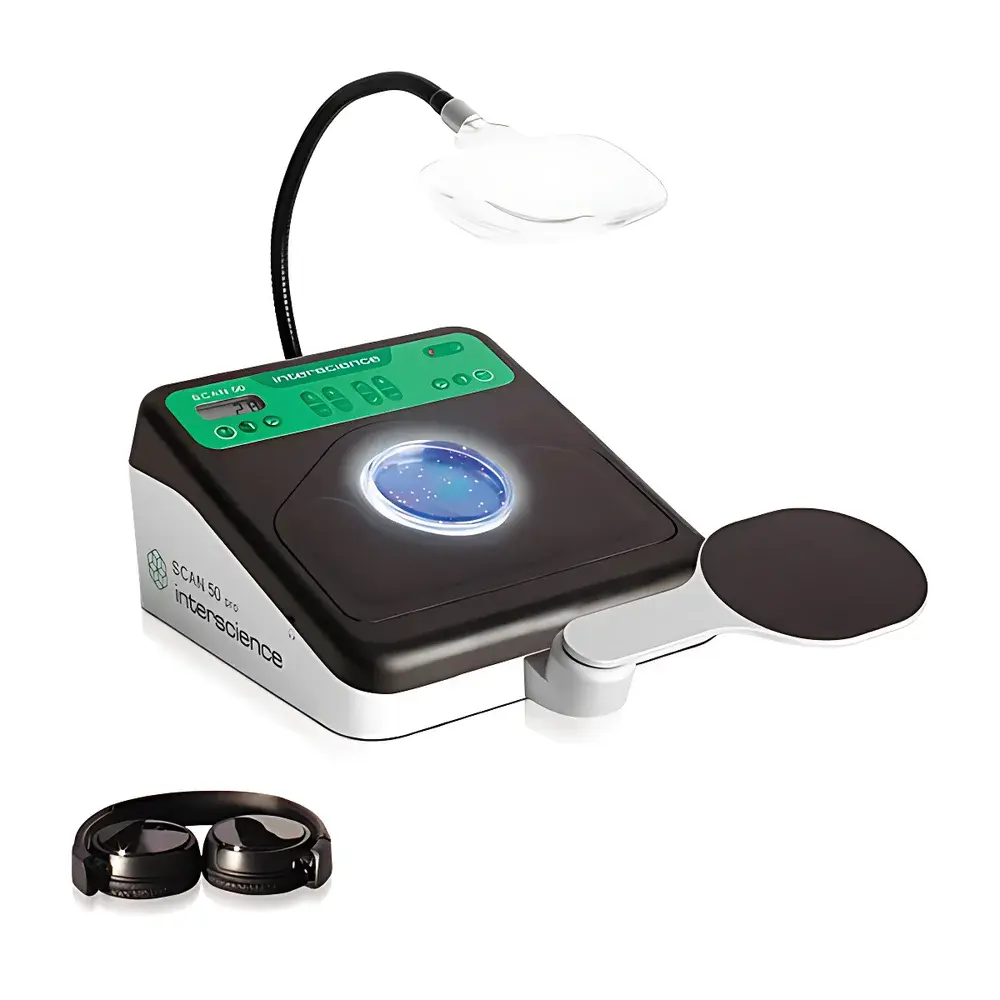
Interscience Scan 50 Pro Manual Colony Counter with Ergonomic HandPad
| Brand | Interscience |
|---|---|
| Origin | France |
| Model | Scan 50 Pro |
| Power Supply | 100–240 V~, 50–60 Hz |
| Max. Power | 20 W |
| IP Rating | IP20 |
| Petri Dish Diameter | 55–90 mm |
| Counting Range | 0–1999 CFU (manual) |
| Illumination | 2nd-generation dark-field indirect LED lighting, 5 intensity levels |
| Touch Surface | Adjustable sensitivity capacitive surface |
| Audio Feedback | 9 selectable tones, 5 volume levels, headphone jack included |
| Connectivity | USB interface for LIMS/SIL integration, barcode reader support |
| Data Export | CSV (Excel™-compatible), plain text (Word™-compatible) |
| Compliance | ISO 7218, FDA Bacteriological Analytical Manual (BAM) |
| Warranty | 3 years (upon registration) |
| Spare Parts Availability | 10 years |
| Accessories | Optional magnifier, detachable glass plate, removable HandPad扶手 |
Overview
The Interscience Scan 50 Pro Manual Colony Counter is an ergonomically engineered microbiological counting workstation designed for high-accuracy, low-fatigue manual colony enumeration in regulated and research laboratory environments. It operates on the principle of visual contrast enhancement—leveraging a proprietary 2nd-generation dark-field indirect illumination system (Dark Field 2) to maximize colony visibility across the entire surface of standard Petri dishes (55–90 mm diameter), including edge regions, agar depth interfaces, and opaque or chromogenic media. Unlike conventional transmitted or reflected light systems, Dark Field 2 uses precisely angled LED arrays and optical reflectors to diffuse light laterally beneath the dish, eliminating glare, hotspots, and shadow artifacts. This ensures uniform contrast between microbial colonies and background medium—critical for accurate detection of small, translucent, or pigmented colonies on chromogenic agars (e.g., CHROMagar™, Oxoid Brilliance™), as well as on rapid-dry formats including Petrifilm™, Compact Dry™, MC-Media Pads™, EasyPlate™, and membrane filters. The device supports all standard plating methods: pour plate, spread plate, spiral plating, and loop dilution assays—enabling full-surface enumeration (100% dish coverage) without repositioning.
