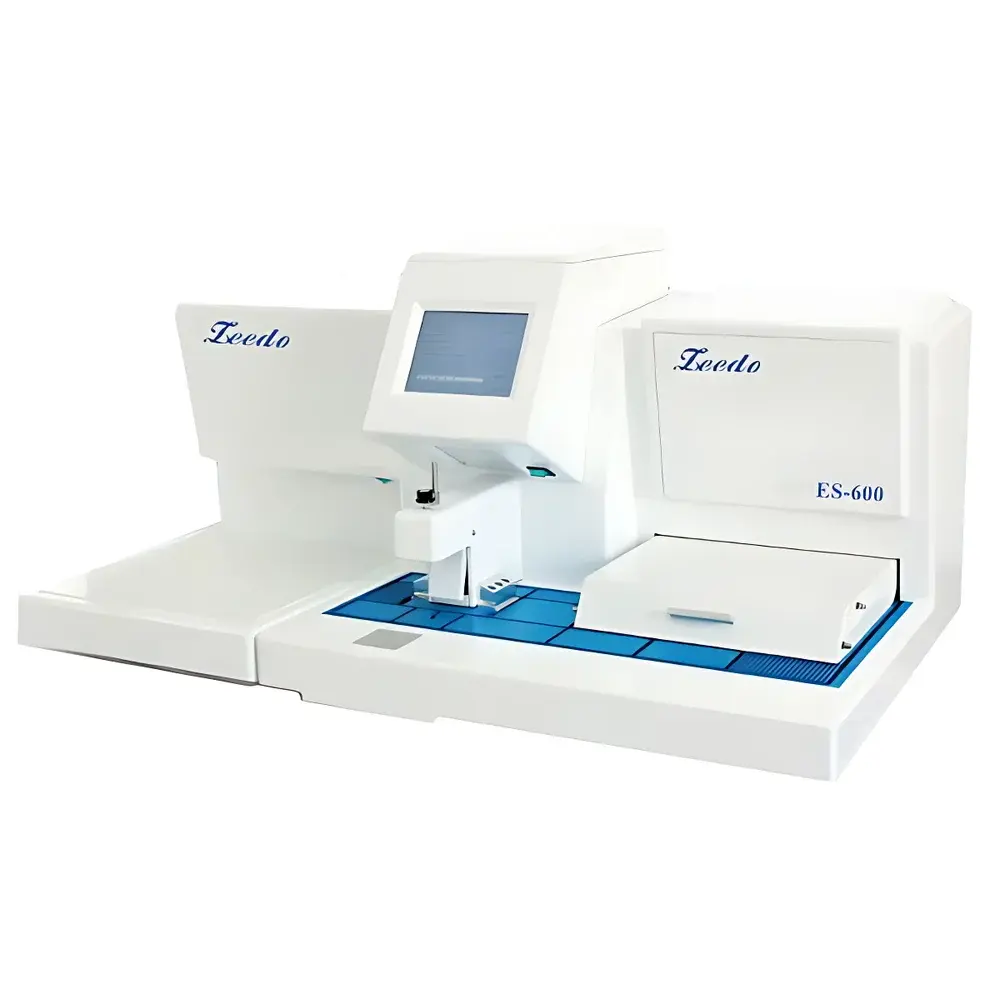
ZEEDO ES-600 Automated Paraffin Embedding and Cryo-Embedding Station
| Brand | ZEEDO (Huasu) |
|---|---|
| Origin | Zhejiang, China |
| Model | ES-600 |
| Wax Reservoir Capacity | 6.5 L |
| Cold Plate Temperature Range | −20 °C to ambient |
| Working Surface Area | 830 cm² (287 mm × 205 mm + 330 mm × 73 mm) |
| Embedded Mold Heater Volume | 0.93 L |
| Cold Plate Dimensions | 365 mm × 330 mm |
| Net Weight | 40 kg (main unit), 25 kg (cold plate) |
| Power Supply | 220 V, 50 Hz |
| Total Rated Power | 800 W (main unit) + 100 VA (cold plate) |
Overview
The ZEEDO ES-600 is a fully automated, dual-function paraffin embedding and cryo-embedding station engineered for high-throughput histopathology laboratories in hospitals, academic research institutions, and contract diagnostic service providers. It integrates precision thermal control, ergonomic workflow design, and fail-safe operational architecture to support reproducible tissue orientation and embedding—critical prerequisites for consistent microtome sectioning and downstream immunohistochemical or molecular analysis. Unlike conventional single-mode embedding units, the ES-600 features synchronized operation between its heated paraffin handling system and an independently controlled cryogenic plate, enabling seamless transition from warm wax infiltration to rapid tissue solidification without manual transfer. Its core thermal architecture employs five isolated PID-controlled zones—including wax reservoir, fluid delivery lines, working surface, upper and lower tissue storage trays—each adjustable within 30–75 °C with ±0.5 °C stability. This multi-zone granularity ensures optimal wax viscosity management across diverse tissue types (e.g., fatty, fibrous, or calcified specimens) while minimizing thermal stress-induced artifact formation.
