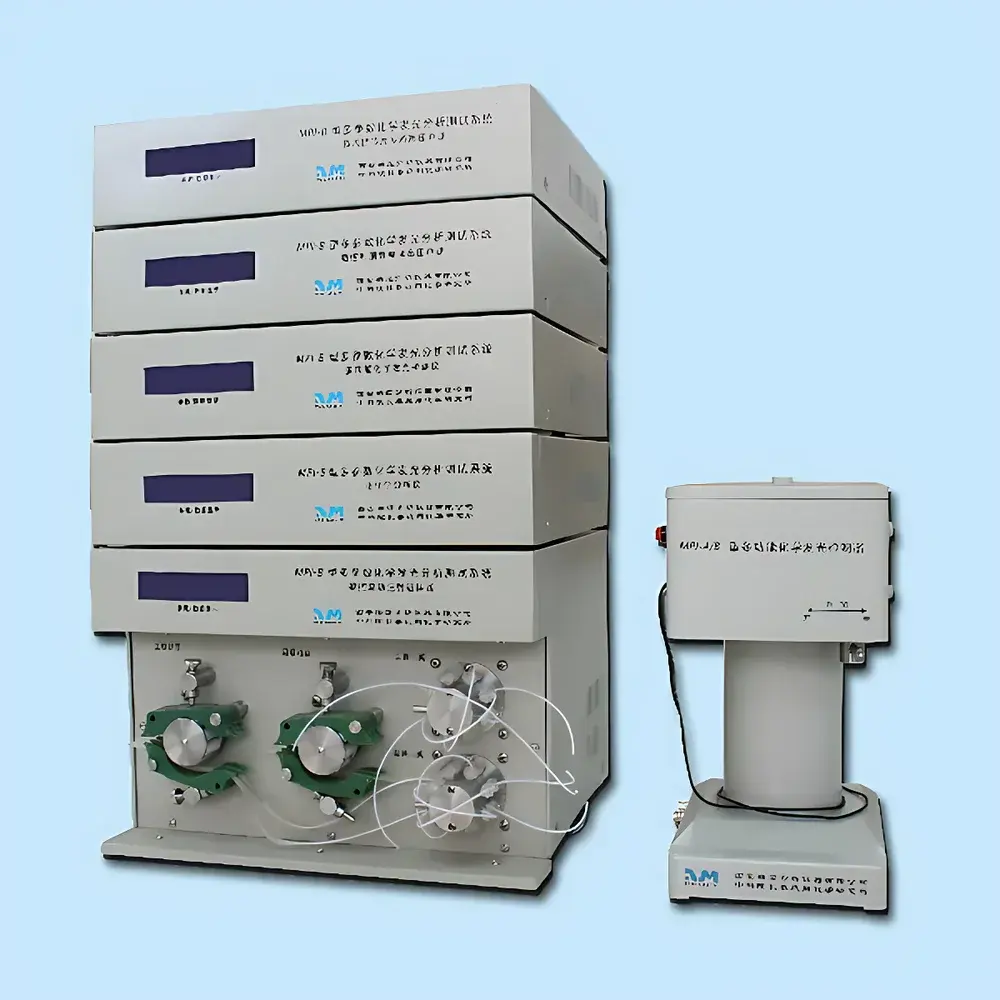
Leica HistoCore Arcadia Modular Tissue Embedding System
| Brand | Leica |
|---|---|
| Origin | Germany |
| Model | Leica HistoCore Arcadia |
| Heating Module | Arcadia H |
| Cooling Module | Arcadia C |
| Operating Temperature (Cold Plate) | –6 °C (environmentally adaptive control) |
| Cold Plate Capacity | >60 embedding cassettes |
| Interface | LCD touchscreen |
| Ergonomic Features | integrated wrist rest, adjustable magnifier |
Overview
The Leica HistoCore Arcadia Modular Tissue Embedding System is an engineered solution for histopathology laboratories requiring precision, reproducibility, and workflow flexibility in paraffin embedding—the critical pre-sectioning step that directly influences section quality, diagnostic accuracy, and downstream staining consistency. Based on the fundamental principle of controlled thermal transition—melting paraffin wax to infiltrate tissue, followed by rapid, uniform solidification—the Arcadia system separates heating and cooling functions into two dedicated, interoperable modules: the Arcadia H heated embedding station and the Arcadia C cold plate. This modular architecture eliminates thermal cross-talk, minimizes operator-induced variability, and supports GLP-compliant embedding protocols by enabling independent validation and calibration of each functional unit.