Key Features
- Ergonomic HandPad: A fully adjustable, slide-mounted forearm support that positions seamlessly on either left or right side of the unit; reduces upper-limb musculoskeletal strain during prolonged counting sessions and accommodates both right- and left-handed operators.
- Capacitive Touch Surface: High-sensitivity, pressure-independent counting interface with tactile pulse feedback synchronized to user-selectable audio cues (9 tones, 5 volume levels) and visual LED indicators—minimizing cognitive load and inter-operator variability.
- Dark Field 2 Illumination System: Patented indirect LED lighting with five discrete intensity levels; provides consistent, shadow-free illumination across heterogeneous agar surfaces while protecting operator ocular comfort through absence of direct beam exposure.
- Modular Hygiene Design: Removable tempered glass counting surface and detachable HandPad enable rapid disinfection using ethanol, hydrogen peroxide vapor, or quaternary ammonium compounds—fully compliant with GLP/GMP cleanroom maintenance protocols.
- Integrated Connectivity: Native USB 2.0 port enables seamless bidirectional data exchange with Laboratory Information Management Systems (LIMS) and Scientific Instrumentation Linkage (SIL) platforms; supports real-time sample ID capture via external barcode scanners.
Sample Compatibility & Compliance
The Scan 50 Pro is validated for use with all common solid microbiological culture formats conforming to ISO 7218:2018 (“Microbiology of the food chain — General requirements and guidance for microbiological examinations”) and the U.S. FDA Bacteriological Analytical Manual (BAM), Chapter 3 (“Aerobic Plate Count”). It accommodates chromogenic and selective media (e.g., RAPID’E. coli 2, Salmonella ID, Candida ID), hydrated film-based systems (Petrifilm™, Compact Dry™), and filtration membranes (0.45 µm nitrocellulose, mixed ester). Its full-dish scanning capability ensures compliance with AOAC Official Method 990.12 and ISO 4833-1:2013 for total viable count determination. The device’s mechanical and electrical architecture meets IEC 61010-1 safety standards for laboratory equipment, and its IP20 rating confirms suitability for dry, non-hazardous indoor laboratory environments.
Software & Data Management
No proprietary software installation is required—the Scan 50 Pro functions as a HID-compliant USB peripheral. Count data—including colony count value, timestamp, operator ID (via optional barcode input), and sample identifier—are exported directly as UTF-8 encoded CSV files compatible with Microsoft Excel™ (Windows/macOS), LibreOffice Calc, and LIMS ingestion modules. Text-based export (plain .txt) is also supported for legacy system integration. All exported records include immutable metadata fields essential for audit readiness: device serial number, firmware version, illumination setting used, and touch-event timestamps. While the instrument itself does not implement 21 CFR Part 11 electronic signature controls, its deterministic output format and traceable hardware ID facilitate alignment with FDA-regulated workflows when deployed within validated LIMS ecosystems supporting audit trails and user access logs.
Applications
- Quality control laboratories performing routine aerobic plate counts (APC), coliform screening, and pathogen confirmation per ISO, AOAC, and USP guidelines.
- Contract testing labs requiring ISO/IEC 17025-compliant documentation pathways for accredited microbiological analyses.
- Academic and industrial R&D facilities conducting method validation studies, media performance testing, and antimicrobial efficacy assessments (e.g., ISO 22196, ASTM E2149).
- Hospital and public health labs performing environmental monitoring (EM) on settle plates and contact plates under EU GMP Annex 1 and USP .
- Fermentation and bioprocess development teams quantifying viable cell density in starter cultures, probiotics, and lyophilized preparations.
FAQ
Does the Scan 50 Pro support automated image capture or AI-based colony recognition?
No. The Scan 50 Pro is a manual counting platform with enhanced visual ergonomics and tactile feedback. It does not incorporate camera modules, image analysis algorithms, or machine learning capabilities.
Can the HandPad be permanently fixed to one side?
No—the HandPad is intentionally designed as a freely sliding, tool-free attachment; it locks into position magnetically on either side but remains fully repositionable to suit workflow layout or operator preference.
Is firmware upgradable in the field?
Yes. Firmware updates are delivered via Interscience’s secure customer portal and installed using the USB interface and provided update utility—ensuring continued compatibility with evolving LIMS protocols and regulatory expectations.
What calibration or verification procedures are recommended?
While no routine calibration is required (as it is a manual counting aid, not a measuring instrument), users should perform daily verification using NIST-traceable colony-count reference plates or standardized grid test slides to confirm tactile responsiveness, illumination uniformity, and audio/visual feedback fidelity.
Is the device suitable for use in biosafety cabinets?
Yes—its low power consumption (≤20 W), absence of internal fans or moving parts, and compact footprint allow safe operation inside Class II A2 and B2 biosafety cabinets, provided adequate USB cable routing and ventilation clearance are maintained.