Ugo Basile Model 38450 Electronic Von Frey Anesthesiometer for Rodent Mechanical Nociception Testing
| Brand | Ugo Basile |
|---|---|
| Origin | Italy |
| Model | 38450 |
| Maximum Force Range | 1000 g |
| Force Resolution | 0.1 g |
Overview
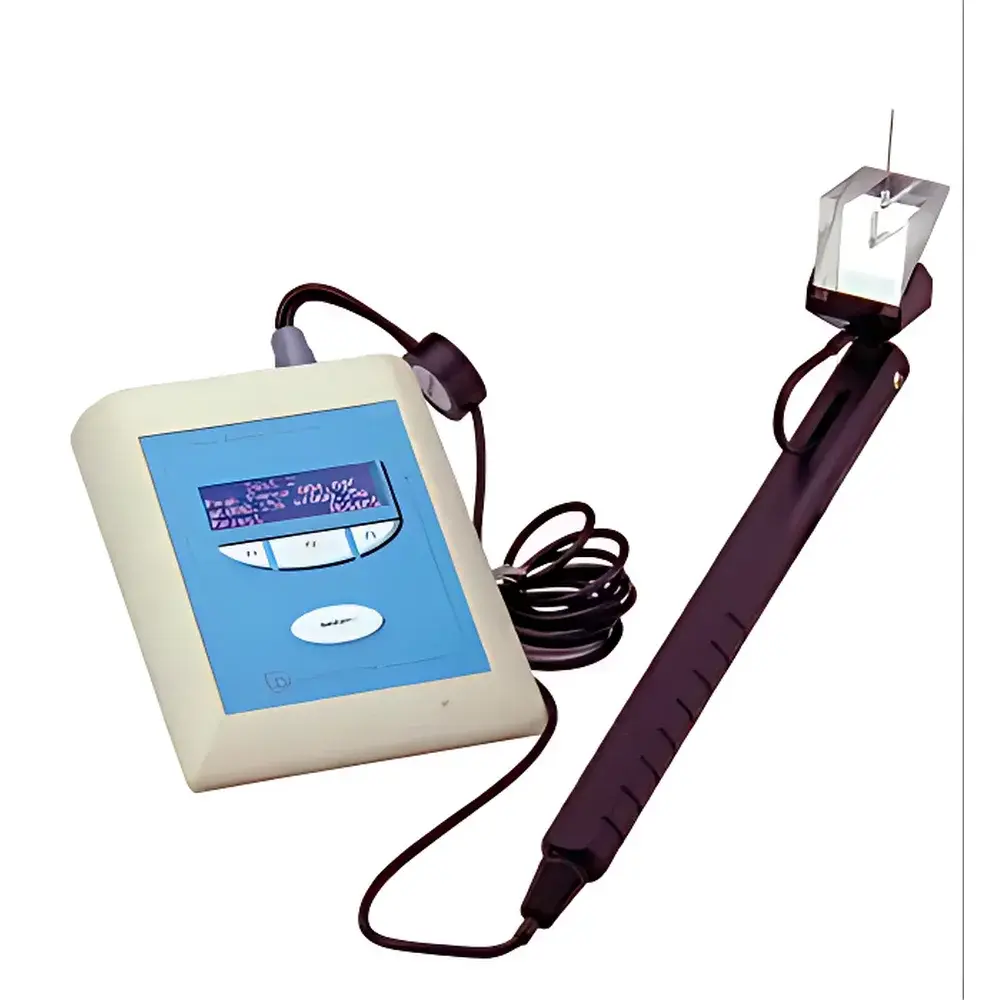
The Ugo Basile Model 38450 Electronic Von Frey Anesthesiometer is an ISO-certified, CE-marked instrument engineered for precise, objective quantification of mechanical nociceptive thresholds in rodents—primarily rats and mice. It operates on the principle of calibrated, linearly increasing mechanical force applied via a rigid stainless-steel probe to the plantar surface of the hind paw. Upon reaching the animal’s withdrawal threshold—the minimal force eliciting a rapid, unambiguous paw withdrawal reflex—the device automatically captures the peak force value (in grams) and reaction latency (in milliseconds). Unlike manual Semmes-Weinstein filaments (Von Frey hairs), which rely on discrete, logarithmically spaced fiber diameters and subjective endpoint interpretation, the Model 38450 delivers continuous, real-time force profiling with digital precision. Its design adheres to the core methodological requirements outlined in IASP guidelines for preclinical pain assessment and supports reproducible longitudinal studies under controlled environmental and behavioral conditions.
