testo 190 CFR Data Logger and Validation System Model 0554 1901
| Brand | testo |
|---|---|
| Origin | Germany |
| Model | 0554 1901 |
| Compliance | 21 CFR Part 11 compliant software platform |
| System Requirements | Windows 7 (32/64-bit), Windows 8, Windows 10 or newer |
| Included Software | testo 190 CFR Software (locally installed, registration required) |
| Maximum Simultaneous Loggers | 8 (programming & readout), 254 (validation-scale data aggregation) |
| Key Functionality | F₀ calculation, photo-annotation of measurement locations, graphical + tabular data visualization, audit-trail-enabled reporting |
Overview
The testo 190 CFR Data Logger and Validation System (Model 0554 1901) is a purpose-engineered solution for regulated thermal process validation in pharmaceutical, biotechnology, and medical device manufacturing environments. Designed to meet the stringent documentation and traceability requirements of FDA 21 CFR Part 11, this system integrates rugged, high-accuracy wireless or wired temperature and pressure data loggers with a locally installed, validated software platform. It operates on the principle of time-synchronized, multi-channel thermal mapping—capturing continuous, timestamped measurements during critical processes such as autoclave sterilization, lyophilization (freeze-drying), environmental chamber qualification, and cleanroom monitoring. Unlike generic data loggers, the testo 190 CFR architecture embeds electronic signature capability, role-based user access control, immutable audit trails, and automated report generation directly into its workflow—eliminating manual data transcription, Excel-based post-processing, and associated risk of human error. The system is not a standalone hardware unit but a tightly coupled hardware-software ecosystem engineered for compliance-driven validation lifecycle management.
Key Features
- Full 21 CFR Part 11 compliance out-of-the-box: digital signatures, hierarchical user management (admin/operator/auditor roles), and tamper-evident audit trail recording all system events—including login attempts, parameter changes, report generation, and data export actions.
- Simultaneous programming and readout of up to eight testo 190-series CFR loggers via USB or docking station—enabling rapid setup and retrieval without sequential bottlenecks.
- Scalable validation support: software architecture supports aggregation and synchronized analysis of measurement data from up to 254 individual loggers across multiple process cycles—essential for large-scale equipment qualification (IQ/OQ/PQ) and media fill studies.
- Integrated F₀ (Fo) value calculation per ICH Q5C and USP , with configurable Z-value, reference temperature, and hold-time thresholds—automatically applied to temperature-time profiles during post-process evaluation.
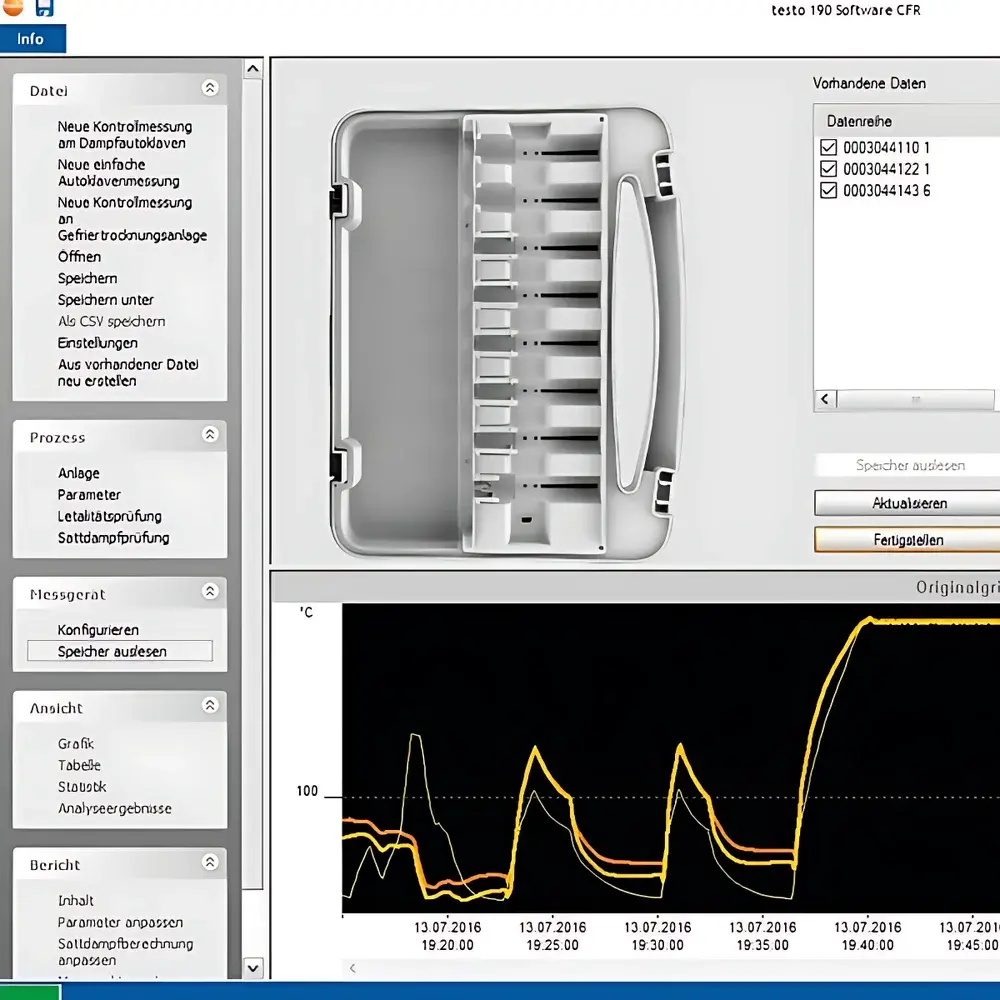
- Geo- and context-aware data annotation: users may import and embed photographs of autoclaves, freeze dryers, or room layouts directly into reports; measurement channels are visually mapped onto images to clarify spatial sensor placement and thermal gradients.
