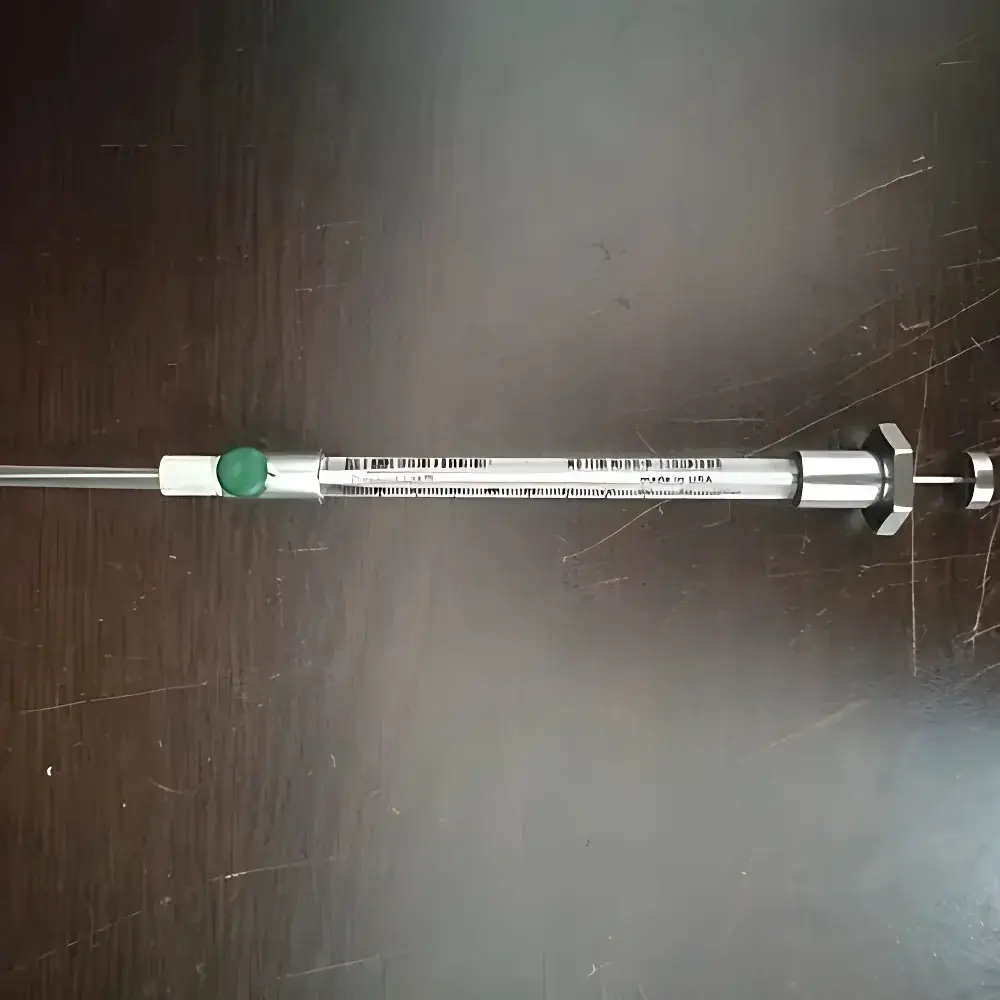
VICI PS-050025 A-2 Series Gas-Tight Syringe with Integrated Push-Button Valve
| Brand | VICI |
|---|---|
| Model | PS-050025 |
| Volume | 100 µL |
| Needle | RN (Removable Needle), Bevel Tip, 0.029" OD × 0.005" ID × 2" L |
| Maximum Pressure Rating | 250 psi |
| Seal Material | PTFE Plunger Tip & Needle Seal |
| Barrel Material | Glass |
| Valve Type | Integrated Push-Button Actuated On/Off Valve |
| Compliance | Designed for GLP/GMP-compliant chromatographic workflows requiring trace-level sample integrity |
Overview
The VICI PS-050025 is a precision-engineered A-2 series gas-tight syringe featuring an integrated push-button actuated valve, specifically designed for quantitative, loss-free handling of volatile, low-boiling-point, and air-sensitive liquid samples in high-performance liquid chromatography (HPLC), ultra-high-performance liquid chromatography (UHPLC), and gas chromatography (GC) sample introduction systems. Unlike standard syringes, the PS-050025 employs a dual-seal architecture—comprising a PTFE plunger tip and a PTFE needle seal—combined with an optically ground glass barrel and ultra-smooth bore finish to eliminate adsorption, leakage, and vapor-phase sample loss during aspiration, storage, and injection. Its valve mechanism enables true “zero-dead-volume” actuation: the fluid path remains fully isolated until the user depresses the button, at which point the internal flow channel opens instantaneously—ensuring reproducible, operator-independent injection timing and volume delivery. This design conforms to fundamental requirements of ISO 17025-accredited laboratories where sample integrity, repeatability, and auditability are critical.
