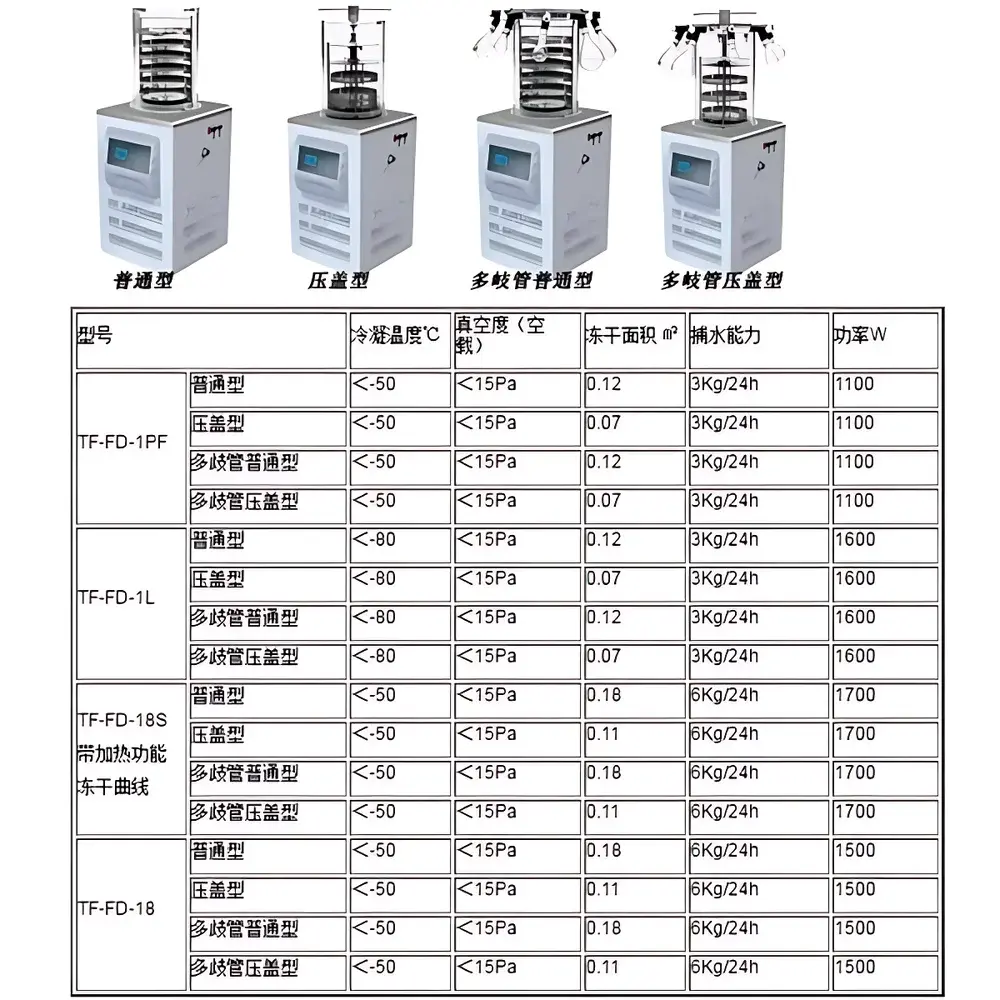
Tianfeng TF-FD-18S Stoppering Benchtop Freeze Dryer
| Brand | Tianfeng |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Direct Manufacturer |
| Instrument Type | Stoppering Freeze Dryer |
| Application Scope | Laboratory-Scale Lyophilization |
| Shelf Area | 0.11 m² |
| Ultimate Vacuum | <15 Pa (at no load) |
| Condenser Temperature | <−60 °C |
| Ice Capacity | 6 kg/24 h |
| Dimensions (W×D×H) | 550 mm × 550 mm × 960 mm |
| Power Supply | 220 V, 50 Hz |
| Rated Power | 1700 W |
| Shelf Configuration | 3-layer Φ220 mm stainless steel shelves with programmable electric heating |
| Control System | 7-inch color touchscreen with real-time lyo-cycle curve display (shelf temperature, chamber pressure, condenser temperature), PID control algorithm |
| Construction | 304 stainless steel condenser and chamber interior |
| Optional Features | Inert gas backfill valve (N₂ or Ar), co-lyophilization monitoring port, shelf temperature uniformity ≤ ±1 °C, optional eutectic point tester and automatic stopper press unit |
Overview
The Tianfeng TF-FD-18S Stoppering Benchtop Freeze Dryer is a laboratory-grade, in-situ lyophilization system engineered for reproducible, controlled freeze-drying of heat-sensitive biologicals, pharmaceuticals, and food-grade samples. It operates on the fundamental principle of sublimation—removing water from frozen material under deep vacuum while maintaining the sample below its eutectic temperature throughout primary drying. This preserves structural integrity, bioactivity, and reconstitution fidelity—critical for vaccines, monoclonal antibodies, enzymes, probiotics, and botanical extracts. Unlike conventional drying methods that induce thermal degradation, collapse, or oxidation, the TF-FD-18S enables drying at cryogenic temperatures (≤ −60 °C condenser), followed by precisely regulated shelf heating during secondary drying to remove bound moisture without exceeding 60 °C. Its integrated stoppering function allows for sterile, aseptic vial sealing post-drying—eliminating exposure to ambient conditions and supporting GLP-compliant workflow continuity.