- Zero-export reporting: all visualizations—including real-time trend graphs, deviation heatmaps, pass/fail overlays, and statistical summaries—are generated natively within the software and exported exclusively as password-protected, digitally signed PDFs meeting ALCOA+ principles.
Sample Compatibility & Compliance
The testo 190 CFR system is validated for use with testo’s Class A-certified temperature loggers (e.g., testo 190-T1, T2, PT100 variants) and pressure-capable models (e.g., testo 190-P1), all conforming to EN 12830, VDI/VDE 2655 Blatt 2, and ASTM E2810 standards for thermal validation instrumentation. Hardware meets IP65 ingress protection where applicable and operates reliably across −20 °C to +125 °C (depending on probe configuration). The entire solution—including firmware, software binaries, installation packages, and default report templates—undergoes periodic vendor-supplied verification against 21 CFR Part 11 Annex 11 and EU GMP Annex 11 requirements. Installation Qualification (IQ) and Operational Qualification (OQ) protocols are provided by testo upon request, supporting GxP-aligned deployment in FDA-, EMA-, and PMDA-regulated facilities.
Software & Data Management
The testo 190 CFR Software is a Windows-native application requiring local installation (no cloud dependency), ensuring full data sovereignty and network isolation per security policies. It employs AES-256 encryption for stored data and leverages Windows’ native credential manager for secure authentication. All user actions generate timestamped, non-erasable entries in the audit trail—including creation/modification of calibration certificates, adjustment of alarm thresholds, and final report approval. Data integrity is enforced through checksum validation at every read cycle, and raw binary logger files remain unaltered throughout the workflow. The software supports version-controlled template management for SOP-aligned report structures and permits custom field injection (e.g., batch numbers, operator IDs, equipment tags) to maintain contextual traceability across validation protocols.
Applications
This system is routinely deployed in: steam sterilization cycle development and routine release testing; lyophilizer shelf temperature uniformity mapping; tunnel washer and depyrogenation oven qualification; cold chain logistics monitoring for clinical trial materials; HVAC system temperature/humidity mapping in Grade A/B cleanrooms; and stability chamber performance verification per ICH Q1. Its deterministic timing engine ensures sub-second synchronization across distributed loggers—critical for detecting transient thermal excursions during critical hold phases. Integration with enterprise systems (e.g., LIMS, MES) is achievable via standardized CSV/XML export—but only after explicit user-initiated, audit-trail-logged export authorization.
FAQ
Does the testo 190 CFR Software require annual re-validation?
Yes—per industry best practice and regulatory expectation, the software must undergo periodic re-verification following any OS update, patch installation, or change in operational environment; testo provides documented re-qualification checklists.
Can third-party loggers be used with this system?
No—the 190 CFR Software is exclusively validated for use with testo-branded 190-series loggers; integration with non-testo devices voids 21 CFR Part 11 compliance claims.
Is remote access supported for real-time monitoring?
No—real-time telemetry is intentionally excluded to preserve data integrity and prevent unauthorized modification; all data retrieval occurs post-process via physical connection or secure offline transfer.
What happens if a user forgets their password?
Password recovery is disabled by design; locked accounts must be reset by an authorized system administrator with dual-control approval—ensuring continuity of accountability per 21 CFR Part 11 §11.200(c).
Are raw data files exportable in unprocessed format?
Yes—raw .tdf (testo data format) files can be exported, but each export action is permanently logged in the audit trail with user ID, timestamp, and destination path; the original file remains cryptographically sealed within the software database.