Shimadzu GCMS-QP2010 Plus Used Gas Chromatography-Mass Spectrometry System
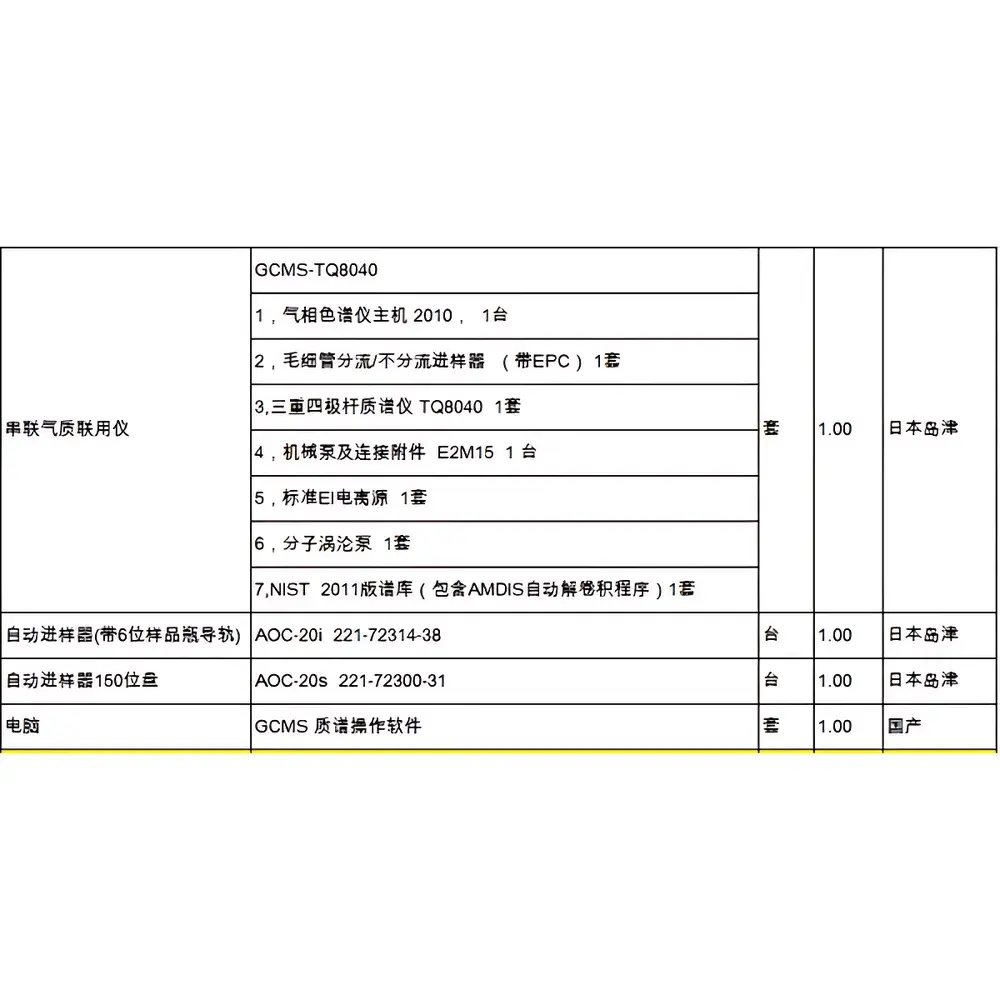
| Brand | Shimadzu |
|---|---|
| Origin | Japan |
| Model | GCMS-QP2010 Plus |
| Configuration | GC-2010 Plus GC, TD-20 Thermal Desorber, AOC-20s 100-position Autosampler Tray, AOC-20i Autosampler, LabSolutions Software Workstation, Dual-inlet Turbo Molecular Pump (≥360 L/s), Vacuum System |
| Warranty | 90 days post-installation verification |
Overview
The Shimadzu GCMS-QP2010 Plus is a high-performance gas chromatography–mass spectrometry (GC-MS) system engineered for precise, reproducible trace-level analysis across environmental monitoring, food safety testing, pharmaceutical quality control, and forensic toxicology. Built on Shimadzu’s proven QP platform, this instrument integrates a robust quadrupole mass spectrometer with an advanced capillary GC system—delivering reliable electron ionization (EI) and optional chemical ionization (CI) capabilities. Its core architecture employs a shielded ion source design that minimizes thermal and electrical interference from the filament, ensuring uniform ion source temperature distribution and enhanced ion transmission efficiency. The dual-inlet turbo molecular pump (≥360 L/s) establishes independent differential vacuum stages for the ion source and mass analyzer, achieving operational base pressures below 1 × 10⁻⁵ Pa—critical for maintaining signal stability during extended acquisition sequences and low-abundance analyte detection.