Key Features
- High-resolution force transduction system with ±0.1 g accuracy across the full 0–1000 g range, traceable to NIST-calibrated standards
- Prism-integrated probe handle enabling ergonomic, visually guided alignment with the target paw region—critical for minimizing inter-operator variability
- Digital peak-hold circuitry that captures and locks the maximum applied force at the moment of withdrawal, eliminating observer bias in endpoint determination
- Integrated footswitch interface allowing hands-free trial initiation and result validation; each trial is timestamped and tagged with operator ID (when configured)
- On-device memory storage for up to 1,000 trials with force-time waveform logging, including real-time display of applied force (red trace) versus user-defined ramp rate (blue reference line)
- Software-controlled ramp rate stabilization algorithm ensuring consistent force application velocity (e.g., 0.5–5.0 g/s), mitigating sensitization artifacts from variable stimulus kinetics
- Modular hardware architecture supporting optional bench-mounted stabilization brackets and species-specific restraint cages (rat/mouse)
Sample Compatibility & Compliance
The Model 38450 is validated for use with C57BL/6, Sprague-Dawley, Wistar, and CD-1 strains under standard IACUC- and AAALAC-accredited housing protocols. Its probe geometry and force range are optimized for adult rodents (20–300 g body weight), with published validation data confirming linear response across age-matched cohorts and both sexes. The instrument conforms to ISO 13485:2016 for medical device quality management systems and incorporates electromagnetic compatibility (EMC) per EN 61326-1. When operated with the optional GLP-compliant software module, it supports 21 CFR Part 11–aligned features—including electronic signatures, immutable audit trails, and role-based access control—making it suitable for regulatory submission packages in analgesic drug development programs.
Software & Data Management
The included Ugo Basile Nociception Suite (v4.2+) provides a secure, Windows-based platform for instrument control, real-time visualization, and statistical post-processing. Raw force-time curves are stored in binary format with metadata (date, operator, animal ID, cage number, ambient temperature/humidity). Export options include tab-delimited TXT and native Excel (.xlsx) files compatible with GraphPad Prism, SPSS, and R. Batch analysis tools enable automated threshold calculation using Dixon up-down or Chaplan logistic regression methods. Audit logs record all parameter changes, data deletions, and export events with timestamps and user credentials—fully compliant with GLP documentation standards. Data backups are encrypted and support network drive synchronization for centralized lab data governance.
Applications
This system is routinely deployed in academic and pharmaceutical research settings for: neuropathic pain modeling (e.g., CCI, SNI, STZ-induced diabetic neuropathy); inflammatory pain assessment (CFA, carrageenan); opioid tolerance and hyperalgesia studies; evaluation of novel TRP channel modulators, Nav1.7 inhibitors, or microglial suppressants; sex-difference analyses in pain sensitivity; and validation of genetic knockouts affecting somatosensory transduction. Over 1,200 peer-reviewed publications (including high-impact journals such as Gastroenterology, Brain, Behavior and Immunity, and Science Advances) cite the Model 38450 or its predecessor (Model 37450 Aesthesiometer), attesting to its methodological robustness and cross-laboratory reproducibility.
FAQ
Is the Model 38450 compatible with existing Ugo Basile accessories?
Yes—the probe tip geometry, calibration protocol, and software communication protocol are fully backward-compatible with the legacy Model 37450 Aesthesiometer, enabling direct comparative analysis across longitudinal studies.
Can the ramp rate be customized per experimental protocol?
Yes—users define ramp velocity (g/s) and hold duration via software; default presets align with IASP-recommended protocols for acute vs. chronic pain models.
Does the system require annual recalibration?
Ugo Basile recommends biannual verification using certified dead-weight standards; full recalibration by an authorized service center is advised every 24 months or after physical impact.
How is animal welfare addressed during testing?
The device enforces automatic force cutoff at 1000 g and includes visual/audible alerts for prolonged stimulation; all protocols must comply with local ethics committee requirements for maximum exposure duration and inter-trial recovery intervals.
Is cloud-based data hosting supported?
Local installation only; however, exported datasets can be ingested into institutional LIMS or cloud analytics platforms via API-ready CSV/Excel formats.