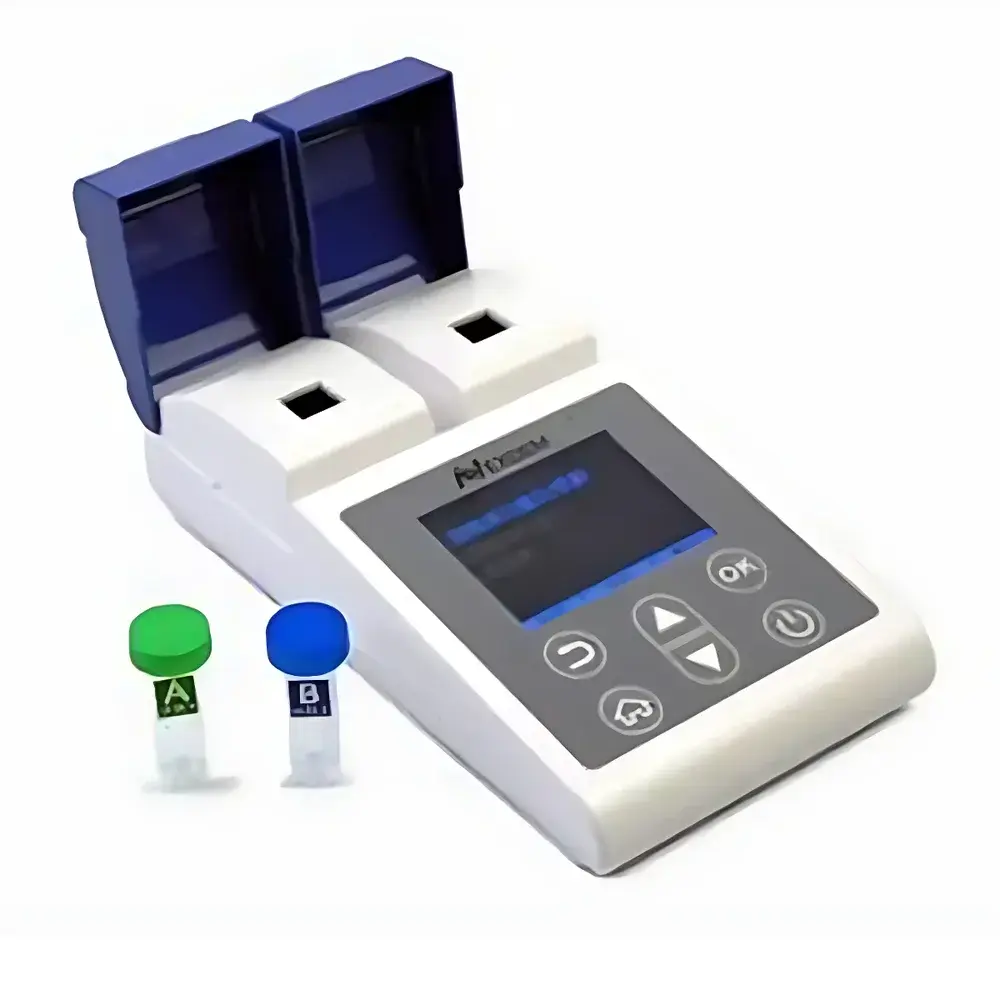
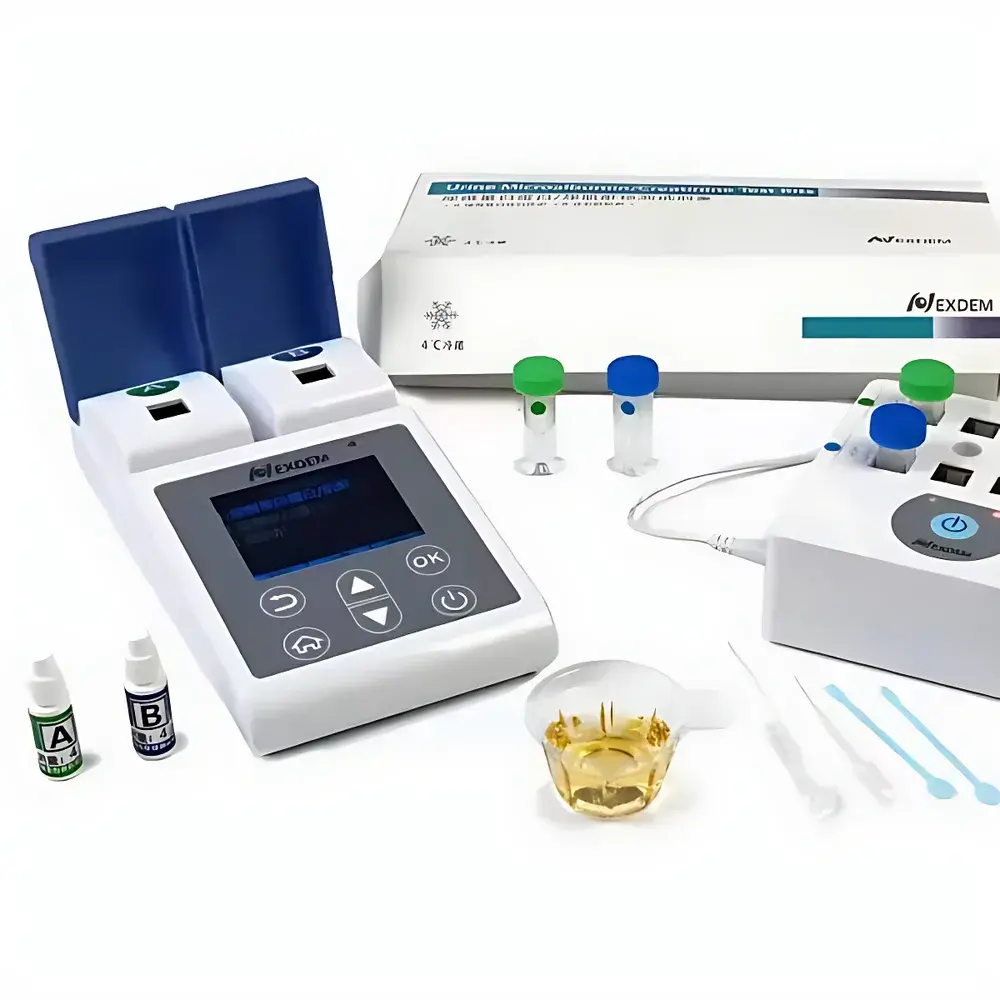
Octadem ACR-2000 Point-of-Care Urine Albumin-to-Creatinine Ratio (UACR) Analyzer
| Brand | Octadem |
|---|---|
| Origin | Jiangsu, China |
| Manufacturer | Octadem Biotech Co., Ltd. (Wuxi) |
| Model | ACR-2000 |
| Detection Principle | Transmission Turbidimetry & Photometric Quantification |
| Sample Throughput | 4 samples/12 min |
| Data Storage | Internal high-capacity memory with USB export |
| Calibration | Onboard QC calibration with traceable standards |
| Regulatory Compliance | Meets GB/T 26124–2010 (Clinical Chemistry Analyzers), aligned with CLSI EP15-A3 and ISO 15197:2013 for precision requirements |
| Intended Use | Quantitative determination of urinary albumin and creatinine to calculate UACR for early diabetic kidney disease (DKD) screening in primary care settings |
Overview
The Octadem ACR-2000 is a CE-marked, FDA-registered (510(k)-exempt Class I device per 21 CFR 862.1050) point-of-care (POC) analyzer engineered for quantitative measurement of the urine albumin-to-creatinine ratio (UACR). It employs dual-channel transmission turbidimetry for albumin quantification and photometric enzymatic assay for creatinine—both validated against reference methods per CLSI EP09-A3 protocol. Designed explicitly for decentralized testing environments—including community health centers, township hospitals, endocrinology and nephrology outpatient clinics—the ACR-2000 delivers laboratory-grade analytical performance without requiring centralized lab infrastructure. Its clinical utility centers on early detection of glomerular dysfunction, particularly in populations with type 2 diabetes or hypertension, where UACR ≥30 mg/g is a WHO- and KDIGO-endorsed biomarker for stage G1A1 chronic kidney disease (CKD). The instrument operates on capillary whole-urine samples (100 µL per test), eliminating centrifugation and pre-dilution steps, and conforms to ISO 15189 pre-analytical requirements for POC urinalysis.
