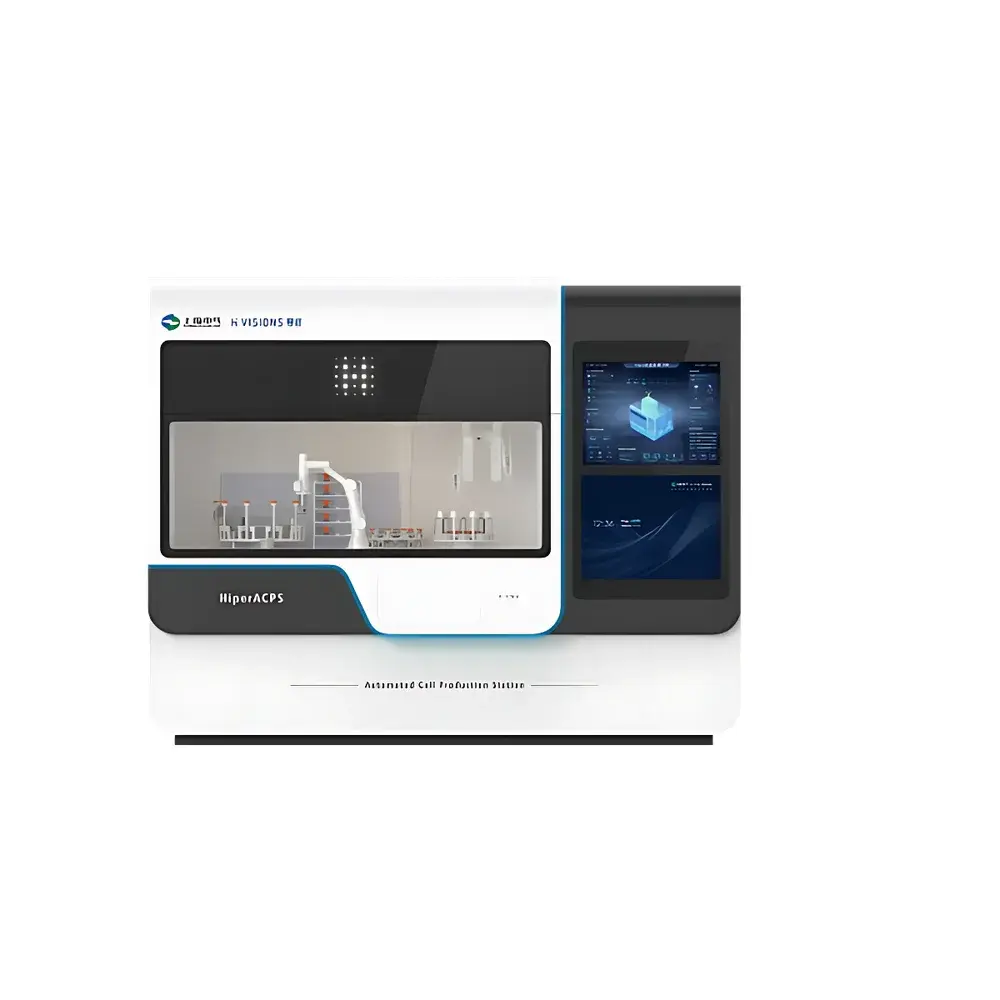
HiperACPS Automated Cell Culture Workstation
| Brand | Shanghai Electric H Visions |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Country of Origin | China |
| Model | HiperACPS |
| Pricing | Upon Request |
Overview
The HiperACPS Automated Cell Culture Workstation is an integrated, closed-system platform engineered for end-to-end automation of mammalian cell culture processes—from cryopreserved vial thawing and seed cell activation to expansion, media exchange, harvest, centrifugation, real-time monitoring, and aliquoting. Designed in accordance with core principles of aseptic processing and process analytical technology (PAT), the system operates under ISO Class 5 (A-grade) laminar airflow conditions within its internal chamber, ensuring environmental control equivalent to Grade A cleanroom standards per EU GMP Annex 1 and USP <1043>. Its architecture supports both adherent and suspension cell lines—including CHO, HEK293, hybridoma, and iPSC-derived lineages—enabling reproducible, operator-independent execution of complex, multi-day culture protocols without manual intervention.
