Shimadzu LabSolutions MD Method Development Software
| Brand | Shimadzu |
|---|---|
| Origin | Japan |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Product Origin | Imported |
| Model | LabSolutions MD |
| Instrument Category | Conventional High-Performance Liquid Chromatography (HPLC) System Software |
| Application Scope | General-Purpose Analytical Method Development for HPLC/UHPLC |
Overview
Shimadzu LabSolutions MD is a dedicated method development software platform engineered for systematic, science-driven analytical method optimization in high-performance liquid chromatography (HPLC) and ultra-high-performance liquid chromatography (UHPLC) environments. Built upon the Analytical Quality by Design (AQbD) framework—aligned with ICH Q8(R3), Q9, and Q14 guidelines—the software enables chromatographers to transition from empirical trial-and-error approaches to structured, risk-based method development. It integrates experimental design (DoE), automated chromatographic data processing, retention modeling, design space visualization, and robustness assessment into a single, validated software environment. LabSolutions MD operates natively within the LabSolutions ecosystem, ensuring full bidirectional communication with Shimadzu LC modules (e.g., Nexera X2, Prominence-i), detectors (SPD-M30A, RF-20Axs), and autosamplers—eliminating file-based data handoffs and preserving audit trail integrity.
Key Features
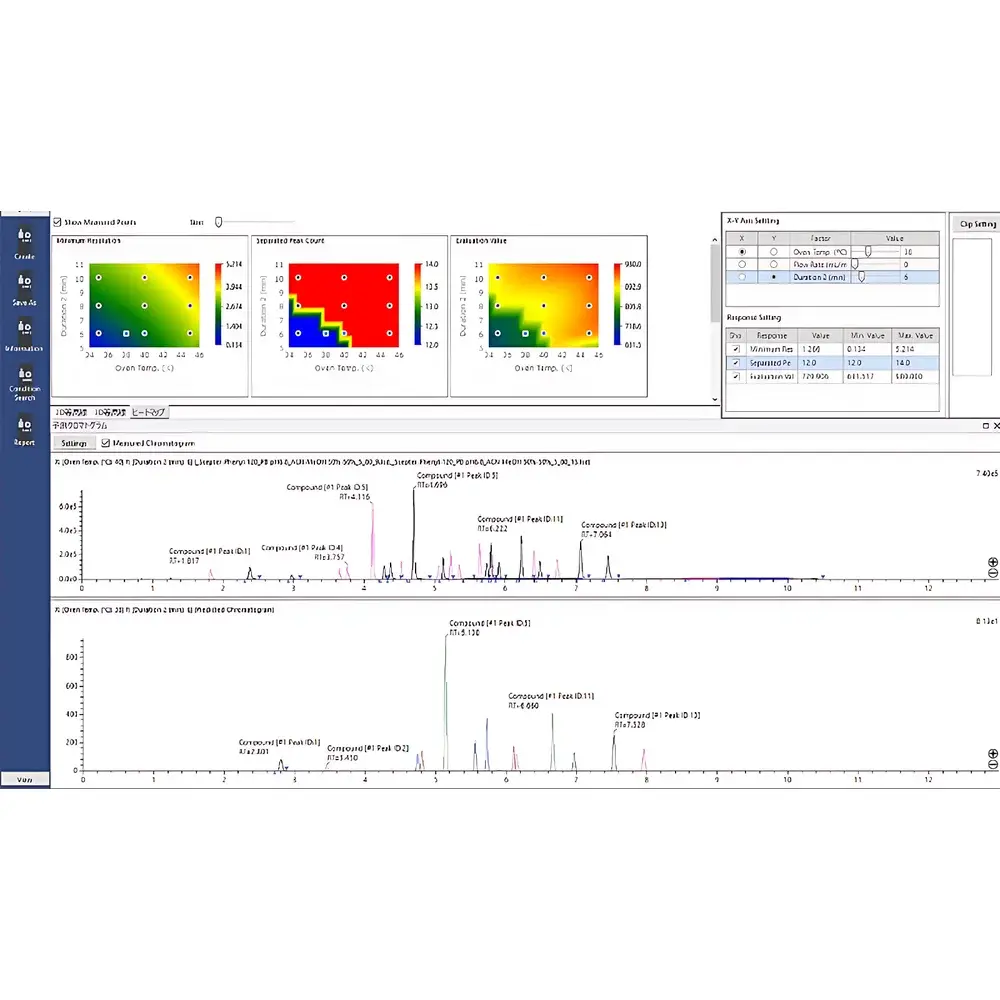
- Integrated DoE Workflow Engine: Supports full-factorial, Box-Behnken, and central composite designs—configurable via intuitive wizards. Automatically generates batch tables with method files, column equilibration steps, and system suitability blanks per DoE matrix point.
