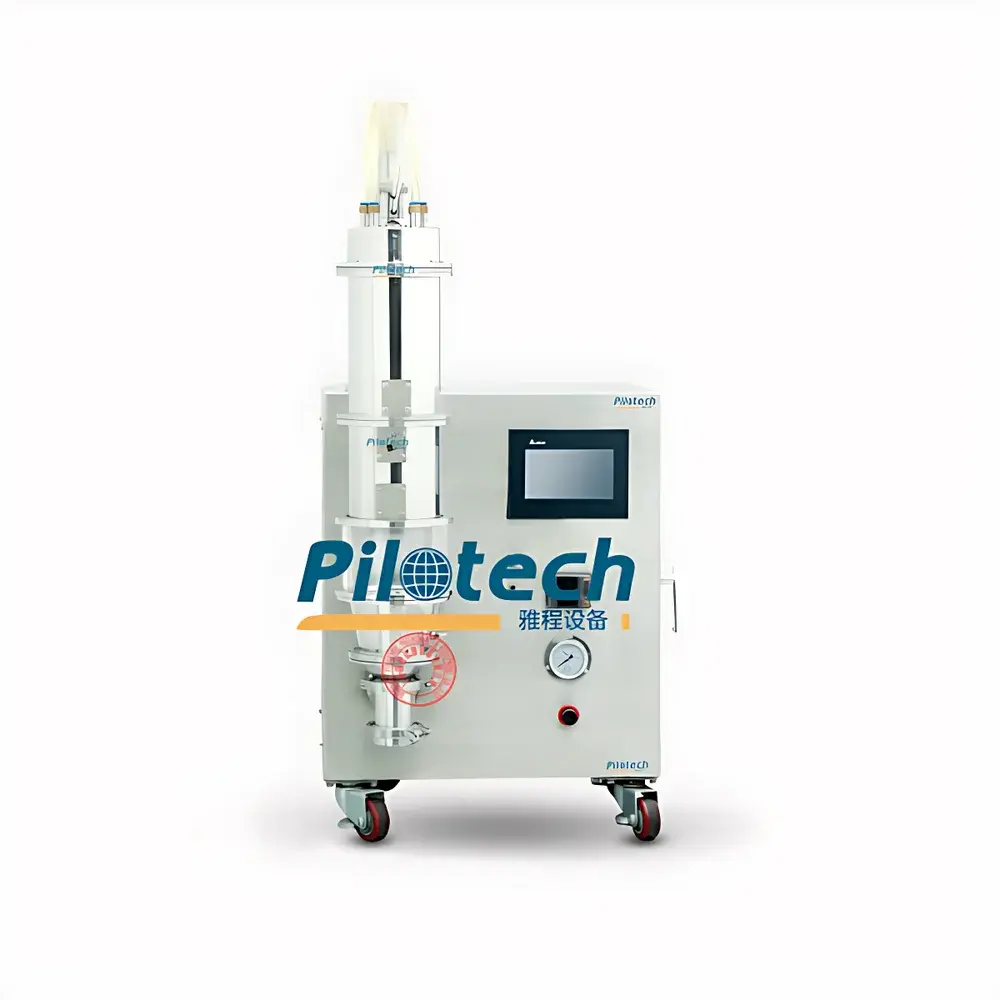
Yacheng YC-310 Laboratory-Scale Fluidized Bed Granulator, Coater & Dryer
| Brand | Yacheng / PILOTECH |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | OEM Manufacturer |
| Model | YC-310 |
| Pricing | Upon Request |
Overview
The Yacheng YC-310 is a compact, integrated laboratory-scale fluidized bed processor engineered for granulation, film coating, and controlled drying of pharmaceutical and nutraceutical powders. It operates on the principle of fluidization—where upward-flowing heated air suspends particulate material in a dynamic, turbulent state—enabling uniform heat and mass transfer during spray-based unit operations. Unlike batch tray or oven drying, the YC-310 leverages fluid dynamics to achieve rapid, thermodynamically balanced moisture removal while preserving particle integrity and morphology. Its dual-nozzle configuration (top-spray and bottom-spray) supports both agglomeration-based granule formation and precision film coating of microspheres or pellets. The system is designed for R&D-scale process development aligned with ICH Q5A–Q5E and FDA Process Validation Guidance, supporting early-stage formulation screening, scale-up studies, and GMP-compliant method development.
