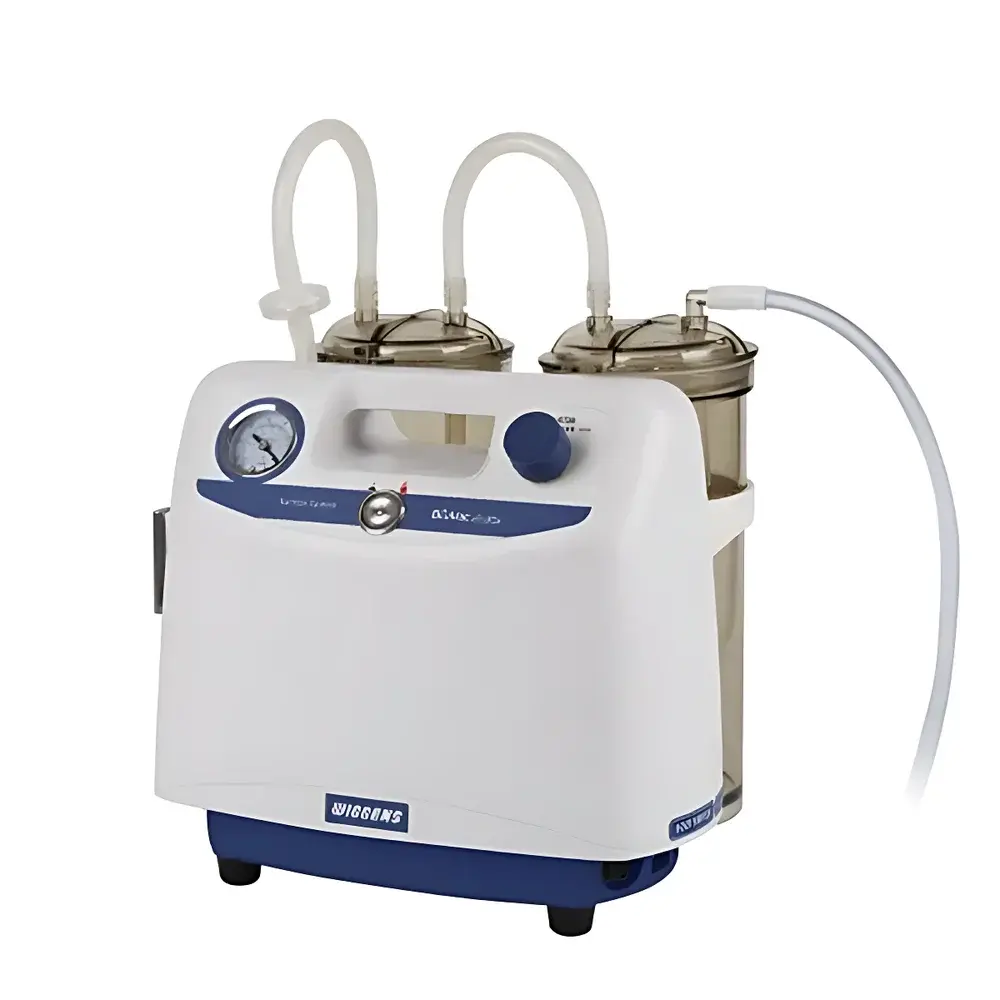
WIGGENS BioVac 235 Portable Liquid Aspiration System
| Brand | WIGGENS |
|---|---|
| Origin | Germany |
| Model | BioVac 235 |
| Vacuum Level | ≥100 mbar |
| Flow Rate | ≤35 L/min |
| Weight | 6.9 kg |
| Dimensions (W×D×H) | 27 × 30 × 28 cm |
| Waste Bottle Capacity | 1500 mL × 2 |
| Filtration | 0.2 µm PTFE disc filter |
| Sterilization | Autoclavable (121 °C, 20 min) |
| Foot Switch | Included |
| Accessories | 4-piece aspiration kit |
| Compliance | CE, ISO 13485-design aligned |
Overview
The WIGGENS BioVac 235 Portable Liquid Aspiration System is an engineered solution for controlled, aseptic liquid removal in life science laboratories. Designed specifically for post-culture processing—including spent media aspiration from cell culture flasks, T-plates, and bioreactor vessels—the system operates on a dry, oil-free vacuum principle. Unlike traditional oil-lubricated pumps, the BioVac 235 utilizes a maintenance-free diaphragm vacuum pump that eliminates risk of oil contamination, ensures consistent vacuum stability over time, and delivers low operational noise (<52 dB(A))—critical in laminar flow hoods and biosafety cabinets where acoustic disturbance and cross-contamination must be minimized. Its compact footprint and integrated handle support true portability, enabling seamless movement between biosafety level 1 (BSL-1) and BSL-2 workstations without compromising vacuum integrity or sterility assurance.
