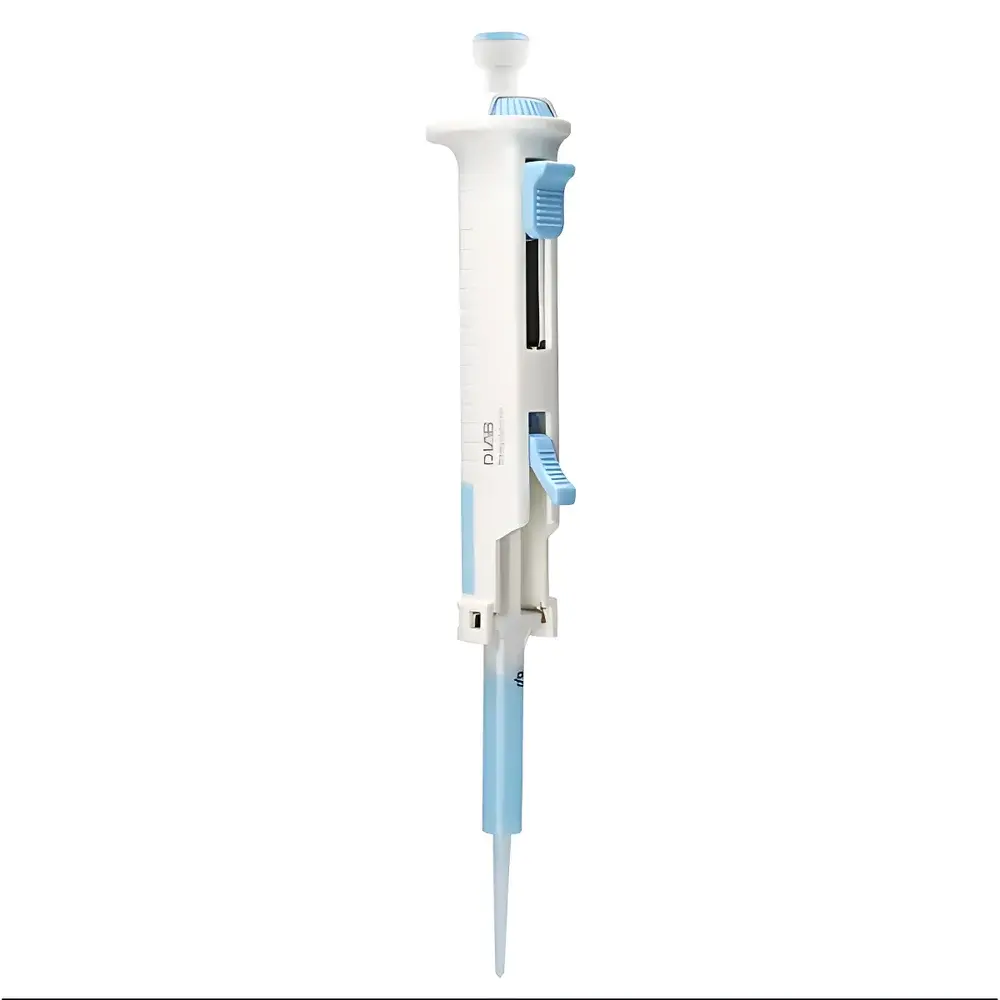
DLAB StepMate Continuous Dispenser
| Brand | DLAB |
|---|---|
| Origin | Beijing, China |
| Model | StepMate |
| Operating Principle | External Piston |
| Max. Sequential Dispensing Cycles | 48 |
| Volume Range (Dispenser Head Dependent) | 10 µL – 50 mL |
| Calibration | Factory-Calibrated per ISO 8655 |
| Dispenser Tips | 7 PP Tip Options (0.5 mL, 1 mL, 2.5 mL, 5 mL, 10 mL, 25 mL, 50 mL) |
| Tip Locking Mechanism | Integrated Mechanical Lock |
| Maintenance | Maintenance-Free Design |
| Ergonomics | Single-Handed Operation, Low Actuation Force, ISO 13485-Informed Handling Geometry |
Overview
The DLAB StepMate Continuous Dispenser is an external-piston–driven precision liquid handling instrument engineered for high-reproducibility sequential dispensing in regulated and research-intensive laboratory environments. Unlike air-displacement pipettes, the StepMate employs a mechanical piston that directly contacts the liquid via disposable polypropylene dispenser tips—eliminating compressible air gaps and minimizing vapor pressure interference. This design ensures stable, pulse-free delivery of challenging fluids including glycerol-rich reagents, viscous enzyme solutions, saturated salt buffers, and volatile organic solvents. Its fixed-volume stepping mechanism—activated by a calibrated rotary dial—enables repeatable dispensing across up to 48 consecutive cycles without manual reaspiration, significantly reducing operator fatigue and inter-cycle variability. The device complies with fundamental metrological requirements outlined in ISO 8655-3 (piston-operated volumetric apparatus) and supports traceable volume delivery under GLP-compliant workflows when used with certified tips and documented calibration records.
