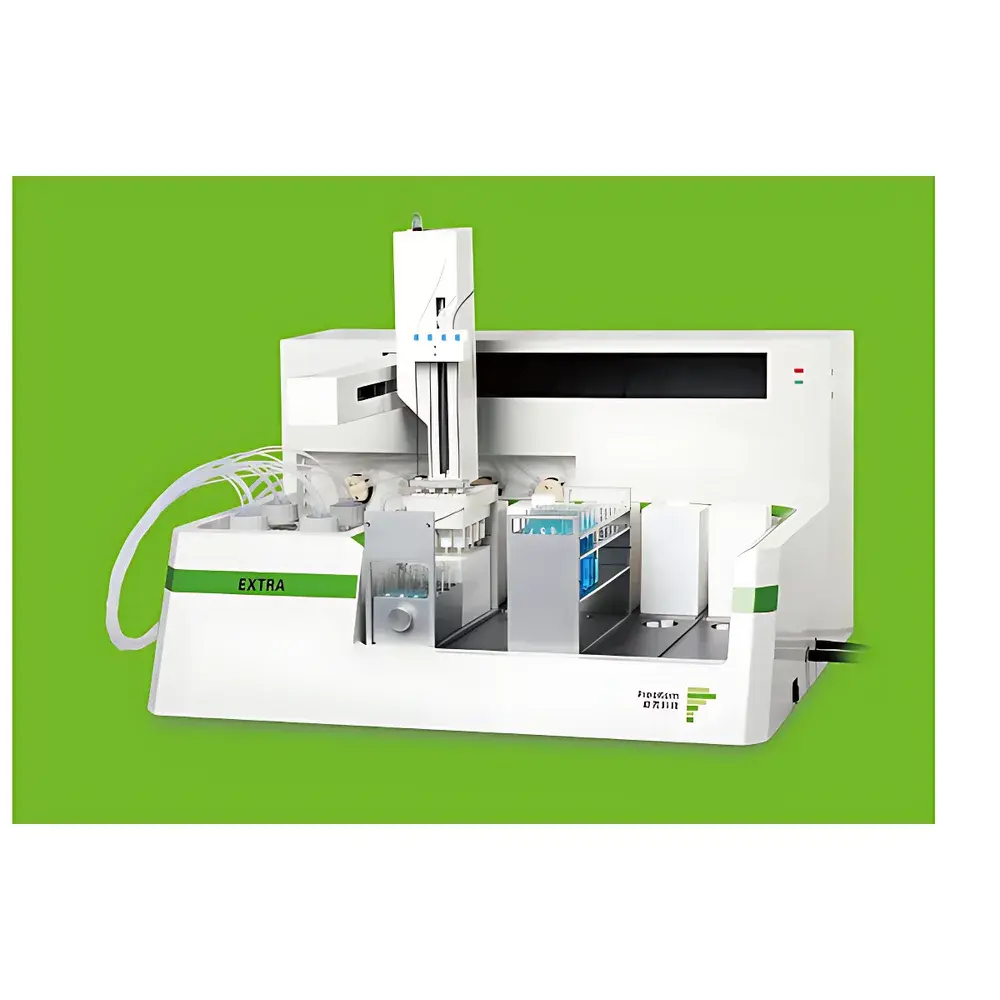
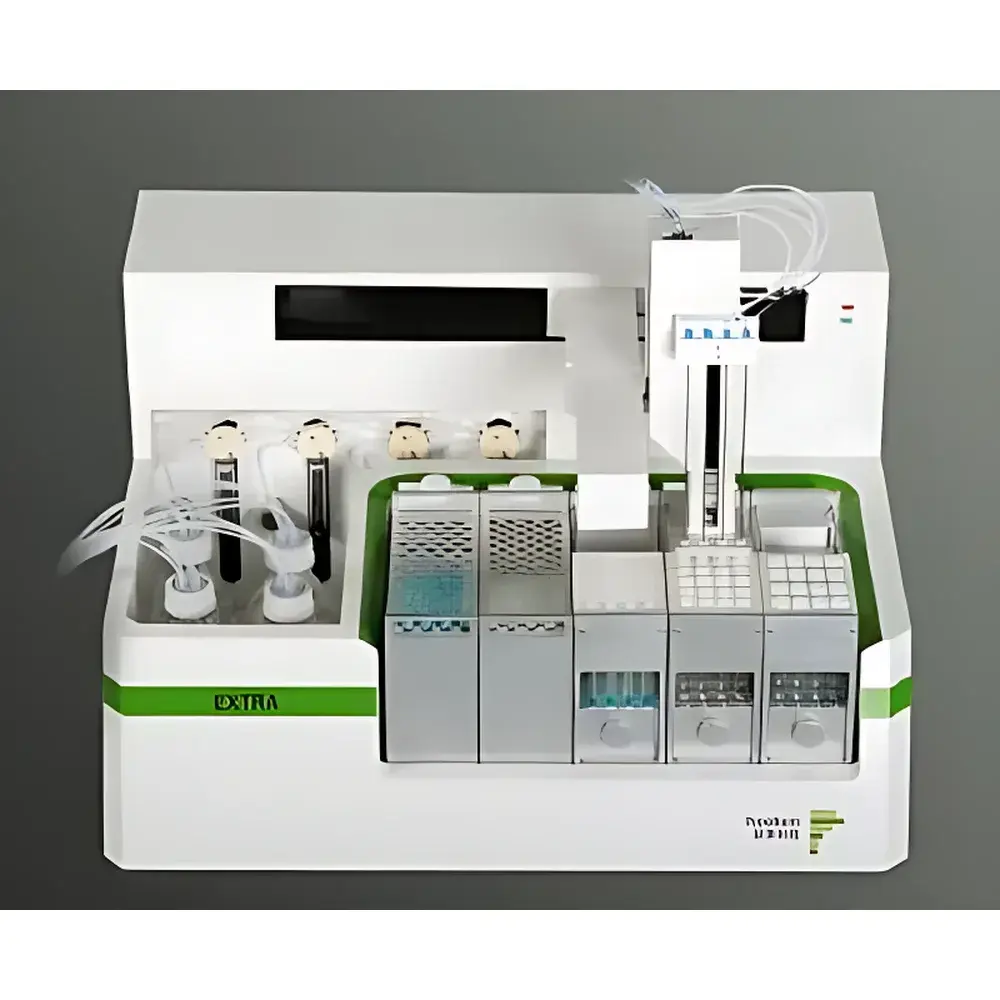
PreeKem YAO EXTRATM Fully Automated Solid Phase Extraction System
| Brand | PreeKem (Yao Technology) |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Direct Manufacturer |
| Country of Origin | China |
| Model | EXTRA |
| Automation Level | Fully Automated |
| Channel Count | 4 Parallel Channels |
| Extraction Format | Cartridge-Based SPE |
| Flow Rate Range | 0–500 mL/min |
| Solvent Selection | 8 Independent Solvent Lines |
| Sample Capacity | 4 Samples Simultaneously, Up to 108 Sequential Samples |
| Sample Loading Volume | 0–1440 mL per Sample |
| Wetted Materials | Stainless Steel (316L), Polytetrafluoroethylene (PTFE), Borosilicate Glass |
Overview
The PreeKem YAO EXTRATM Fully Automated Solid Phase Extraction System is an engineered solution for high-throughput, reproducible sample preparation in modern analytical laboratories. Based on standardized cartridge-based solid phase extraction (SPE) methodology, the system automates all critical steps—including conditioning, loading, washing, elution, and optional nitrogen-assisted drying—without manual intervention. Designed for compliance-driven environments, it supports method traceability, audit-ready operation logs, and seamless integration into regulated workflows aligned with ISO/IEC 17025, FDA 21 CFR Part 11 (when paired with validated software configurations), and GLP/GMP documentation requirements. Its architecture prioritizes precision fluid handling, minimal carryover, and environmental safety through segregated waste management and reduced solvent exposure.
