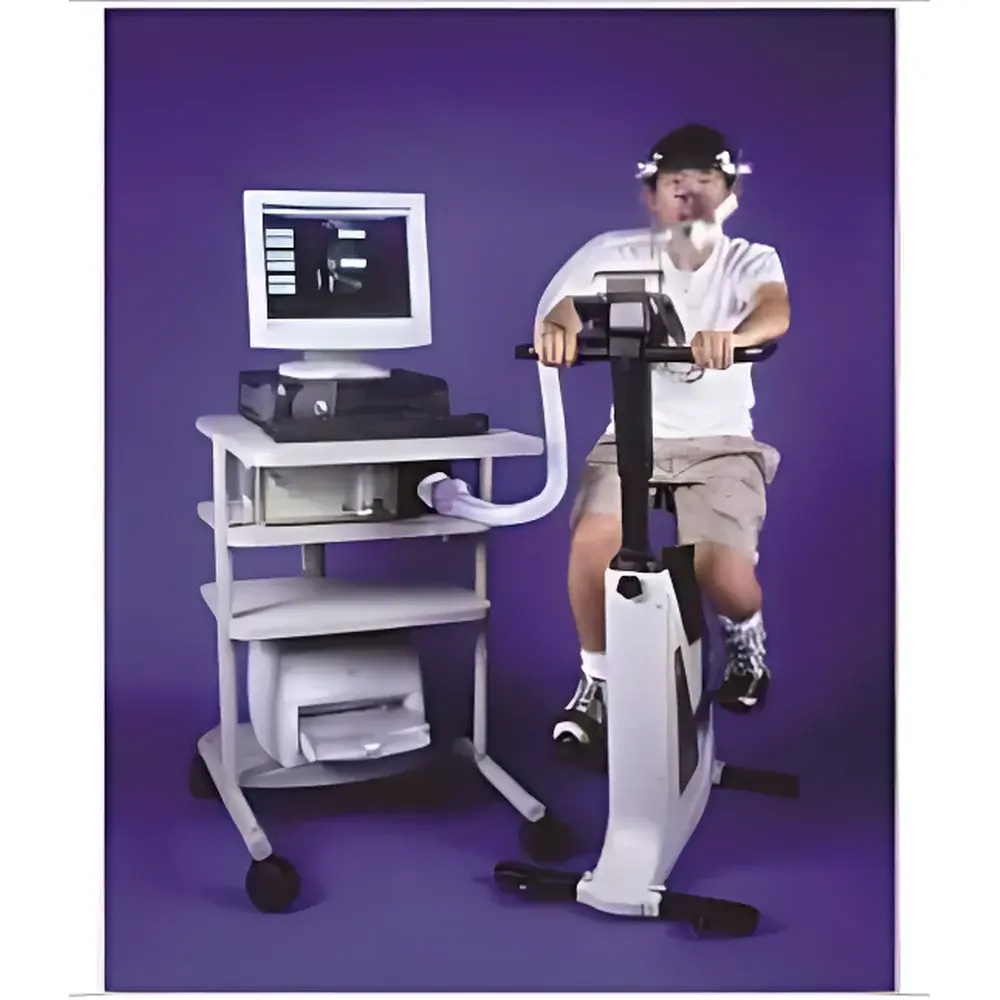
Parvo Medics True One2400 Metabolic Measurement System
| Brand | Parvo Medics |
|---|---|
| Origin | USA |
| Model | True One2400 |
| O₂ Analysis Range | 0–100% or 0–25% |
| O₂ Accuracy | ±0.02% |
| O₂ Response Time | 200 ms (21% → 16% in 4 L mixing chamber) |
| CO₂ Analysis Method | NDIR (Non-Dispersive Infrared) |
| CO₂ Range | 0–15% |
| CO₂ Accuracy | ±0.03% (for 0–10% CO₂) |
| CO₂ Response Time | 100 ms |
| Flow Measurement Accuracy | ±0.5% |
| Flow Range | 0–800 L/min |
| Calibration | Dual-stage (3 L syringe, 16% O₂ / 4% CO₂ gas mixture) |
| Operating Temperature | 14–30 °C |
| Relative Humidity | 20–80% RH |
| Warm-up Time | 30 min |
| Power Supply | 100–240 VAC, 50–60 Hz |
| Software Platform | Windows XP–compatible proprietary metabolic analysis suite |
| Data Export | Native Excel (.xls) output |
Overview
The Parvo Medics True One2400 Metabolic Measurement System is a fully integrated, research-grade indirect calorimetry platform engineered for high-fidelity assessment of pulmonary gas exchange and substrate oxidation kinetics. It employs dual-sensor physiology—paramagnetic O₂ detection and non-dispersive infrared (NDIR) CO₂ analysis—combined with a precision laminar-flow turbine sensor calibrated via the peer-reviewed Yeh algorithm. This architecture ensures traceable, reproducible measurements of VO₂ (oxygen consumption), VCO₂ (carbon dioxide production), VE (minute ventilation), RER (respiratory exchange ratio), and energy expenditure under dynamic physiological conditions. Designed for clinical exercise laboratories, sports science centers, and preclinical translational research facilities, the system complies with American Thoracic Society (ATS)/European Respiratory Society (ERS) guidelines for cardiopulmonary exercise testing (CPET) and adheres to ISO 9001-certified manufacturing protocols. Each unit undergoes pre-shipment validation using the Douglas bag method—a gold-standard reference technique for volumetric gas collection—and is certified per JIS T 9121:2001 Annex B for computerized gas exchange measurement accuracy.
