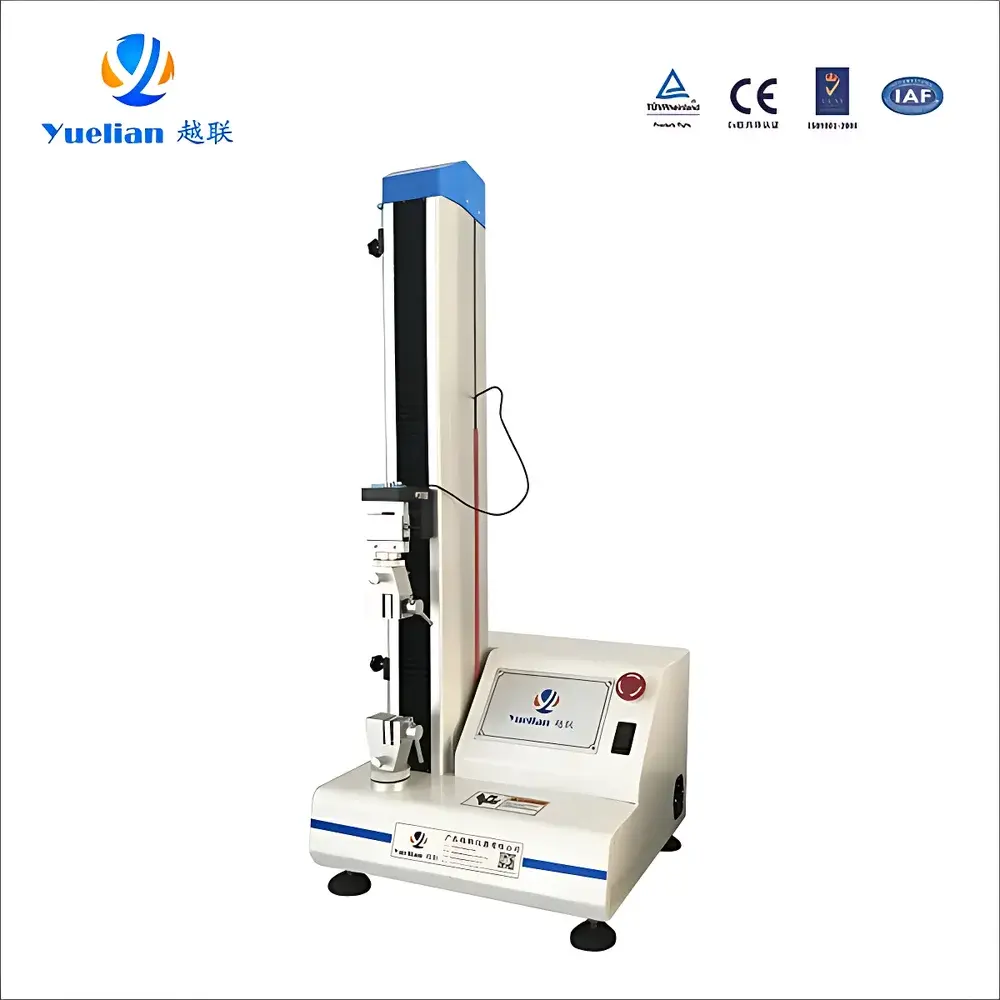
Yuelian YL-8801AB Pharmacopoeial Initial Adhesion Tester (Rolling Ball Method)
| Brand | Yuelian |
|---|---|
| Model | YL-8801AB |
| Measurement Principle | Rolling Ball Inclined Plane Method (ChP 2020 Vol. IV, General Chapter 0952 Method I) |
| Compliance Standards | ChP 0952, GB/T 2792, ASTM D3330, GB/T 12914, GB/T 13022 |
| Force Capacity Options | 10 N, 20 N, 50 N, 100 N, 200 N, 500 N, 1 kN |
| Force Resolution | 1/500,000 full scale |
| Force Accuracy | ≤ ±0.5% FS |
| Displacement Resolution | 0.005 mm |
| Test Speed Range | 5–500 mm/min (programmable) |
| Max Travel | 500 mm |
| Test Width | 55 mm |
| Drive System | Stepper Motor + Precision Ball Screw |
| Power Supply | 220 V, 50/60 Hz, 10 A |
| Dimensions (W×D×H) | 400 × 300 × 700 mm |
| Weight | ~28 kg |
| Safety Protections | Dual limit switches (upper/lower travel), overload cutoff, emergency stop, automatic halt on specimen failure, programmable force/travel limits |
Overview
The Yuelian YL-8801AB Pharmacopoeial Initial Adhesion Tester is an ISO/GB-compliant instrument engineered for precise determination of initial tack—the instantaneous adhesive resistance developed upon light contact between a pressure-sensitive adhesive (PSA) surface and human skin or standardized substrate. It implements the rolling ball inclined plane method as defined in the Chinese Pharmacopoeia (ChP) Volume IV, General Chapter 0952 Method I, and aligns with parallel international standards including ASTM D3330 and GB/T 2792. Unlike static peel or shear-based adhesion assessments, initial tack quantifies the microscale interfacial energy and viscoelastic response at momentary contact under minimal applied pressure—critical for evaluating transdermal patches, medical tapes, wound dressings, and topical drug delivery systems during early-stage formulation screening and QC release testing.
