YUYANBIO e-Celsius® Human Core Body Temperature Telemetry System
| Brand | YUYANBIO |
|---|---|
| Origin | USA |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Product Category | Imported Medical Device |
| Model | e-Celsius® Human Core Temperature Telemetry System |
| Pricing | Available Upon Request |
Overview
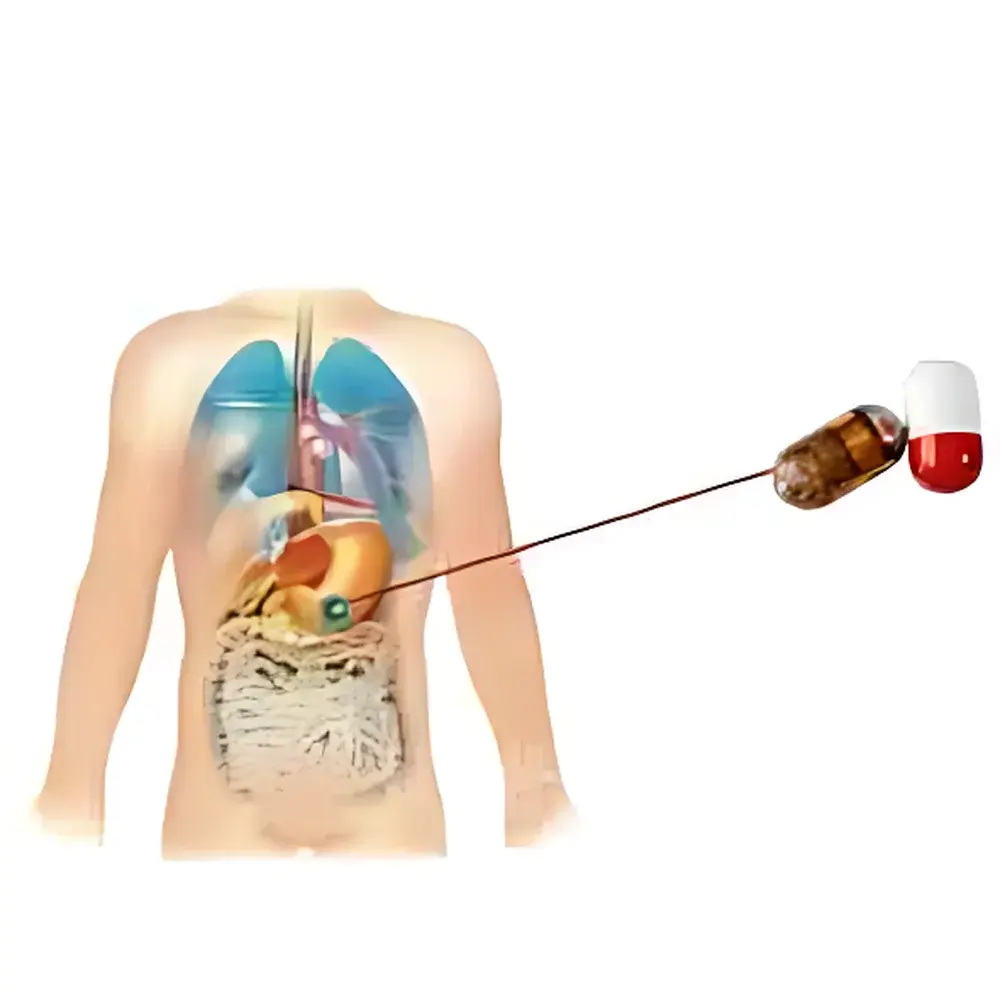
The YUYANBIO e-Celsius® Human Core Body Temperature Telemetry System is an ingestible, wireless physiological monitoring platform engineered for continuous, non-invasive measurement of core body temperature in human subjects under free-living or controlled experimental conditions. Based on validated ingestible sensor capsule technology, the system operates on the principle of thermistor-based temperature transduction within a biocompatible, hermetically sealed capsule that traverses the gastrointestinal (GI) tract via peristalsis. As the capsule moves from ingestion through gastric passage, small intestinal transit, and colonic elimination, it acquires high-fidelity thermal data at programmable sampling intervals. This approach eliminates external sensor artifacts associated with skin-surface probes, rectal thermistors, or tympanic measurements—enabling true core temperature profiling aligned with circadian physiology, metabolic demand, and thermoregulatory stress responses.