Key Features
- Oil-free diaphragm pump: Eliminates need for lubricant replenishment, reduces long-term maintenance overhead, and prevents hydrocarbon vapor ingress into sensitive biological workflows.
- Dual-stage overflow protection: Combines a buoyancy-activated float valve inside the autoclavable polycarbonate waste bottle with a sterile-grade 0.2 µm hydrophobic PTFE disc filter positioned inline between bottle and pump—preventing liquid ingress into the pump head under overfill or sudden pressure fluctuation.
- Autoclavable components: Both 1500 mL waste bottles and the 0.2 µm PTFE filter discs are validated for repeated sterilization at 121 °C for 20 minutes (EN 285-compliant cycles), supporting GLP/GMP-aligned reuse protocols.
- Ergonomic aspiration control: Includes a hands-free foot switch with momentary-action logic and a four-piece aspiration kit (sterile tubing, reusable suction tips, and modular connectors) optimized for variable vessel geometries—from 6-well plates to 1000 mL culture bottles.
- Compact, benchtop-integrated design: Weighing only 6.9 kg with dimensions of 27 × 30 × 28 cm, the unit fits beneath standard fume hood sashes and integrates directly into ISO Class 5 clean environments without requiring dedicated utility hookups.
Sample Compatibility & Compliance
The BioVac 235 is compatible with aqueous-based biological fluids including cell culture supernatants, PBS washes, enzymatic digestion mixtures, and low-viscosity transfection reagents. It is not intended for use with organic solvents, highly corrosive acids/bases, or viscous suspensions (>500 cP). All wetted materials—including tubing, bottle gaskets, and filter housings—comply with USP Class VI biocompatibility standards. The system’s electrical architecture conforms to IEC 61010-1 for laboratory equipment safety, and its construction aligns with ISO 13485 principles for medical device–adjacent instrumentation. While not a medical device itself, its design supports workflows referenced in ISO 56002 (innovation management) and ASTM E2659 (standard practice for nonclinical laboratory study conduct).
Software & Data Management
The BioVac 235 operates as a standalone analog-controlled system with no embedded firmware or digital interface. This intentional design choice prioritizes reliability, electromagnetic compatibility (EMC), and regulatory simplicity—eliminating validation burdens associated with software-driven devices under FDA 21 CFR Part 11 or EU Annex 11. Vacuum output remains stable across ambient temperature ranges of 15–30 °C and relative humidity up to 80% non-condensing. For laboratories requiring audit trails, manual log entries (date, operator ID, usage duration, bottle change timestamp) are recommended per internal SOPs—consistent with ALCOA+ data integrity principles.
Applications
- Aseptic removal of spent growth media during adherent and suspension cell passaging
- Decanting supernatants after centrifugation or sedimentation steps in primary tissue isolation
- Draining wash buffers in ELISA plate preparation and immunostaining workflows
- Controlled aspiration of conditioned media for exosome or cytokine harvesting
- Low-risk liquid transfer in teaching labs and QC environments where reproducibility and contamination control are prioritized over high-throughput automation
FAQ
Is the BioVac 235 suitable for use inside a biosafety cabinet?
Yes—the oil-free pump generates minimal heat and particulate emission, and its noise profile (<52 dB(A)) meets ISO 15707 recommendations for BSC-integrated equipment.
Can the PTFE filter be reused after autoclaving?
Yes—each 0.2 µm hydrophobic PTFE disc filter is rated for ≥20 autoclave cycles at 121 °C/20 min; visual inspection for discoloration or warping is advised prior to reuse.
What is the maximum recommended viscosity for safe aspiration?
For optimal pump longevity and flow consistency, liquids exceeding 500 cP (e.g., methylcellulose solutions or collagen gels) should be pre-diluted or processed using gravity-assisted methods.
Does the system include vacuum regulation capability?
No—BioVac 235 delivers fixed vacuum output (≥100 mbar); users requiring adjustable vacuum levels should consider the BioVac 335 series with integrated electronic pressure control.
Are replacement parts traceable and documented for quality audits?
Yes—WIGGENS provides batch-specific CoA documentation for all consumables (filters, bottles, tubing kits), with full traceability back to manufacturing lot and material certifications per EN ISO 13485 Annex A.