- Automated Peak Tracking: Uses UV spectral similarity (correlation coefficient ≥0.995), peak height/area consistency, and retention time drift compensation to track target analytes across varying gradient, temperature, and mobile phase conditions—reducing manual peak annotation effort by >70% in multi-condition studies.
- Design Space Visualization: Constructs interactive 2D/3D response surface plots (e.g., resolution vs. pH & organic modifier %; tailing factor vs. column temperature & flow rate). Enables identification of robust operating regions compliant with ICH Q5A and USP system suitability requirements.
- In Silico Chromatogram Simulation: Predicts elution profiles under untested parameter combinations using empirically derived retention models—supporting prospective robustness evaluation prior to physical experimentation.
- End-to-End Data Traceability: All DoE definitions, raw chromatograms, peak tables, design space models, and final method reports are stored in a single project database with immutable timestamps, user attribution, and version-controlled metadata—meeting FDA 21 CFR Part 11 and EU Annex 11 electronic record requirements.
Sample Compatibility & Compliance
LabSolutions MD is validated for use with Shimadzu HPLC/UHPLC systems employing reversed-phase, HILIC, ion-exchange, and size-exclusion chromatography modes. It supports data acquisition from PDA, fluorescence, and single-wavelength UV-Vis detectors. The software complies with ISO/IEC 17025:2017 (clause 7.7 on data control), adheres to ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available), and supports GLP/GMP-compliant workflows through configurable electronic signatures, audit trail review tools, and report export in PDF/A-1b format. All method development outputs—including design space documentation and robustness test summaries—meet ICH Q5A stability-indicating method expectations.
Software & Data Management
LabSolutions MD utilizes a centralized project database architecture (.lsmdproj) that stores all method development artifacts—including DoE matrices, instrument methods, sequence tables, processed chromatograms, peak integration results, and design space models—in a relational structure. Database backups support automated daily archiving with SHA-256 hash verification. Reporting modules generate customizable PDF reports containing executive summaries, DoE summary statistics (ANOVA, p-values, R²), contour plots, simulation validation metrics (RMSE ≤ 0.2 min retention error), and compliance statements. Integration with LabSolutions CDS allows direct launch of validated QC methods derived from the design space without reconfiguration.
Applications
- Development of stability-indicating assays for pharmaceutical small molecules and biologics (per ICH Q5C)
- Optimization of chiral separations under varying temperature and mobile phase composition
- Robustness testing of dissolution methods per USP
- Method transfer between HPLC and UHPLC platforms using predictive retention modeling
- Supporting regulatory submissions (e.g., NDA, MAA) with documented AQbD evidence packages
FAQ
Is LabSolutions MD compatible with non-Shimadzu HPLC hardware?
No. LabSolutions MD requires native communication with Shimadzu LC control modules and is not designed for third-party instrument integration.
Does the software support multivariate robustness assessment per ICH Q5A?
Yes. It enables simultaneous evaluation of ≥3 critical method parameters (e.g., pH, organic %, column temperature) and quantifies their interaction effects on resolution and tailing factor.
Can design space models be exported for use in external statistical packages?
Yes. Response surface equations and coefficient matrices are exportable in CSV format for further analysis in JMP or Minitab.
How is data integrity maintained during long-term method development projects?
All user actions—including method edits, peak reintegration, and design space recalculations—are logged in an encrypted, read-only audit trail with operator ID, timestamp, and before/after values.
Is 21 CFR Part 11 compliance enabled by default?
Yes. Electronic signature enforcement, audit trail review permissions, and report digital signing are preconfigured and validated per Shimadzu’s Part 11 Implementation Guide v3.2.