Key Features
- Integrated push-button on/off valve enabling precise, repeatable, and hands-free control over fluid path isolation—eliminating manual needle disconnection and minimizing exposure to ambient atmosphere
- Gas-tight performance rated up to 250 psi (17.2 bar), validated via helium leak testing per ASTM F2391, ensuring reliable containment of pressurized or volatile analytes
- 100 µL nominal capacity with RN (removable needle) configuration; beveled stainless-steel needle (0.029″ OD × 0.005″ ID × 2″) optimized for minimal wall contact and reduced carryover in autosampler interfaces
- PTFE-plunger tip and PTFE needle seal provide chemical resistance across broad solvent compatibility (including acetonitrile, methanol, THF, chloroform, and dichloromethane) while maintaining low-friction movement and long-term dimensional stability
- Optically polished borosilicate glass barrel with sub-micron surface roughness (<0.2 µm Ra) minimizes sample adhesion and facilitates complete rinsing—critical for trace-level analysis and multi-analyte methods
- Backstop mechanism prevents plunger extrusion under pressure for volumes ≥250 µL; though this unit is 100 µL, its structural reinforcement ensures consistent stroke linearity and force response across repeated cycles
Sample Compatibility & Compliance
The PS-050025 is routinely deployed in regulated environments where sample representativeness directly impacts method validity. It supports direct coupling to autosamplers from Agilent, Waters, Thermo Fisher, and Shimadzu via standardized mounting adapters (e.g., 10-32 UNF thread interface). Its construction meets material compatibility guidelines outlined in USP for pharmaceutical excipient testing and complies with ICH Q2(R2) expectations for injection system robustness in method validation. The valve’s mechanical actuation eliminates reliance on electronic signals—making it suitable for use in explosion-proof zones (ATEX Zone 1/2) and electrostatic-sensitive environments. Documentation packages—including Certificate of Conformance, materials traceability (ASTM E2918), and pressure test reports—are available upon request to support FDA 21 CFR Part 11–aligned instrument qualification (IQ/OQ/PQ).
Software & Data Management
While the PS-050025 operates as a passive, hardware-level component, its integration into automated workflows is enabled through vendor-specific autosampler firmware protocols (e.g., Agilent OpenLab CDS, Waters Empower, Thermo Chromeleon). The valve’s binary open/closed state allows synchronization with injection trigger events via TTL or dry-contact input—enabling precise temporal alignment between valve actuation and detector acquisition windows. Audit trails generated by the host software capture timestamped injection events, including valve activation status, which satisfies ALCOA+ data integrity principles (Attributable, Legible, Contemporaneous, Original, Accurate). For laboratories implementing electronic lab notebooks (ELN) or LIMS, metadata fields such as syringe lot number, calibration date, and pressure rating can be mapped to sample records via configurable import templates.
Applications
- Quantitative analysis of volatile organic compounds (VOCs) in environmental water matrices using purge-and-trap GC–MS
- Stabilized delivery of thermally labile standards (e.g., aldehydes, ketones, lactones) in UHPLC–HRMS metabolomics workflows
- Calibration standard preparation for residual solvent testing (ICH Q3C) where sub-µL accuracy and zero evaporation loss are mandatory
- On-line derivatization injections requiring sequential reagent/sample introduction without cross-contamination
- Method transfer between HPLC platforms where syringe-to-syringe variability must remain below ±0.5% RSD (n=6) for retention time and peak area
FAQ
What is the maximum recommended operating pressure for the PS-050025?
The syringe is rated for continuous operation up to 250 psi (17.2 bar) at 25 °C. Exceeding this limit may compromise seal integrity and invalidate calibration traceability.
Can this syringe be sterilized using autoclaving?
No. Autoclaving is not recommended due to thermal expansion mismatch between glass and PTFE components, which may induce microfractures or seal deformation. Ethanol wipe-down or isopropanol rinse followed by nitrogen purging is the validated cleaning protocol.
Is the RN needle compatible with all VICI and third-party autosampler needle holders?
Yes—the RN interface conforms to VICI’s universal 10-32 UNF threading standard and is mechanically interchangeable with Hamilton 1700-series and SGE 800-series holders when used with appropriate adapter sleeves.
How often should the PTFE seals be replaced under routine use?
Under typical laboratory conditions (≤100 injections/day, aqueous/organic solvents), PTFE plunger tips and needle seals maintain functional integrity for ≥6 months. Visual inspection for scoring or discoloration is advised prior to each calibration cycle.
Does the valve mechanism require periodic lubrication or maintenance?
No. The valve is a dry-acting, spring-loaded polymer mechanism with no serviceable parts. No lubricants are specified or permitted—introduction of oils or greases may impair sealing performance and violate USP analytical instrument qualification criteria.