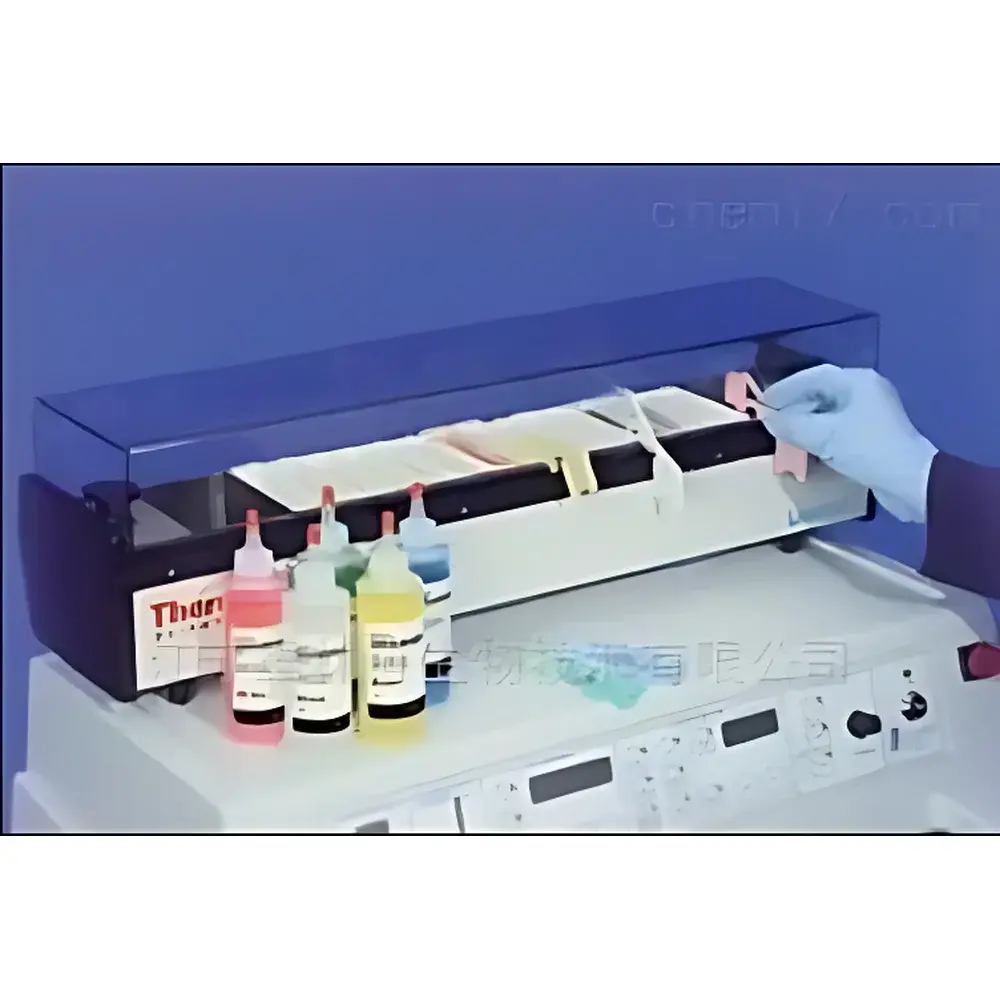
Thermo Fisher Linistat Linear Staining System
| Brand | Thermo Fisher |
|---|---|
| Origin | United Kingdom |
| Model | Linistat |
| Automation Level | Semi-Automatic |
| Instrument Type | Non-Rotary Linear Stainer |
| Staining Stations | 14 (20 sec/station) |
| Throughput | ≤5 min per batch (13 slides) |
| Slide Capacity | 13-slide collection tank |
| Water Rinse Tank | Position-adjustable |
| Footprint | Compact, designed for placement atop cryostats |
| Compatible Stain | Optimized for Hematoxylin-Eosin (H&E) staining of frozen sections |
| Compatible Reagents | Thermo Scientific Cryochrome™ embedding media and color-coded slide holders |
Overview
The Thermo Fisher Linistat Linear Staining System is an engineered solution for high-integrity, rapid hematoxylin-eosin (H&E) staining of frozen tissue sections in pathology laboratories. Operating on a non-rotary linear transport principle, the system advances glass slides sequentially through 14 precisely timed reagent stations—each with a fixed 20-second dwell time—enabling full-cycle staining in under five minutes. Its compact footprint and low-profile design allow direct integration atop standard clinical cryostats, eliminating inter-instrument transfer delays and minimizing benchtop spatial demand. Unlike rotary or orbital stainers, the Linistat employs gravity-assisted, linear slide carriage mechanics with positive-position indexing, ensuring consistent immersion depth and reproducible reagent contact across all slides in a batch. This architecture supports GLP-aligned workflow standardization while maintaining compatibility with manual override protocols for troubleshooting or validation runs.