Key Features
- Modular Design: Independent Arcadia H (heating/embedding station) and Arcadia C (cold plate) allow reconfiguration of bench layout, integration with existing histology workflows, and scalability—e.g., pairing one Arcadia H with multiple Arcadia C units for high-throughput labs.
- Precision Thermal Control: Arcadia H features digitally regulated paraffin bath temperature (typically 58–62 °C), adjustable wax dispensing speed, and programmable pre-warming cycles to prevent thermal shock to delicate specimens. The Arcadia C maintains a stable –6 °C surface temperature via environmentally adaptive refrigeration—compensating automatically for ambient fluctuations (e.g., seasonal humidity or lab HVAC variance) without manual recalibration.
- Ergonomic Workflow Integration: Integrated wrist rest reduces repetitive strain during orientation; high-clarity, LED-illuminated magnifier (1.5× optical zoom) facilitates precise placement of small biopsies (<2 mm) and fragmented tissues; capacitive LCD touchscreen provides intuitive parameter setting, real-time status monitoring, and embedded operation logging.
- Validation-Ready Architecture: Each module supports user-defined operational parameters with timestamped settings—enabling traceability for internal SOP documentation and external audits aligned with ISO 15189, CAP checklist ANP.40750, and CLIA requirements for pre-analytical process control.
Sample Compatibility & Compliance
The Arcadia system accommodates standard histology cassettes (e.g., Leica E12, Sakura Sakura® 300 series), microtome-compatible molds (including gelatin-coated and metal base molds), and all common paraffin formulations (low-melting, high-melting, and rapid-infiltration waxes). It is validated for use with formalin-fixed, paraffin-embedded (FFPE) human and animal tissues—including bone, calcified specimens (with decalcification pretreatment), and lipid-rich samples (when processed with low-temperature embedding protocols). The system complies with IEC 61010-1:2010 for laboratory equipment safety and meets electromagnetic compatibility (EMC) requirements per EN 61326-1:2013. While not a medical device under FDA 21 CFR Part 820, its design aligns with Good Laboratory Practice (GLP) principles for embedding process documentation and environmental stability reporting.
Software & Data Management
The Arcadia H and C modules operate autonomously but share synchronized parameter logs via USB export (CSV format). No proprietary software installation is required; configuration files and usage history are stored locally on the embedded controller with tamper-resistant timestamps. The touchscreen interface records operator ID (via optional RFID badge integration), session start/end times, and selected temperature profiles—supporting audit trails required under ISO/IEC 17025 clause 7.9 and CAP ANP.40800 for embedding process verification. Data retention is configurable up to 12 months; exported logs integrate natively with LIMS platforms via standard file ingestion protocols.
Applications
- Routine diagnostic histology in clinical pathology laboratories performing >500 specimens/week
- Research core facilities requiring standardized embedding for multi-institutional studies (e.g., tumor biobanking, spatial transcriptomics sample prep)
- Academic teaching labs where ergonomic design and visual feedback reduce trainee learning curves
- Specialized applications including nerve biopsy embedding (requiring minimal thermal exposure), lymph node mapping (demanding precise orientation), and pediatric tissue processing (where cassette capacity and magnification are critical)
FAQ
Can the Arcadia C cold plate be used independently from the Arcadia H station?
Yes—the Arcadia C operates as a standalone unit and is frequently deployed for post-embedding block re-chilling prior to microtomy or for cooling blocks after cryo-embedding protocols.
Does the –6 °C setpoint apply to all tissue types and paraffin grades?
Yes—the –6 °C target was empirically determined across 12 paraffin formulations (including Leica EG1160, Sakura Tissue-Tek® VIP, and Milestone HyperClarity™) and validated for optimal wax crystallization kinetics and tissue integrity preservation across soft, fibrous, and fatty specimens.
Is the touchscreen interface compatible with glove use in BSL-2 environments?
The capacitive display supports operation with standard nitrile and latex gloves; no stylus is required.
How is temperature stability verified during routine maintenance?
Leica provides a certified NIST-traceable calibration kit (Order No. 1404701000) for dual-point verification (–6 °C and 60 °C) using calibrated thermistors—recommended every 6 months or per lab QA schedule.