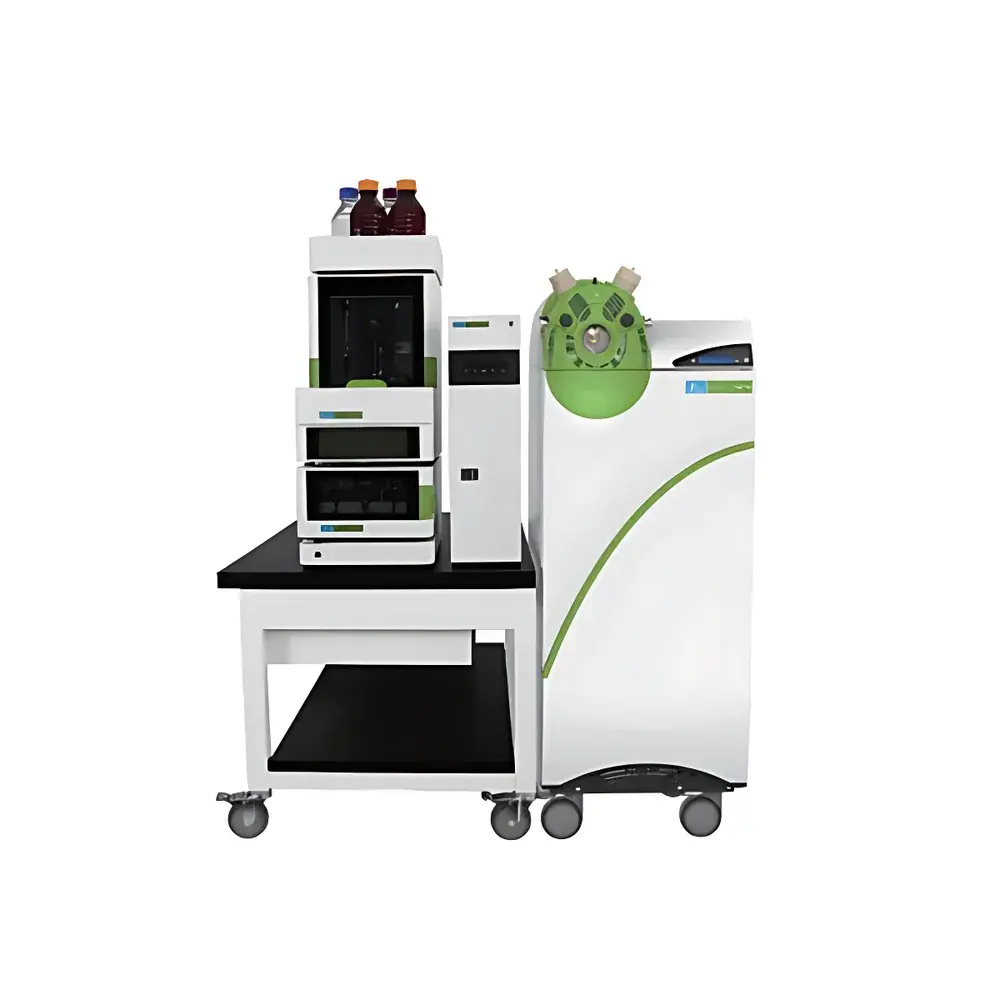
PerkinElmer QSight 500 Triple Quadrupole Liquid Chromatography Tandem Mass Spectrometer
| Brand | PerkinElmer |
|---|---|
| Origin | Singapore |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Import Status | Imported |
| Model | QSight 500 |
| Instrument Type | Triple Quadrupole (TQ) LC/MS/MS |
| Application Scope | General-purpose quantitative and confirmatory analysis in regulated and research laboratories |
Overview
The PerkinElmer QSight 500 is a high-performance triple quadrupole liquid chromatography tandem mass spectrometer engineered for robust, reproducible, and regulatory-compliant quantitative analysis across diverse sample matrices. Operating on the proven principles of electrospray ionization (ESI) and atmospheric pressure chemical ionization (APCI), the system leverages parallel reaction monitoring (PRM)-capable scanning modes and optimized collision-induced dissociation (CID) in Q2 to deliver exceptional selectivity, sensitivity, and dynamic range. Designed as the latest evolution of the QSight platform, it integrates seamlessly with the QSight LX50 UHPLC system—enabling sub-2-µm particle separations, low dispersion flow paths, and precise gradient delivery—to form a fully coordinated LC/MS/MS workflow. Its architecture supports both targeted quantitation (e.g., MRM, dMRM) and qualitative screening (e.g., enhanced product ion scans), making it suitable for routine compliance testing, method development, and multi-residue analysis in food safety, pharmaceutical quality control, environmental monitoring, and clinical research laboratories.