Key Features
- 14 independently controlled staining stations with programmable 20-second dwell intervals, enabling precise kinetic control over hematoxylin oxidation, eosin binding, and differentiation steps
- Integrated position-adjustable water rinse tank—configurable to align with specific cryostat height constraints or lab ergonomic requirements
- 13-slide capacity collection tank with tapered geometry to prevent slide stacking and ensure uniform drainage post-staining
- Color-coded slide holders matched to Thermo Scientific Cryochrome™ embedding media formulations—supporting traceable reagent-slide pairing and reducing mislabeling risk
- Semi-automatic operation mode: fully automated cycle execution with optional manual slide immersion override at any station—no auxiliary staining dishes required
- Cryostat-top mounting interface with vibration-dampened base plate and integrated slide feed alignment guides for seamless transfer from microtome to stainer
Sample Compatibility & Compliance
The Linistat is validated for use with standard 25 × 75 mm glass microscope slides bearing frozen-section tissue mounted in Thermo Scientific Cryochrome™ or equivalent low-viscosity OCT-compatible embedding media. It accommodates section thicknesses from 3 µm to 15 µm without mechanical interference. The system complies with ISO 13485:2016 requirements for in vitro diagnostic (IVD) ancillary equipment and meets electrical safety standards IEC 61010-1 for laboratory instrumentation. While not an IVD device itself, its operational parameters are documented per CLIA and CAP guidelines for histopathology pre-analytical processing. All wetted components—including reagent tanks, rinse channels, and slide carriers—are constructed from chemically resistant polypropylene and stainless steel, facilitating routine decontamination per CDC-recommended protocols.
Software & Data Management
The Linistat operates via embedded firmware with no external PC dependency. Cycle logs—including start time, station dwell durations, rinse tank position setting, and operator-initiated manual overrides—are timestamped and stored internally for up to 500 cycles. Audit trail functionality records user ID (via physical key switch activation), date/time stamps, and event type (e.g., “manual immersion at Station 3”), satisfying basic FDA 21 CFR Part 11 data integrity expectations for non-automated recordkeeping. Export is supported via USB port for CSV-formatted log retrieval; no cloud connectivity or remote access capability is implemented, preserving local data sovereignty in regulated environments.
Applications
Primary application focus includes intraoperative frozen section analysis, where turnaround time directly impacts surgical decision-making. The system enables standardized H&E staining for neuropathology, oncology margin assessment, and dermatopathology workflows requiring morphologic fidelity within tight temporal windows. It also supports quality assurance staining for cryostat performance verification, embedding medium lot testing, and training of histotechnologists in reproducible frozen-section handling. Due to its open reagent architecture, it may be adapted for alternative nuclear-counterstains (e.g., methyl green) or modified eosin formulations—subject to internal validation per laboratory-developed test (LDT) protocols.
FAQ
Can the Linistat be used for permanent section staining (e.g., paraffin-embedded tissues)?
No—the system is specifically engineered for frozen sections. Its dwell times, reagent volumes, and slide transport kinetics are optimized for aqueous-based staining of unfixed or lightly fixed cryosections; paraffin sections require deparaffinization and antigen retrieval steps incompatible with this platform.
Is the 20-second dwell time adjustable per station?
No—dwell time is fixed at 20 seconds per station to maintain regulatory consistency and minimize user-dependent variability; however, operators may pause or manually advance slides at any station during semi-auto mode.
Does the system support barcode scanning or LIS integration?
Not natively—the Linistat lacks built-in barcode readers or HL7/ASTM interface hardware; integration with laboratory information systems requires third-party middleware or manual log reconciliation.
What maintenance is required for long-term reliability?
Daily wipe-down of slide tracks and tank interiors; monthly inspection of carriage belt tension and rinse nozzle alignment; annual calibration verification using certified reference slides per Thermo Fisher Service Bulletin LIN-2023-04.