Key Features
- Full-process automation: Programmable workflow modules cover cryovial retrieval, controlled-rate thawing, seeding, feeding, passaging, harvest, centrifugation, and post-harvest dispensing—all within a single enclosed platform.
- A-grade internal environment: Integrated HEPA-filtered laminar flow (≥99.995% @ 0.3 µm), positive pressure control, and real-time particle monitoring ensure sustained ISO Class 5 air quality during operation.
- Modular hardware design: Interchangeable pipetting arms, robotic grippers, incubator-integrated bioreactor modules (5–500 mL working volume), and on-board centrifuge (max. 3,000 × g) support flexible configuration for diverse cell types and scale requirements.
- Process traceability & audit readiness: All actions—including temperature, CO₂, humidity, liquid handling volumes, and timing—are logged with timestamped metadata compliant with FDA 21 CFR Part 11 requirements for electronic records and signatures.
- Fail-safe contamination mitigation: UV-C sterilization cycles between runs, surface decontamination via hydrogen peroxide vapor (HPV) integration option, and glove-port-free operation eliminate human-mediated bioburden introduction.
Sample Compatibility & Compliance
The HiperACPS accommodates standard labware including cryovials (1.8–2.0 mL), T-flasks (T25–T175), Corning® CellSTACK® chambers, microcarrier-based spinner flasks, and single-use bioreactor bags (up to 500 mL). It supports both serum-containing and serum-free, chemically defined media formulations. The system conforms to ISO 13485:2016 for medical device quality management systems and aligns with ICH Q5D (characterization of cell substrates), ICH Q5A(R2) (viral safety), and EU GMP Annex 1 (manufacture of advanced therapy medicinal products). Validation documentation—including IQ/OQ/PQ templates, risk assessments (FMEA), and cleaning verification protocols—is provided to support regulatory submissions under EMA CAT/ICH or FDA CBER pathways.
Software & Data Management
The workstation is operated via HiperControl™ software—a Windows-based, 21 CFR Part 11-compliant platform featuring role-based user access, electronic batch record (EBR) generation, and configurable SOP-driven workflows. All sensor data (temperature, pH, dissolved oxygen, metabolite levels via optional inline probes), actuator status, and error logs are stored in an encrypted SQL Server database with automated daily backups and immutable audit trails. Raw data export supports CSV, PDF, and XML formats; integration with LIMS (e.g., LabVantage, STARLIMS) and MES systems is enabled through RESTful API and OPC UA interfaces. Electronic signature workflows include dual authorization for critical steps such as master cell bank release or final harvest approval.
Applications
- Accelerated cell line development (CLD): Parallel screening of >24 clones under identical environmental conditions to identify high-expressing, stable producers.
- Consistent seed train preparation: Standardized generation of working cell banks (WCBs) and manufacturing cell banks (MCBs) meeting USP <1043> and Ph. Eur. 5.2.12 requirements.
- Process characterization studies: Systematic DoE-based evaluation of feed strategies, passage ratios, and environmental setpoints across multiple bioreactor vessels.
- ATMP manufacturing support: GMP-aligned production of autologous and allogeneic cell therapies, including CAR-T, MSC, and dendritic cell products.
- Academic and translational research: High-throughput functional assays, differentiation protocol optimization, and long-term stability testing under controlled physiological parameters.
FAQ
Does the HiperACPS support integration with external bioreactors or upstream/downstream equipment?
Yes—via standardized digital I/O, Modbus TCP, and optional OPC UA connectivity, enabling synchronization with external stirred-tank reactors, perfusion systems, or chromatography skids.
What validation support is included with the system?
Comprehensive IQ/OQ documentation, FAT/SAT protocols, URS template, and a vendor-qualified risk assessment report are supplied. PQ support is available upon request with qualified third-party partners.
Can the system be configured for GMP-compliant manufacturing environments?
Yes—the platform meets core GMP infrastructure requirements for ATMPs when installed in ISO Class 7 or better cleanrooms and operated under validated procedures and trained personnel.
Is remote monitoring and troubleshooting supported?
Remote desktop access (with customer-configured firewall policies) and cloud-based telemetry dashboards (optional subscription) enable real-time performance oversight and predictive maintenance alerts.
What training options are available for operators and engineers?
On-site installation training (5 days), SOP development workshops, and annual refresher courses—including GMP documentation practices and change control procedures—are offered by certified application specialists.