Shimadzu LCMS-8080 Triple Quadrupole Liquid Chromatography-Mass Spectrometer (Refurbished Demo Unit)
| Brand | Shimadzu |
|---|---|
| Origin | Japan |
| Instrument Type | Triple Quadrupole LC-MS/MS System |
| Model | LCMS-8080 |
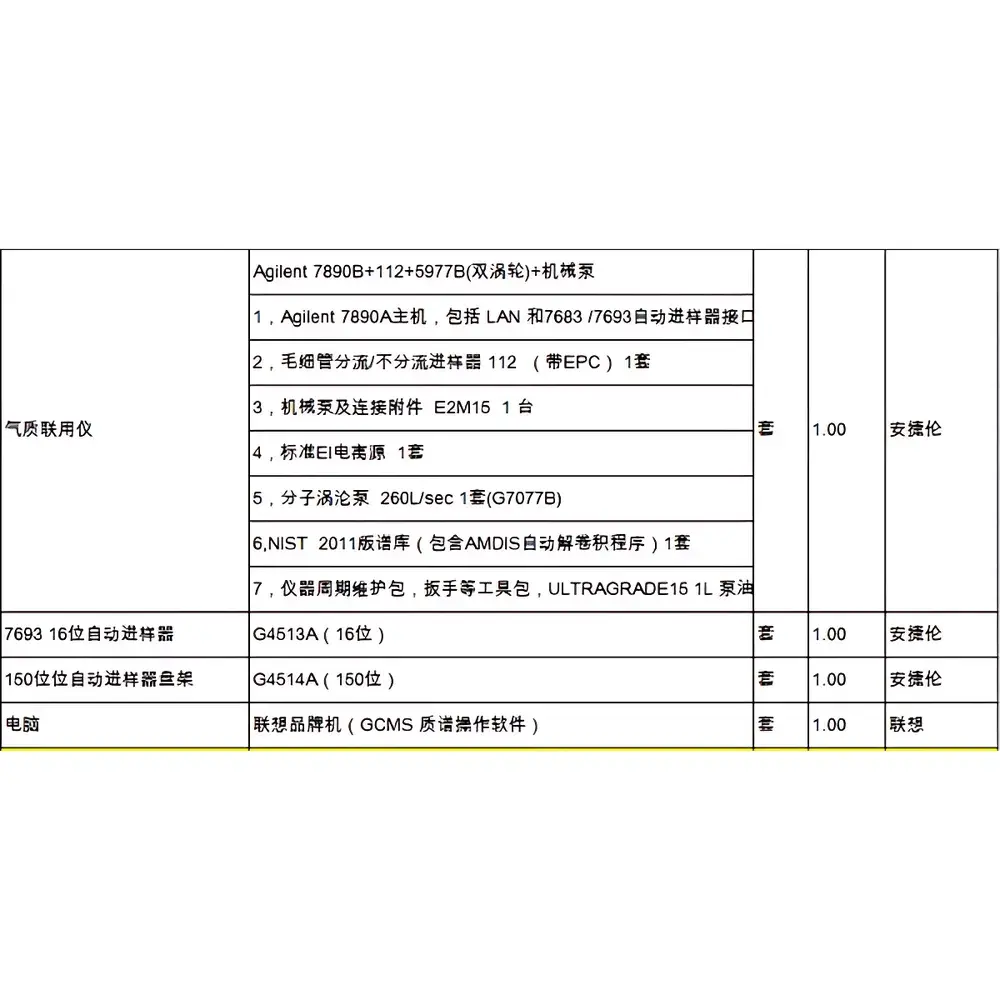
| Configuration | Prominence UFLC + LCMS-8080 + Vacuum Pump + Autosampler Injection Pump + Integrated Control PC with LabSolutions Software |
| Age | 3–4 years |
| Warranty | 6 months |
| Compliance | Designed to meet ISO/IEC 17025-relevant operational requirements for quantitative bioanalysis and routine QC labs |
Overview
The Shimadzu LCMS-8080 is a high-performance triple quadrupole liquid chromatography–tandem mass spectrometry (LC-MS/MS) system engineered for robust, reproducible quantitative analysis in regulated and research environments. Built upon Shimadzu’s proven UFLC (Ultra-Fast Liquid Chromatography) platform and advanced ion optics architecture, the LCMS-8080 employs electrospray ionization (ESI) and atmospheric pressure chemical ionization (APCI) sources to deliver exceptional sensitivity—particularly critical for trace-level quantitation of small molecules in complex matrices. Its core design emphasizes ion transmission efficiency via heated electrospray ionization (HESI), a high-sensitivity ion guide (HSID), and laminar-flow ion transport pathways that minimize ion loss across the interface and mass analyzer stages. This configuration enables sub-pg/mL detection limits for many pharmaceutical analytes under optimized conditions—making it suitable for pharmacokinetic (PK), toxicokinetic (TK), and biomarker validation workflows where precision, linearity, and low-end accuracy are non-negotiable.