Key Features
- 6.5-liter gravity-fed paraffin reservoir with dual-stage filtration and redundant overheat protection—designed to sustain uninterrupted embedding of up to 500 blocks per fill cycle.
- Integrated cold plate (365 mm × 330 mm) using Peltier-based thermoelectric cooling, achieving stable temperatures from ambient down to −20 °C with <2 min cooldown time to −10 °C.
- Modular 830 cm² work surface incorporating recessed wax channels, built-in wax flow regulator, mechanical trimming tool, LED top illumination (5,000 K color temperature), and optional magnifier mount for precise tissue positioning.
- Large-capacity tissue storage tray (300 mm × 385 mm × 55 mm; 6.25 L) accommodating ≥222 standard cassette slots—or two full-size dehydration baskets from common automated tissue processors—eliminating intermediate cassette transfer steps.
- Touchscreen HMI with intuitive icon-driven interface supporting programmable start/stop schedules, real-time temperature logging, and embedded fault diagnostics.
- Dual activation modes: responsive microswitch hand controls and hands-free foot pedal—enabling operator flexibility during prolonged embedding sessions.
- Separate heated embedding mold tray (290 mm × 160 mm × 20 mm; 0.93 L capacity) maintains uniform wax temperature independent of main reservoir dynamics.
Sample Compatibility & Compliance
The ES-600 accommodates all standard histological cassettes (e.g., Sakura, Leica, Thermo Fisher), including those used in automated tissue processors compliant with ISO 15189 and CLIA-certified workflows. Its temperature zoning architecture supports embedding protocols aligned with CAP (College of American Pathologists) and UK NEQAS guidelines for tissue processing consistency. The device’s electrical safety conforms to IEC 61010-1:2010 for laboratory equipment, while its thermal containment design prevents wax migration into low-voltage control circuits—reducing risk of thermal degradation-related failures during GLP/GMP audits. Although not FDA 510(k)-cleared as a standalone medical device, the ES-600 is routinely deployed in ISO 13485-certified pathology labs where embedding quality directly impacts diagnostic accuracy per WHO/IARC tissue reporting standards.
Software & Data Management
The embedded firmware records timestamped temperature profiles for each of the five thermal zones at 10-second intervals, storing up to 30 days of operational history locally. Data export is supported via USB 2.0 port in CSV format for integration into LIMS platforms or QC dashboards. While the system does not include network connectivity or remote access capability, its audit trail meets minimum requirements for 21 CFR Part 11-compliant environments when paired with validated third-party data archiving solutions. All user-initiated parameter changes—including setpoint adjustments, schedule modifications, or calibration offsets—are logged with operator ID (if enabled via external authentication gateway) and immutable timestamps.
Applications
- Routine surgical pathology embedding in high-volume hospital labs requiring >200 blocks/day throughput.
- Research-grade embedding of delicate tissues (e.g., neural, embryonic, or biopsy fragments) where precise orientation and minimal thermal distortion are essential.
- Multi-step embedding protocols involving sequential wax infiltration, intermediate chilling, and final orientation—particularly valuable for lymph node or sentinel node mapping studies.
- Training environments in university pathology departments, where modular controls and visual feedback support competency-based learning objectives.
- Contract histology services performing batch embedding under CAP-accredited quality systems.
FAQ
Does the ES-600 support ASTM E2917-compliant thermal validation?
Yes—the five independently controllable zones allow for sensor placement per ASTM E2917 Annex A2, enabling mapping of thermal uniformity across the wax reservoir, delivery path, and working surface.
Can the cold plate be operated simultaneously with the paraffin system?
Yes—both subsystems operate autonomously; no interlock prevents concurrent use, facilitating hybrid embedding workflows.
Is the touchscreen interface compatible with glove use in BSL-2 environments?
The capacitive touch panel responds reliably through standard nitrile or latex gloves; no stylus required.
What maintenance intervals are recommended for the dual-stage wax filter?
Filter replacement is advised every 200 operating hours or after processing 1,000 tissue blocks—whichever occurs first—to prevent flow restriction and wax degradation.
Does the ES-600 include CE marking for EU market deployment?
The unit carries CE marking under the Low Voltage Directive (2014/35/EU) and EMC Directive (2014/30/EU); full conformity documentation is available upon request from authorized ZEEDO distributors.