Key Features
- Pharmacopoeia-validated mechanical design: Dedicated inclined plane assembly with precision angle adjustment (±0.1°) and calibrated stainless-steel ball set (0.5–20 mm diameter, AISI 440C, Ra ≤ 0.02 µm surface finish) per ChP 0952 requirements.
- Integrated electromechanical test platform: High-resolution stepper motor drive coupled with ground ball screw transmission ensures repeatable positioning accuracy (±0.005 mm) and stable velocity control across the full 5–500 mm/min range.
- Dual-capacity load cell architecture: Optional force transducers (10 N to 1 kN) with ≤ ±0.5% full-scale accuracy and 1/500,000 internal resolution enable traceable measurement of both low-tack films and high-adhesion hydrogel matrices.
- Comprehensive safety interlocks: Hardware-based upper/lower travel limits, real-time overload monitoring, emergency stop circuitry, and automatic halt upon specimen rupture or displacement threshold breach—fully compliant with IEC 61000-6-2 EMC and GB/T 29803 functional safety guidelines.
- Modular fixture interface: Standardized T-slot base accommodates interchangeable test plates (stainless steel, aluminum, or synthetic skin mimics), ball release guides, and alignment jigs for rapid method reconfiguration between initial tack, peel, and tensile modes.
Sample Compatibility & Compliance
The YL-8801AB supports standardized testing of pharmaceutical and biomedical adhesive products including transdermal patches, surgical tapes, silicone-based wound contact layers, acrylic PSAs, and laminated composite films. Substrate compatibility extends to stainless steel (GB/T 2792), polyethylene terephthalate (PET), glass, and ISO 10993-5-certified synthetic skin surrogates. All operational parameters—including inclination angle (30° ± 0.5°), ball release height (100 mm ± 1 mm), and ambient conditioning (23 ± 2°C, 50 ± 5% RH per ISO 291)—are programmatically enforced via firmware logic to ensure audit-ready reproducibility. The system meets GLP documentation requirements through built-in electronic signatures, time-stamped test logs, and 21 CFR Part 11–compliant user access controls when paired with validated software modules.
Software & Data Management
Control and analysis are executed via a Windows-based PC application featuring a dual-mode interface: manual mode for direct parameter adjustment and method mode for pre-configured pharmacopoeial protocols (e.g., “ChP 0952 Initial Tack – Ball Series B”). Raw force/displacement data are acquired at ≥1 kHz sampling rate and stored in encrypted MS-ACCESS databases with mandatory metadata tagging (operator ID, lot number, environmental conditions, calibration certificate ID). Export options include CSV, PDF reports with embedded chromatographic-style force–time curves, and XML files compatible with LIMS integration. Audit trail functionality records all parameter changes, test initiations, and result modifications with immutable timestamps—supporting FDA and NMPA regulatory submissions.
Applications
This instrument serves critical roles across R&D, quality assurance, and regulatory compliance laboratories. It enables comparative ranking of PSA formulations during polymer selection; batch-to-batch consistency verification for GMP-manufactured patches; stability-indicating assessment of adhesive degradation under accelerated aging (ICH Q1A); and root-cause analysis of delamination failures in multi-layer drug-eluting devices. Validated use cases include evaluation of temperature-sensitive adhesives (e.g., thermoreversible hydrogels), moisture-activated bioadhesives, and nanostructured microneedle patch backing layers where conventional peel tests lack sensitivity to surface-energy-driven initial bonding dynamics.
FAQ
What pharmacopoeial standards does the YL-8801AB directly support?
It implements ChP Volume IV General Chapter 0952 Method I (rolling ball), ASTM D3330 Type A, and GB/T 2792 with hardware and software configurations certified for method equivalence.
Can the same unit perform peel strength and holding power tests?
No—the YL-8801AB is purpose-built for initial tack per ChP 0952 Method I. For holding power (Method II) and 180° peel (Method III), Yuelian offers dedicated instruments: YL-8807 (vertical shear) and YS-90 (peel tester), respectively.
Is calibration traceable to national metrology institutes?
Yes—load cells are supplied with NIST-traceable calibration certificates (CNAS-accredited lab), and inclinometer accuracy is verified using optical autocollimator reference standards.
What sample dimensions are required for valid initial tack measurement?
Specimens must be ≥25 mm wide × 100 mm long, mounted on rigid backing plate with ≥50 mm free length for ball roll path, per ChP 0952 Section 3.1.
Does the system support automated pass/fail judgment based on ball retention criteria?
Yes—the software compares observed stopping ball number against predefined specification limits (e.g., “Ball #7 ±1” per internal SOP) and flags deviations in real time with configurable alarm thresholds.