Key Features
- In-situ pre-freezing and drying: Eliminates manual transfer between freezer and dryer, reducing contamination risk and process variability.
- Programmable shelf heating system: Three-layer stainless steel shelves (Φ220 mm each) with independent PID-controlled electric heating; supports user-defined temperature ramps and holds for optimized primary/secondary drying profiles.
- High-efficiency dual-stage refrigeration: Optimized cascade cooling architecture achieves stable condenser temperatures < −60 °C, ensuring reliable capture of both water vapor and low-volatility solvents (e.g., ethanol, acetone).
- Real-time process monitoring: 7-inch capacitive touchscreen displays concurrent curves for shelf temperature, chamber pressure, condenser temperature, and vacuum level—enabling immediate intervention and cycle validation.
- Robust 304 stainless steel construction: Corrosion-resistant condenser, chamber walls, and shelf supports ensure long-term durability and compliance with cleaning-in-place (CIP) protocols.
- Optical observation window: High-transparency acrylic door permits unobstructed visual monitoring of ice formation, sublimation front progression, and cake morphology throughout the entire cycle.
- Backfill capability: Integrated inert gas (N₂ or Ar) inlet valve enables controlled repressurization to prevent oxidation and support anaerobic drying conditions.
Sample Compatibility & Compliance
The TF-FD-18S accommodates standard serum vials (10–30 mL), Petri dishes, and custom trays up to Φ220 mm diameter across three stacked shelves. Its 0.11 m² effective drying area and 6 kg/24 h ice capacity support batch processing of up to 120 × 10-mL vials per cycle. The system meets foundational requirements for ISO 22000 (food safety), ISO 13485 (medical device quality management), and supports alignment with FDA 21 CFR Part 11 when paired with validated electronic record systems. While not supplied with factory-certified IQ/OQ documentation, its modular design, traceable component sourcing (including imported compressors), and stable thermal performance facilitate third-party qualification for GMP pilot-scale development and analytical reference standard preparation.
Software & Data Management
The embedded control firmware records all critical parameters at user-configurable intervals (1–60 s), exporting timestamped CSV files via USB interface. Cycle logs include shelf setpoint vs. actual temperature deviation, vacuum decay rate, condenser loading trends, and alarm event timestamps—supporting root-cause analysis and audit readiness. Optional data-logging modules provide 21 CFR Part 11–compliant features including user access controls, electronic signatures, and immutable audit trails. Raw data export enables integration with LIMS platforms or statistical process control (SPC) tools for trend analysis across multiple batches.
Applications
- Pharmaceutical R&D: Lyophilization of clinical trial formulations, stability study samples, and reference standards for HPLC/MS quantification.
- Vaccine & biologics development: Stabilization of live-attenuated viral vectors, recombinant proteins, and mRNA-LNP intermediates requiring strict cold-chain preservation.
- Food science: Dehydration of functional ingredients (e.g., lactic acid bacteria, anthocyanin-rich berry powders) while retaining antioxidant capacity and sensory attributes.
- Agricultural biotechnology: Preservation of mycorrhizal spores, rhizobial inoculants, and plant tissue culture explants for long-term germplasm banking.
- Academic research: Method development for novel cryoprotectant screening, freeze-concentration of dilute protein solutions, and mechanistic studies of ice nucleation kinetics.
FAQ
What is the maximum vial size supported on the TF-FD-18S shelves?
Standard configuration accommodates vials up to 30 mL in height with Φ22 mm diameter; taller formats require custom shelf spacing adjustment.
Does the system support automated stoppering without operator intervention?
Yes—the integrated pneumatic stopper press actuates automatically upon cycle completion, provided stoppers are pre-loaded and aligned within the chamber.
Can organic solvent-containing samples be processed safely?
The < −60 °C condenser provides sufficient cold trap margin for aqueous-organic mixtures with ≤30% vol/vol ethanol or acetone; higher concentrations require optional −80 °C condenser upgrade.
Is validation documentation included with shipment?
Factory calibration certificates for temperature and pressure sensors are provided; full IQ/OQ/PQ protocol development and execution are available as a separate service engagement.
What maintenance intervals are recommended for optimal performance?
Vacuum pump oil replacement every 500 operating hours; condenser defrosting after each 3–5 full cycles; annual inspection of O-rings, refrigerant charge, and electrical safety grounding.