Key Features
- Ingestible capsule design (17.7 × 8.9 mm, 1.7 g) compliant with ISO 10993-5 biocompatibility standards for short-term internal use
- High-resolution thermal sensing: ±0.02 °C accuracy over 25–45 °C range, with user-configurable sampling frequency up to 30 Hz
- Onboard memory capacity: 2,000 temperature readings stored internally; data retention stable for ≥1 year prior to activation, ≥20 days post-activation
- Low-power RF telemetry: 433 MHz ISM-band transmission with reliable reception within ≤1 m line-of-sight distance
- Multi-capsule support: Single receiver unit concurrently processes data from up to three capsules in real time
- Scalable deployment: Up to seven receivers can be daisy-chained via USB or Ethernet for synchronized multi-subject studies
- Receiver storage: 80,000 data points per unit (equivalent to >9 hours of continuous 30-Hz recording from one capsule)
Sample Compatibility & Compliance
The e-Celsius® system supports both human clinical research and preclinical animal studies, with model variants certified for use in rats, mice, and beagles. Human-use capsules are manufactured under ISO 13485-certified quality management systems and comply with FDA guidance for ingestible electronic devices (FDA Guidance for Industry: Ingestible Devices – General Considerations, 2021). The system meets essential requirements of IEC 60601-1 (medical electrical equipment safety) and IEC 62304 (software lifecycle processes), and its data acquisition workflow supports audit trails compatible with GLP and GCP environments. All firmware and software components adhere to FDA 21 CFR Part 11 requirements for electronic records and signatures when deployed with validated configurations.
Software & Data Management
Data acquisition and visualization are managed via the e-Celsius® Desktop Suite, a cross-platform application (Windows/macOS) supporting real-time waveform display, timestamped event annotation, and export in CSV, MATLAB (.mat), and HDF5 formats. The software includes built-in tools for circadian rhythm analysis (e.g., cosinor modeling of temperature minima/maxima), thermal drift correction, and synchronization with external physiological inputs (e.g., ECG, accelerometry, respirometry). All data files include embedded metadata (capsule ID, calibration coefficients, sampling parameters), ensuring traceability across longitudinal studies. Raw binary logs are digitally signed and encrypted during transfer to prevent tampering—critical for regulatory submissions and peer-reviewed publication workflows.
Applications
- Exercise physiology: Continuous core temperature monitoring during endurance sports (cycling, marathon running, triathlon), team sports (rugby, American football), and aquatic disciplines (diving, open-water swimming)
- Thermoregulatory assessment: Evaluation of heat acclimatization protocols, cold exposure response, and pharmacological modulation of thermogenesis
- Circadian biology: Quantification of core temperature nadir/phase shifts in shift-work, jet lag, and sleep disorder studies
- Clinical thermometry: Early detection of febrile response in immunocompromised cohorts, perioperative temperature tracking, and sepsis surveillance
- Occupational health: Monitoring thermal strain in firefighters, military personnel, and industrial workers operating in extreme ambient conditions
- Preclinical research: Comparative thermoregulation studies across rodent and canine models under pharmacologic or genetic intervention
FAQ
How long does the capsule remain functional inside the human GI tract?
Typical transit duration ranges from 24 to 48 hours, depending on individual motility patterns; complete excretion occurs without intervention.
Is the capsule reusable?
No—each capsule is single-use, sterilized, and intended for one-time ingestion and disposal per regulatory clearance.
Can the system integrate with third-party data acquisition platforms?
Yes, the e-Celsius® SDK provides TCP/IP and serial API access for integration with LabVIEW, Python (via PySerial), and MATLAB Simulink environments.
What environmental conditions affect signal reliability?
Signal integrity remains stable across ambient temperatures of −10 °C to +50 °C and relative humidity up to 95% RH (non-condensing); metal shielding or dense tissue mass may attenuate RF transmission at the 1-m limit.
Does the system require IRB or ethics committee approval for human use?
Yes—human ingestion studies must undergo institutional review board (IRB) evaluation per local regulatory frameworks (e.g., FDA IDE, EU MDR Annex XV, or equivalent national guidelines).