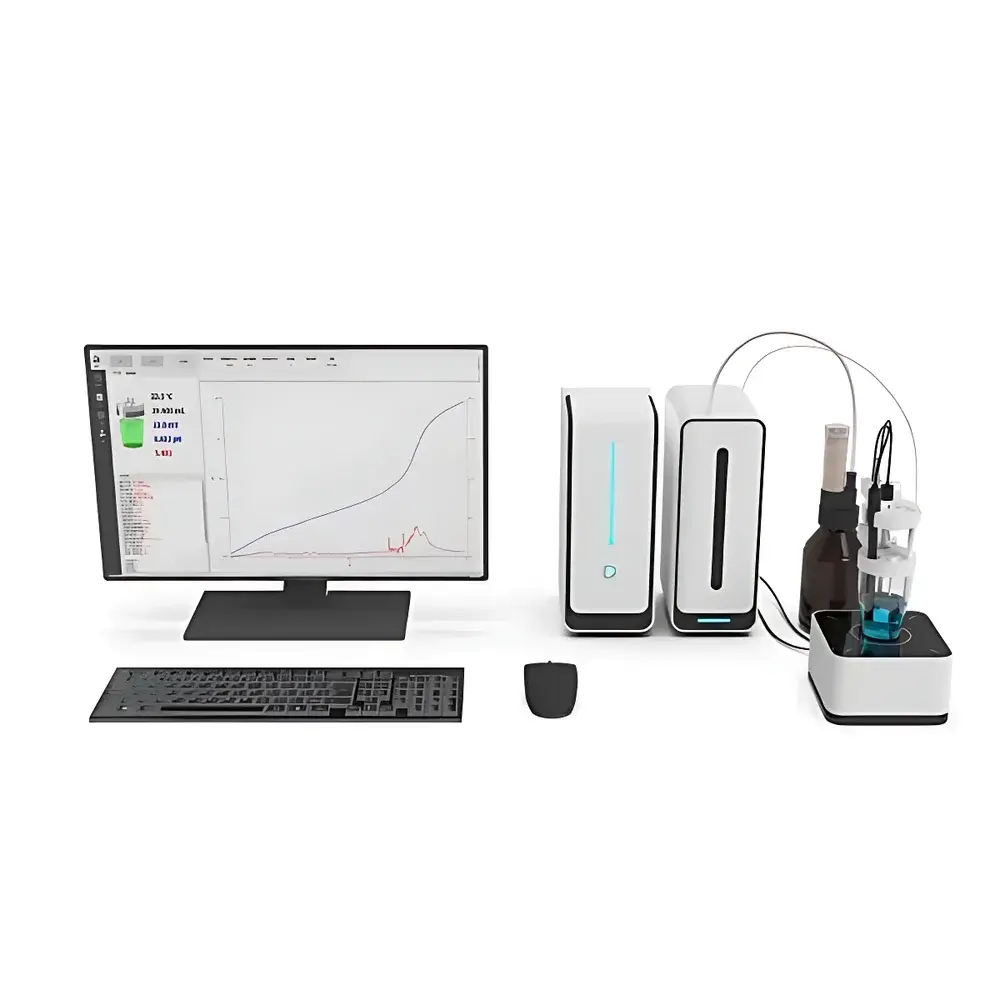
DELEITE APT-2000 Fully Automated Potentiometric Titrator
| Brand | DELEITE |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Domestic (China) |
| Model | APT-2000 |
| Pricing | Available Upon Request |
| Measurement Range | mV: −2000.0 to +2000.0 mV |
| pH | −20.000 to +20.000 pH |
| Temperature | −5 to 120 °C |
| Resolution | 0.1 mV, 0.001 pH, 0.1 °C |
| Accuracy | ±0.1 mV (±0.03% of reading), ±0.003 pH, ±0.1 °C |
| Burette Options | 1 mL / 5 mL / 10 mL / 25 mL (standard: 10 mL) |
| Burette Resolution | 1/48000 steps |
| Refill Time (100%) | 16 s |
| Stirring Method | Magnetic stirring |
| Electrode Interfaces | mV/pH input, reference electrode input, temperature probe input |
| Connectivity | USB, power interface |
| Burette Material | PTFE |
| Drive Mechanism | Micro-stepping motor |
Overview
The DELEITE APT-2000 Fully Automated Potentiometric Titrator is an engineered solution for high-precision, traceable, and reproducible potentiometric titration in regulated and research-intensive laboratory environments. Based on the fundamental principle of measuring potential difference (mV) across a working electrode–reference electrode pair during controlled reagent addition, the APT-2000 delivers quantitative endpoint detection for acid-base, redox, complexometric, precipitation, and non-aqueous titrations. Its modular architecture—comprising independent titration module, magnetic stirrer, reagent rack, and main control unit—enables flexible spatial configuration while maintaining electromagnetic isolation between subsystems. This design minimizes signal crosstalk and ensures stable electrochemical signal acquisition under variable lab conditions. The system operates within a validated measurement range of −2000.0 to +2000.0 mV and −20.000 to +20.000 pH, with temperature compensation from −5 to 120 °C—supporting both ambient and elevated-temperature titration protocols.