Key Features
- Shielded ion source with optimized thermal management and reduced filament-induced background noise
- Dual-inlet turbo molecular pumping system enabling simultaneous high-vacuum operation in ion source and mass analyzer compartments
- FASST (Fast Automated Scan/SIM Transition) mode allowing concurrent full-scan and selected-ion monitoring (SIM) data acquisition within a single run
- High-speed scanning capability up to 10,000 u/sec—preserving spectral fidelity even for narrow, high-efficiency GC peaks (e.g., <1 s width at base)
- Molybdenum quadrupole mass filter with pre-rod assembly to suppress ion scattering and mitigate contamination-related signal drift
- Extended mass range up to m/z 1090—supporting structural characterization of high-molecular-weight compounds including brominated flame retardants (e.g., PBDEs) and certain pesticides
- AART (Automatic Adjustment of Retention Time) algorithm using n-alkane retention indices to dynamically correct retention time shifts caused by column aging or replacement
- Direct insertion probe (DI) interface enabling rapid qualitative MS analysis of thermally labile, non-volatile, or solid-phase samples without GC separation
- LabSolutions GC/MS software suite compliant with GLP/GMP documentation requirements—including audit trail, user access control, and electronic signature support per FDA 21 CFR Part 11
Sample Compatibility & Compliance
The GCMS-QP2010 Plus accommodates diverse sample introduction methods: split/splitless capillary injection, headspace analysis (via optional HS-20 or TD-20 thermal desorption), direct probe analysis, and purge-and-trap interfaces. It supports standard 0.25–0.53 mm ID fused silica columns and is compatible with common stationary phases (e.g., DB-5ms, HP-5MS, Rtx-CLP). Regulatory alignment includes method validation support per EPA Methods 502.2, 524.2, 8270D, and 8321B; ISO 17025-compliant operation when integrated into accredited laboratory workflows; and compliance-ready data handling for ISO/IEC 17025, USP , and EU Annex 11 environments.
Software & Data Management
LabSolutions GC/MS provides integrated acquisition, processing, and reporting tools—including Compound Composer (preloaded with >900 environmental contaminants and food safety markers), Method Package libraries for multi-residue pesticide, PAH, PCB, and dioxin analysis, and automated calibration curve generation with internal standard correction. All raw data files (.qgd) are stored in vendor-neutral formats supporting third-party reprocessing. Audit trails record operator actions, parameter changes, and report generation events. Data integrity safeguards include write-protected archives, timestamped metadata, and role-based permissions—fully aligned with 21 CFR Part 11 Subpart B requirements for electronic records and signatures.
Applications
This system is routinely deployed in regulatory laboratories for residual solvent quantification in pharmaceuticals (ICH Q3C), pesticide residue screening in fruits and vegetables (EU MRL enforcement), volatile organic compound (VOC) profiling in drinking water (EPA 524.2), polycyclic aromatic hydrocarbon (PAH) analysis in soil and sediment (EPA 8270D), and targeted metabolite identification in clinical research. Its DI capability supports rapid confirmation of synthetic cannabinoids or novel psychoactive substances (NPS) directly from seized material. When coupled with Prep-Q (GPC-GC-MS), it enables automated cleanup and analysis of complex food matrices—reducing solvent consumption by >60% versus conventional SPE-GC-MS workflows.
FAQ
Is this unit refurbished or used-as-is?
This GCMS-QP2010 Plus system has undergone full functional verification at SpectraLab Scientific’s ISO 17025-accredited calibration facility, including vacuum integrity testing, mass axis calibration, sensitivity verification (using DFTPP), and detector linearity assessment. It is sold as a verified pre-owned instrument with documented performance history.
What consumables and spare parts are included?
The system ships with all original accessories: GC-2010 Plus oven module, QP2010 Plus MS detector, TD-20 thermal desorber, AOC-20i autosampler with AOC-20s 100-position tray, LabSolutions v5.8x software license, dedicated PC workstation, vacuum lines, carrier gas fittings, and a certified oil-lubricated rotary vane backing pump.
Does the 90-day warranty cover labor and parts?
Yes—the warranty includes onsite technical support, remote diagnostics, and replacement of defective components attributable to pre-delivery condition. Extended service plans covering preventive maintenance and annual recalibration are available upon request.
Can this system be upgraded to support GCxGC or HRMS configurations?
While the QP2010 Plus platform does not support native time-of-flight or magnetic sector upgrades, it is fully compatible with Shimadzu’s MDGC-2010 multidimensional GC interface and can be retrofitted with newer-generation AOC-5000 series autosamplers or HS-20 headspace units without hardware modification.
Is LabSolutions software provided with full licensing rights?
Yes—the system includes a perpetual, transferable LabSolutions GC/MS software license with current version activation and access to Shimadzu’s official software update portal for patch releases and security updates.