Key Features
- Paramagnetic O₂ analyzer with dual-range capability (0–100% or 0–25%), ±0.02% full-scale accuracy, and 200 ms response time (21% → 16% step change in 4 L mixing chamber)
- NDIR CO₂ sensor covering 0–15% range with ±0.03% accuracy (0–10% CO₂) and 100 ms response time
- Laminar-flow turbine transducer calibrated to ±0.5% across 0–800 L/min, implementing the Yeh algorithm for real-time flow compensation during variable breathing patterns
- Automated dual-stage gas calibration: primary (3 L precision syringe) and secondary (certified 16% O₂ / 4% CO₂ gas mixture)
- Integrated hardware interface support for ECG, treadmill, cycle ergometer, power meters, and bidirectional Rudolph valves
- Modular physical design: compact analyzer module (12″ W × 10″ D × 5″ H), mixing chamber (5″ W × 12.5″ D × 5″ H), and mobile cart (24″ W × 30″ D × 35″ H) optimized for lab workflow efficiency
Sample Compatibility & Compliance
The True One2400 supports human subjects across diverse populations—including elite athletes, pediatric cohorts, geriatric patients, and individuals with chronic cardiopulmonary disease—when used with appropriate mouthpieces, nose clips, bacterial/viral filters, and moisture traps. Its validated performance envelope extends to altitudes up to 3,000 m above sea level without recalibration. The system meets ASTM E2573-22 requirements for respiratory gas analyzers used in clinical exercise testing and satisfies data integrity prerequisites outlined in FDA 21 CFR Part 11 when operated with audit-trail-enabled software configurations. All firmware and calibration records are timestamped and user-identifiable to support GLP/GCP-compliant study documentation.
Software & Data Management
The proprietary Windows XP–based metabolic analysis software provides real-time visualization of O₂, CO₂, and flow waveforms with configurable time bases and scaling. Users define custom display layouts, overlay multiple parameters, and generate breath-by-breath summaries or time-averaged reports (e.g., 15-, 30-, or 60-second epochs). The software computes derived metrics—including resting metabolic rate (RMR), exercise energy cost, fat/carbohydrate oxidation rates (via stoichiometric equations), and ventilatory thresholds (V-slope and ventilatory equivalents)—using algorithms published in the Journal of Applied Physiology and validated against whole-room calorimetry. Raw and processed data export natively to Microsoft Excel (.xls) format; metadata (subject ID, test protocol, calibration logs) embed automatically within exported files for traceability.
Applications
- Clinical CPET for diagnosis and functional staging of heart failure, COPD, pulmonary hypertension, and metabolic myopathies
- Sports performance assessment: VO₂max determination, lactate threshold estimation, and training zone optimization
- Nutritional science: longitudinal monitoring of energy balance, dietary intervention efficacy, and adaptive thermogenesis
- Rehabilitation medicine: objective quantification of functional capacity before/after cardiac or pulmonary rehabilitation programs
- Academic research: mechanistic studies of mitochondrial respiration, hypoxia adaptation, and pharmacological modulation of substrate utilization
FAQ
What regulatory standards does the True One2400 comply with?
The system conforms to JIS T 9121:2001 Annex B for computerized gas exchange measurement, ASTM E2573-22 for respiratory gas analyzers, and ISO 9001 quality management requirements. When configured with audit-trail functionality, it supports FDA 21 CFR Part 11 compliance.
Is altitude compensation required for field deployments?
No. The paramagnetic O₂ sensor and NDIR CO₂ detector maintain specified accuracy up to 3,000 m elevation without manual barometric correction or recalibration.
How is flow calibration validated prior to shipment?
Each unit undergoes end-to-end verification using the Douglas bag method—a gravimetrically traceable reference standard—as documented in the Certificate of Conformance included with delivery.
Can the system interface with third-party exercise equipment?
Yes. Standard analog/digital I/O ports support synchronization with commercially available treadmills, cycle ergometers, ECG systems, and metabolic carts from major OEMs via TTL, RS-232, or USB protocols.
What level of technical support is provided post-installation?
Parvo Medics delivers one full day of on-site installation, hardware verification, software configuration, and operator training conducted by factory-certified application specialists. Remote diagnostics and firmware updates are available through secure customer portal access.