Key Features
- Modular, decentralized hardware layout: Each functional unit—including titration head, stirrer base, reagent station, and control console—is physically separable and independently mountable, facilitating ergonomic integration into fume hoods, glove boxes, or multi-instrument workstations.
- High-fidelity electrochemical sensing: Dual-input electrode interface supports simultaneous connection of indicator and reference electrodes; integrated temperature probe input enables real-time thermal compensation per IUPAC-recommended titration practices.
- PTFE fluidic pathway: All wetted components—including burette, tubing, and valve manifolds—are constructed from chemically inert polytetrafluoroethylene, ensuring compatibility with aggressive titrants (e.g., concentrated HClO₄, KMnO₄, AgNO₃) and minimizing adsorption or carryover.
- Precision micro-stepping motor drive: Delivers volumetric accuracy of ≤0.1% RSD across full burette capacity (1–25 mL options); resolution of 1/48,000 steps enables sub-microliter dispensing increments.
- Comprehensive self-diagnostic suite: Includes automated pH electrode calibration (single- or multi-point), burette volume verification, stirring speed validation, and electrical grounding integrity check—each logged with timestamp and operator ID.
- IPX4-rated enclosure: Front-panel seals and grommeted cable entries provide protection against incidental liquid splashes, supporting safe operation near open reagent vessels and reducing corrosion risk in humid environments.
Sample Compatibility & Compliance
The APT-2000 accommodates diverse sample matrices—including aqueous solutions, organic solvents (e.g., glacial acetic acid, ethanol, DMF), suspensions, and heterogeneous slurries—via programmable stirring profiles and adjustable electrode immersion depth. It supports standardized methods aligned with ASTM E2087 (potentiometric titration of acids), ISO 8587 (sensory analysis—general guidance), and USP (acid–base titrimetry). When configured with audit trail and role-based access control (RBAC), the system meets data integrity requirements of FDA 21 CFR Part 11 and EU Annex 11 for electronic records and signatures. All user actions—including method edits, calibration events, and result exports—are time-stamped, immutable, and attributable to authenticated roles (e.g., Analyst, Supervisor, Administrator).
Software & Data Management
The proprietary TitraSoft™ PC software provides full instrument control, method development, and compliant reporting. Users define custom titration sequences—including dynamic slope detection, fixed-volume additions, or equivalence-point interpolation—using a graphical workflow editor. The embedded formula editor supports user-defined calculations (e.g., % purity = (Vₜᵢₜᵣₐₙₜ × Mₜᵢₜᵣₐₙₜ × F × 100) / Wₛₐₘₚₗₑ), enabling direct derivation of regulatory-compliant results without post-processing. Real-time titration curves (mV vs. volume or dE/dV vs. volume) are rendered with dual-axis scaling; historical runs are searchable by sample ID, date, method name, or operator. Reports export natively to PDF (with digital signature support) and Excel (.xlsx), preserving metadata such as calibration logs, environmental readings, and audit trail summaries.
Applications
- Pharmaceutical QC: Quantification of active pharmaceutical ingredients (APIs) via non-aqueous titration (e.g., amine hydrochlorides in acetic anhydride), residual chloride in excipients, and assay of chelating agents (EDTA) in parenteral formulations.
- Food & Beverage: Total acidity (TA) in wines and juices, salt content (as NaCl) in processed foods, and preservative (sorbic/benzoic acid) determination using potentiometric back-titration.
- Environmental Analysis: Cyanide speciation (free vs. complexed) in wastewater, alkalinity profiling in surface water, and chloride monitoring in drinking water per EPA Method 325.1.
- Materials Science: Acid number and saponification value in lubricants and biodiesel, free fatty acid content in edible oils, and catalyst residue quantification in polymer synthesis intermediates.
- Academic Research: Development and validation of novel titration endpoints for redox-active metal complexes, kinetic studies of slow-reacting systems via stop-flow titration modules, and teaching laboratories requiring GLP-aligned student data handling.
FAQ
Does the APT-2000 support GLP/GMP-compliant operation?
Yes—when deployed with enabled audit trail, electronic signatures, and role-based permissions, the system satisfies core ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate) and aligns with FDA 21 CFR Part 11 Subpart B requirements.
Can the instrument perform automatic reagent preparation?
Yes—the built-in liquid handling engine supports programmable multi-step dilution and standardization workflows, including sequential aspiration, mixing, and dispensing with defined intervals and repetitions.
Is remote monitoring or network integration possible?
The APT-2000 connects via USB 2.0 to Windows-based host PCs; optional Ethernet-to-USB bridge devices enable integration into local area networks for centralized method distribution and backup—though native TCP/IP control is not supported.
What burette sizes are validated for ISO/IEC 17025 traceability?
All factory-supplied burettes (1 mL, 5 mL, 10 mL, 25 mL) include individual calibration certificates traceable to NIM (National Institute of Metrology, China) standards, with uncertainty budgets provided per ISO/IEC 17025:2017 Clause 6.4.
How is electrode drift compensated during long-duration titrations?
The software applies real-time baseline correction using pre-titration stabilization periods and adaptive slope filtering; users may also configure periodic zero-point recalibration at user-defined volume intervals.