Key Features
- Modular fluidized bed architecture with interchangeable conical product containers (standard 310 mL working volume, scalable via optional vessels)
- Dual-mode spray capability: top-spray for granulation/drying and bottom-spray for functional enteric or sustained-release coating of pellets ≥100 µm
- PLC-based control system with color touchscreen HMI, real-time parameter adjustment (inlet air temperature, blower frequency, peristaltic pump speed, pulse reverse-jet cleaning interval)
- Integrated PID-controlled temperature regulation with inlet-air ↔ product-bed thermal feedback loop to prevent surface case-hardening and ensure consistent drying kinetics
- Optimized airflow design featuring dual-inlet wind channels beneath the draft tube (bottom-spray mode) and expansion chamber geometry (top-spray mode) to maintain stable fountain-type fluidization
- High-efficiency reverse-jet cleaning system minimizing filter clogging and improving granule yield by >15% compared to conventional fixed-pulse designs
- Compliance-ready data handling: USB export for raw parameter logs (CSV), optional audit trail functionality for 21 CFR Part 11–aligned environments
Sample Compatibility & Compliance
The YC-310 accommodates a broad range of hygroscopic and thermolabile materials, including herbal extracts, amorphous APIs, lactose-based excipients, and polymer dispersions (e.g., Eudragit® L100, HPMC AS). Particle size input range spans from submicron aerosols to coarse milled powders (D90 ≤ 500 µm); final granule output typically falls within 100 µm – 3 mm, depending on binder viscosity, atomization pressure, and residence time. The system meets mechanical safety requirements per ISO 12100 and electrical compliance per IEC 61000-6-2/6-4. For regulated labs, it supports GLP documentation workflows and can be qualified per ASTM E2500–22 (User Requirement Specification → Design Qualification → Operational Qualification).
Software & Data Management
The embedded control software records time-stamped operational parameters at 1-second intervals—including inlet/outlet air temperature, relative humidity, bed pressure drop, spray rate, and blower RPM—enabling full traceability of each run. Data export is native to CSV format via USB 2.0 interface; no proprietary drivers required. Optional firmware upgrade enables electronic signature capture, user-level access control (admin/operator/reviewer roles), and automatic generation of IQ/OQ protocol templates compliant with Annex 11 and EU GMP Annex 15. All logged events—including parameter overrides, alarm triggers, and cleaning cycles—are timestamped and immutable post-recording.
Applications
- Development of immediate-release granules from aqueous or alcoholic herbal extracts
- Functional coating of mini-tablets and extruded pellets for pH-dependent release (e.g., gastric protection of probiotics)
- Moisture-sensitive API stabilization via low-temperature fluidized bed drying (residual moisture < 2.0% w/w achievable)
- Process mapping studies for QbD implementation: DoE-driven optimization of inlet temperature vs. binder concentration vs. spray rate
- Bridging studies between lab-scale (YC-310) and pilot-scale (e.g., Glatt GPCG-1) fluid bed systems
- Accelerated stability testing of coated dosage forms under controlled humidity/temperature cycling
FAQ
What is the maximum batch capacity for top-spray granulation?
The standard conical bowl supports up to 310 mL of loose powder (typically 80–120 g, depending on bulk density). Optional larger bowls (600 mL, 1 L) are available for extended scalability studies.
Can the YC-310 perform solvent-based coating without explosion risk?
Yes—when equipped with nitrogen inerting option (N₂ purge + O₂ sensor interlock), it safely handles ethanol-, acetone-, or IPA-based coating suspensions per ATEX Zone 22 guidelines.
Is remote monitoring supported?
Standard Ethernet port enables Modbus TCP integration with SCADA or MES platforms; optional Wi-Fi module allows secure cloud-based run supervision via TLS 1.2 encrypted channel.
How is cleaning validation addressed?
The unit features fully drainable fluidization chamber, tool-free nozzle removal, and CIP-compatible surfaces (Ra ≤ 0.8 µm stainless steel 316L); residue sampling ports align with PDA TR72 recommendations.
Does the system comply with FDA 21 CFR Part 11?
Out-of-the-box, it meets ALCOA+ principles; full Part 11 compliance requires activation of optional audit trail, electronic signature, and role-based access modules—validated per vendor-supplied IQ/OQ protocols.