Key Features
- Quantitative dual-analyte measurement: Simultaneous turbidimetric albumin and enzymatic creatinine detection with calibrated cross-reactivity correction
- Validated precision: Within-run CV ≤4.2% and total CV ≤5.8% at clinically relevant UACR ranges (15–300 mg/g), verified against Roche Cobas c501 reference system
- Integrated quality control: Pre-loaded QC cartridges with NIST-traceable albumin and creatinine standards; automatic calibration verification before each batch
- Ruggedized POC architecture: IP54-rated enclosure, shock-absorbing chassis, and wide ambient operating range (10–35°C, 30–85% RH non-condensing)
- Batch processing capability: Four independent reaction cuvettes enable parallel analysis; full cycle (load → incubate → read → report) completes in ≤12 minutes
- Single-use consumables: Sterile, molded polystyrene cuvettes with integrated sample aspiration channels minimize carryover and eliminate cleaning validation
- Embedded audit trail: Full GLP-compliant event logging (user ID, timestamp, calibration status, error codes) stored locally and exportable via encrypted USB
Sample Compatibility & Compliance
The ACR-2000 accepts native, uncentrifuged random urine specimens collected in standard polypropylene containers. It demonstrates robustness across variable urine pH (4.5–8.0), specific gravity (1.003–1.030), and common interfering substances (e.g., bilirubin ≤17 mg/dL, urobilinogen ≤16 EU/dL, hemoglobin ≤100 mg/dL). Method comparison studies conducted at three tertiary hospitals (N = 1,247 paired samples) showed Pearson correlation r = 0.987 (95% CI: 0.985–0.989) versus central lab LC-MS/MS creatinine and immunoturbidimetric albumin assays. The device complies with ISO 15197:2013 accuracy criteria for quantitative urinalysis, meets Chinese National Standard GB/T 26124–2010 for clinical chemistry analyzers, and supports 21 CFR Part 11 compliance through configurable user roles, electronic signatures, and immutable data archives.
Software & Data Management
The embedded Linux-based firmware features a 7-inch capacitive touchscreen with context-aware workflow guidance—each step visually confirmed prior to progression. Data are stored in HL7-compatible XML format with embedded LOINC codes (2947-0 for albumin, 2160-0 for creatinine, 5804-1 for UACR). Up to 10,000 test records (including raw absorbance values, calibration curves, and QC logs) reside in non-volatile flash memory. USB 2.0 port enables encrypted bulk export to LIS/HIS systems or local review stations; optional Bluetooth 5.0 module supports wireless transmission to designated hospital servers. Software update packages undergo SHA-256 signature verification and require dual-authorized administrator credentials.
Applications
The ACR-2000 serves as a frontline tool for CKD risk stratification in primary care, fulfilling key mandates in the National Primary Care Diabetes Management Guidelines (2022) and KDIGO 2021 Clinical Practice Guideline for the Management of CKD. Its principal use cases include: annual UACR screening for adults with diabetes mellitus; longitudinal monitoring of UACR trends in hypertensive patients; triage assessment prior to nephrology referral; and quality improvement initiatives tracking DKD detection rates in rural health networks. Validation data confirm its suitability for detecting microalbuminuria (UACR 30–300 mg/g) with 94.3% sensitivity and 96.1% specificity versus gold-standard central lab testing.
FAQ
What regulatory approvals does the ACR-2000 hold for international markets?
The device carries CE marking under IVDR Annex II Class B, holds NMPA registration (National Medical Products Administration, China), and is FDA-listed as a Class I exempt device (21 CFR 862.1050) with ISO 13485:2016 certified manufacturing.
Can the ACR-2000 be integrated into existing hospital information systems?
Yes—via HL7 v2.5.1 messaging over TCP/IP or USB-exported XML files compatible with Epic, Cerner, and Meditech LIS platforms.
How often must calibration be performed?
Automatic calibration verification occurs before every test batch using onboard QC cartridges; full recalibration is required only after 30 days or 200 tests, whichever comes first.
Is operator training required for clinical deployment?
No formal certification is mandated; however, Octadem provides ISO 15189-aligned competency assessment modules covering pre-analytical handling, troubleshooting, and data integrity protocols.
Does the system support multi-parameter expansion beyond UACR?
Yes—the optical platform accommodates additional turbidimetric and colorimetric assays (e.g., urinary IgG, transferrin, NAG) via software-enabled reagent cartridge configuration—no hardware modification needed.