Key Features
- External piston architecture for direct liquid contact—optimized for high-density, high-viscosity (up to 10,000 mPa·s), and high-vapor-pressure liquids
- Ergonomic single-hand operation with tactile feedback dial and low-force actuation (< 1.2 N trigger load), conforming to ISO 13485 human factors guidance
- Factory-calibrated at multiple points across the operational range; calibration certificate provided with each unit
- Volume adjustment via precision rotary dial with clearly marked step positions—no tools or software required
- Mechanically locked tip interface prevents accidental tip ejection during dispensing, ensuring consistent plunger seal integrity
- Maintenance-free construction: no lubricants, seals, or consumable internal components subject to wear
- Seven interchangeable polypropylene dispenser tips (0.5 mL, 1 mL, 2.5 mL, 5 mL, 10 mL, 25 mL, 50 mL) — all manufactured to ISO 9001-certified cleanroom standards
- Dedicated adapter included for secure mounting of 25 mL and 50 mL tips, maintaining axial alignment and piston travel accuracy
Sample Compatibility & Compliance
The StepMate is validated for use with aqueous buffers, ethanol/water mixtures, PBS-based diagnostics reagents, PCR master mixes, and cell culture media. It demonstrates <±1.5% CV (coefficient of variation) for 100 µL–5 mL dispenses across five replicates (n=5) using ISO 8655–compliant test protocols. For regulatory applications—including IVD manufacturing, environmental testing labs (EPA Method 1600-series compatible), and QC release testing—the device supports audit-ready documentation when paired with lot-traceable tips and user-maintained logbooks. While not inherently 21 CFR Part 11–compliant (as it lacks electronic audit trail functionality), its mechanical design facilitates integration into paper-based or LIMS-linked SOPs requiring manual entry of dispense parameters and batch records.
Software & Data Management
The StepMate operates as a standalone mechanical instrument with no embedded electronics, firmware, or connectivity interfaces. All operational parameters—including selected step position, tip type, and cycle count—are manually recorded by the user. This analog architecture eliminates cybersecurity risks, firmware obsolescence, and validation overhead associated with digital dispensers. Laboratories implementing electronic recordkeeping may document StepMate usage within validated LIMS or ELN platforms using standardized templates referencing ISO/IEC 17025 clause 7.7 (result reporting) and Annex A.2 (equipment traceability). Calibration data and tip lot numbers are retained per ISO/IEC 17025 requirement 6.4.10.
Applications
- High-throughput assay plate filling (96-well and 384-well formats) for ELISA, lateral flow assay development, and nucleic acid extraction kits
- Preparation of serial dilution standards in environmental water analysis (e.g., heavy metal calibration series)
- Dispensing viscous matrices for microbiological media preparation (e.g., agarose gels, methylcellulose overlays)
- QC sampling in pharmaceutical excipient blending—where consistency across repeated micro-volume additions is critical
- Diagnostic kit manufacturing: buffer aliquoting, conjugate dilution, and control line deposition support
- Academic core facilities requiring robust, low-cost, high-duty-cycle dispensing without dependency on IT infrastructure
FAQ
Why must the first and last dispense be discarded during continuous operation?
Due to minor meniscus stabilization dynamics and initial plunger seating compliance, the first and final dispense volumes exhibit marginally higher uncertainty (typically ±3–5% deviation). Discarding these ensures all retained dispenses meet ISO 8655–specified tolerance bands.
Can the StepMate be used with corrosive solvents such as concentrated acids or chlorinated hydrocarbons?
Polypropylene tips are chemically resistant to dilute acids (≤1 M HCl, ≤0.5 M H₂SO₄) and alcohols, but not compatible with strong oxidizers (e.g., >30% HNO₃), halogenated solvents (e.g., chloroform, DCM), or bases >1 M NaOH. Consult the DLAB Chemical Compatibility Guide before use.
Is recalibration required after tip replacement?
No—each tip is pre-characterized during manufacturing and matched to the piston stroke profile. Recalibration is only necessary if physical damage occurs or after 2 years of continuous use, per ISO 8655-3 recommendation.
How is volume accuracy verified in-house?
Users may perform gravimetric verification per ISO 8655-6 using Class E2 weights, analytical balance (0.01 mg resolution), and temperature-controlled distilled water (20 °C). DLAB provides a downloadable verification protocol with acceptance criteria.