Key Features
- Triple quadrupole mass analyzer with unit-mass resolution and fast polarity switching (≤ 15 ms)
- UFLC front-end delivering ≤ 0.001 min retention time repeatability and gradient precision down to 0.1% RSD
- Heated ESI source with coaxial sheath gas and adjustable desolvation temperature (up to 300 °C) for enhanced ionization stability
- High-Sensitivity Ion Guide (HSID) with active collision cell cooling for reduced chemical noise and improved signal-to-noise ratios
- Integrated vacuum system with dual-stage turbomolecular pump and backing pump for stable high-vacuum operation (≤ 1×10⁻⁶ Pa in Q3 region)
- Dedicated control PC preinstalled with LabSolutions LCMS software (v5.9 or later), supporting AIA-compliant data acquisition, batch processing, and 21 CFR Part 11–ready audit trails when configured with appropriate user access controls
Sample Compatibility & Compliance
The LCMS-8080 supports broad sample compatibility across biological fluids (plasma, serum, urine), tissue homogenates, food extracts, environmental water samples, and synthetic chemical standards. It accommodates standard 2–5 µL injection volumes and interfaces seamlessly with reversed-phase, HILIC, and mixed-mode LC columns (1.0–4.6 mm ID, 50–250 mm length). From a regulatory standpoint, the system operates within parameters aligned with ICH M10 guidance on bioanalytical method validation and supports GLP-compliant workflows when paired with validated SOPs, instrument qualification (IQ/OQ/PQ), and electronic record integrity controls. While not supplied with full GMP validation documentation, the platform is routinely deployed in laboratories adhering to ISO/IEC 17025:2017 for testing and calibration competence.
Software & Data Management
LabSolutions LCMS provides an integrated environment for method development, acquisition, processing, and reporting. Key modules include MultiReaction Monitoring (MRM) optimization tools, peak integration with customizable baseline algorithms, calibration curve fitting (linear, quadratic, weighted), and automated QC flagging based on predefined acceptance criteria (e.g., %CV, %bias, IS response stability). Raw data files (.qgd) are stored in vendor-native format; export to open formats (mzML, CSV) is supported for third-party processing. The software architecture permits role-based user accounts, electronic signatures, and audit trail generation—enabling alignment with FDA 21 CFR Part 11 requirements when deployed on domain-joined Windows systems with appropriate IT governance policies.
Applications
This refurbished LCMS-8080 demo unit has been verified for use in multiple high-value application domains: therapeutic drug monitoring (TDM) in clinical laboratories; residue analysis of pesticides and veterinary drugs in food safety testing; environmental screening of persistent organic pollutants (POPs); impurity profiling and stability-indicating assays during pharmaceutical development; and high-throughput compound quantification in early-stage ADME studies. Its combination of speed, specificity, and quantitative fidelity makes it especially well-suited for multi-analyte panels requiring simultaneous detection of structurally diverse compounds without compromising sensitivity or dynamic range (typically >4 orders of magnitude).
FAQ
Is this unit fully refurbished and tested prior to shipment?
Yes—this LCMS-8080 was originally a factory-fresh demonstration system, unpacked only for functional verification at SpectraLab Scientific. All modules underwent comprehensive performance verification including sensitivity checks (reserpine S/N ≥ 10,000:1 at 1 pg/µL), mass accuracy (< 0.2 Da), retention time stability (< 0.005 min RSD), and MRM transition reproducibility (< 5% RSD over 10 injections).
What does the 6-month warranty cover?
The warranty includes parts and labor for all core components: UFLC pumps, autosampler, ESI source, vacuum system, detector electronics, and mass analyzer. Exclusions include consumables (columns, ESI tips, pump seals), accidental damage, or misuse outside specified operating conditions.
Can this system be qualified for GLP or 21 CFR Part 11 compliance?
Yes—the hardware and LabSolutions software support IQ/OQ/PQ execution. Customers are responsible for implementing site-specific validation protocols, network security configurations, and electronic signature policies required for formal regulatory submissions.
Is original Shimadzu documentation included?
Yes—electronic copies of the user manuals, maintenance guides, and factory calibration certificates are provided. Physical printed manuals are not included unless requested separately.
Do you offer installation and on-site training?
Remote startup assistance and basic software training are included. On-site installation, qualification support, and advanced method development training are available as optional services at additional cost.