COMECAUSE IN-Y500 Zone of Inhibition Measuring Instrument for Antibiotic Potency Assay
| Brand | COMECAUSE |
|---|---|
| Origin | Shandong, China |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Country of Manufacture | China |
| Model | IN-Y500 |
| Price | USD 11,400 (FOB Qingdao) |
Overview
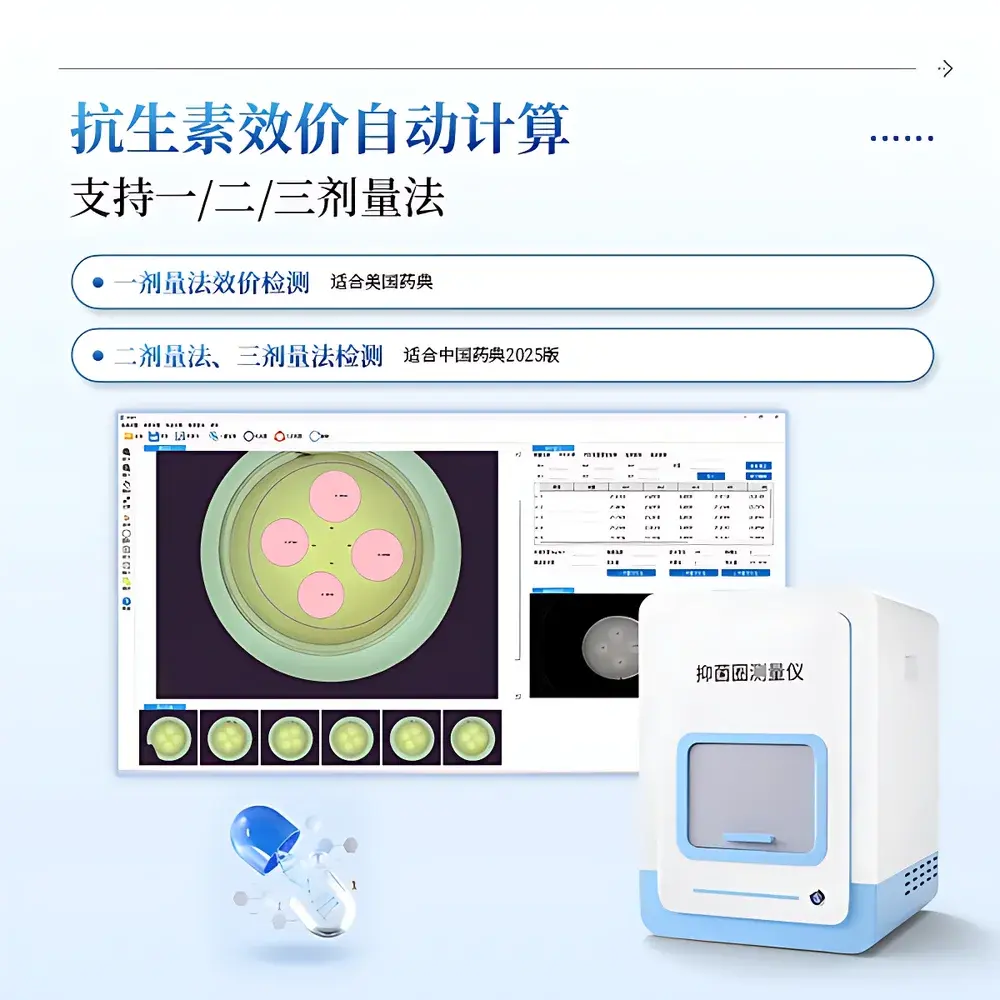
The COMECAUSE IN-Y500 Zone of Inhibition Measuring Instrument is a fully integrated, regulatory-compliant digital imaging system engineered for quantitative antibiotic potency determination via the agar diffusion (disk or well) method. It operates on the pharmacopoeial principle that the diameter of the microbial growth inhibition zone surrounding an antibiotic-impregnated disk or well is logarithmically proportional to the logarithm of its concentration—enabling precise, traceable potency calculation per USP <81>, EP 2.6.7, and ChP 2025 Volume IV General Chapter 9202. Designed for GLP- and GMP-aligned laboratories, the IN-Y500 eliminates subjective manual caliper measurements by combining high-fidelity optical imaging, adaptive edge detection algorithms, and validated dose-response modeling—ensuring metrological integrity across pharmaceutical QC, academic research, food safety testing, and environmental microbiology applications.