Key Features
- Patented self-cleaning ion optics design minimizes downtime by reducing routine maintenance intervals and mitigating signal drift from matrix buildup.
- HSID™ (High Sensitivity Ion Drive) interface ensures stable, high-efficiency ion transmission across wide flow-rate ranges (0.05–1.5 mL/min), supporting both conventional HPLC and UHPLC applications without hardware reconfiguration.
- Dual-source configuration (ESI/APCI) enables rapid, software-controlled source switching within a single analytical run—eliminating manual intervention and preserving column integrity.
- UniField Detector™ provides linear response over five orders of magnitude with <1% relative standard deviation (RSD) for replicate injections, meeting stringent ICH Q2(R2) precision requirements.
- EDIT™ (Enhanced Dual Ionization Technology) source delivers improved ionization efficiency for thermally labile and low-proton-affinity compounds, including per- and polyfluoroalkyl substances (PFAS), polar pesticides, and endogenous metabolites.
- Modular hardware architecture allows field-upgradable components—including collision cell gas control, detector voltage tuning, and vacuum pump sequencing—ensuring long-term adaptability to evolving regulatory and methodological demands.
Sample Compatibility & Compliance
The QSight 500 accommodates complex biological fluids (plasma, urine, tissue homogenates), environmental extracts (soil leachates, wastewater), food digests (meat, dairy, cereals), and pharmaceutical formulations (tablet extracts, stability samples). It complies with key international standards including ASTM D7968 (PFAS in soil), EPA Method 1633 (PFAS in water), USP <621> (chromatographic system suitability), and ISO/IEC 17025:2017 (general competence requirements for testing laboratories). The system supports 21 CFR Part 11-compliant audit trails, electronic signatures, and role-based access control when operated with PerkinElmer’s Simplicity™ 2.0 software—facilitating GLP and GMP-aligned workflows in regulated environments.
Software & Data Management
Simplicity™ 2.0 software provides an integrated environment for instrument control, data acquisition, processing, and reporting. It features automated peak integration with customizable retention time windows, batch processing with template-driven report generation, and built-in compliance tools such as electronic lab notebook (ELN) export, raw data archiving in open-format .d directories, and secure user authentication. Method validation modules support linearity, accuracy, precision, LOD/LOQ determination, and carryover assessment—all traceable to NIST-traceable reference standards. Data files are compatible with third-party platforms including OpenLab CDS, Chromeleon, and Skyline for cross-platform verification.
Applications
- Quantitative residue analysis of PFAS compounds (e.g., PFOA, PFOS, GenX) in drinking water and biosolids per EPA Draft Method 1633.
- Pharmaceutical impurity profiling and genotoxic impurity quantification in accordance with ICH M7 guidelines.
- Multi-analyte pesticide screening in fruits and vegetables using QuEChERS-extracted samples.
- Endogenous biomarker quantification (e.g., cortisol, vitamin D metabolites) in clinical diagnostics.
- Environmental contaminant monitoring, including brominated flame retardants (PBDEs), phthalates, and pharmaceuticals in wastewater effluents.
FAQ
What regulatory standards does the QSight 500 support for PFAS analysis?
It meets the performance criteria outlined in EPA Draft Method 1633 and ASTM D7968, with validated MRM transitions, internal standard compensation, and matrix-matched calibration protocols.
Can the system operate unattended for extended batches?
Yes—integrated autosampler cooling (4–40 °C), column oven temperature stability (±0.1 °C), and predictive maintenance alerts enable >72-hour continuous operation with minimal operator intervention.
Is method transfer possible from other triple quadrupole platforms?
Simplicity™ 2.0 includes migration wizards that convert common MRM methods from Thermo TSQ, Sciex 6500+, and Agilent 6470 formats into native QSight syntax with automatic dwell time optimization and collision energy adjustment.
How is data integrity ensured during audits?
Full 21 CFR Part 11 compliance is achieved through immutable audit trails, electronic signature enforcement, and cryptographic hashing of raw data files at acquisition—verified during FDA or EMA inspection readiness reviews.