Key Features
- Optical Imaging Subsystem: Fully enclosed light-tight dark chamber with dual independently controllable illumination: top-mounted 360° quad-color (RGB+white) LED array for shadow-free visible-light imaging; bottom-mounted 254 nm UV-C lamp for in-chamber sterilization between runs.
- High-Resolution Capture: 25 MP color CMOS sensor coupled with a 12 MP, 12 mm focal length macro lens optimized for flat-field uniformity across standard Petri dishes (90–110 mm diameter); accommodates smaller plates via central positioning.
- Three-Mode Detection Engine: Auto-detection (edge-based contour segmentation), Circle-Fitting (least-squares circular regression for non-ideal zones), and Manual 3-Point Circle Definition (for low-contrast or fragmented inhibition boundaries).
- Dose-Response Compliance: Implements one-, two-, and three-dose assay protocols aligned with USP <81> (single-point reference) and ChP 2025 (parallel-line analysis with slope validation and validity checks per ICH Q5C).
- Metric Calibration Framework: Dual-mode calibration—automated pixel-to-mm conversion using intrinsic camera parameters, and user-verifiable physical calibration via NIST-traceable stainless steel standard plate (included); supports custom agar-height compensation.
Sample Compatibility & Compliance
The IN-Y500 supports all standard agar diffusion formats including paper disks (6 mm), stainless-steel cylinders (6.0–8.0 mm), and wells (6–10 mm diameter), compatible with common test organisms such as Staphylococcus aureus ATCC 6538, Bacillus subtilis ATCC 6633, and Escherichia coli ATCC 25922. All measurement outputs comply with ISO/IEC 17025:2017 requirements for testing laboratories—including documented uncertainty estimation per GUM (JCGM 100:2008). The system meets essential performance criteria defined in JJG (Hebei) 185–2022: maximum indication error ≤ ±0.08 mm, repeatability RSD ≤ 0.01%, and uniformity deviation ≤ 0.01% across rotational quadrants.
Software & Data Management
The embedded Windows-based application provides full 21 CFR Part 11 compliance through role-based multi-user authentication, electronic signatures, immutable audit trails (timestamped operator ID, action type, parameter changes, and result modifications), and write-protected raw image archiving. Statistical outputs—including potency (IU/mg), confidence intervals, parallelism p-values, and system suitability metrics—are exportable in PDF/A-1b format with embedded digital signatures. Cloud synchronization enables secure remote access to historical datasets via TLS 1.3–encrypted API endpoints. All calibration records, user logs, and assay reports are retained indefinitely with automatic version control and hash-based integrity verification.
Applications
- Pharmaceutical QC: Release testing of β-lactams (penicillins, cephalosporins), aminoglycosides, tetracyclines, and macrolides per compendial monographs.
- Antibiotic Residue Screening: Quantitative assessment of veterinary drug residues in milk, meat, and honey against EU MRLs (Commission Regulation (EU) No 37/2010).
- Academic Research: Dose-response curve generation, synergy studies (checkerboard assays), and antimicrobial susceptibility profiling under CLSI M07-A11 guidelines.
- Environmental Monitoring: Evaluation of biocidal efficacy in wastewater treatment effluents and surface disinfectants per EN 13697.
- Method Validation Support: Facilitates ICH Q2(R2) analytical procedure validation for specificity, linearity, accuracy, precision, and robustness.
FAQ
Does the IN-Y500 support automated validation of assay validity criteria (e.g., parallelism, linearity)?
Yes—the software applies built-in statistical tests per ChP 2025 and USP <81>, automatically flagging failures in slope ratio, residual sum of squares, and lack-of-fit during parallel-line analysis.
Can the system be integrated into an existing LIMS environment?
Yes—it supports HL7 v2.5.1 and ASTM E1384-compliant data exchange via configurable RESTful API with OAuth 2.0 authentication.
Is hardware calibration traceable to national standards?
The included stainless-steel calibration plate is certified to ISO/IEC 17025 by a CNAS-accredited metrology lab, with certificate of calibration valid for 12 months.
What environmental conditions are required for operation?
Ambient temperature: 15–25 °C; relative humidity: ≤75% RH; no direct sunlight or strong electromagnetic fields; stable AC power (220 V ±10%, 50 Hz).
How is data integrity maintained during software updates or system migration?
All databases use SQLite WAL mode with atomic transaction logging; firmware and application updates preserve audit trail continuity and do not overwrite historical metadata.