Key Features
- Four independent parallel processing channels enable simultaneous SPE treatment of up to four samples, significantly reducing batch turnaround time while maintaining inter-channel consistency.
- High-precision dual-threaded lead screw robotic arm ensures sub-millimeter positioning accuracy, eliminating cartridge puncture or column misalignment during probe insertion and retraction.
- Eight-solvent selection manifold with ceramic-core multi-position valves guarantees chemical resistance against aggressive organic solvents (e.g., acetonitrile, dichloromethane, methanol) and eliminates leakage risk over extended operational cycles.
- Variable-speed syringe pump delivers flow rates from 0.0625 to 500 mL/min with coefficient of variation (C.V.) < 0.05% and absolute accuracy ≤ ±0.5%, ensuring robust method transfer across diverse matrix types.
- Dynamic liquid-level tracking probe continuously adjusts vertical position during sample loading, minimizing probe immersion depth and reducing surface contact area—critical for viscous or particulate-laden extracts.
- Dual-waste collection system separates aqueous and organic effluents into dedicated reservoirs, facilitating safe disposal and regulatory compliance with local hazardous waste classifications.
- Integrated nitrogen gas switching module enables programmable air/nitrogen purge for cartridge drying and solvent evaporation, supporting downstream concentration protocols.
- Up to nine-fraction sequential eluate collection allows method flexibility for analyte fractionation, multi-target recovery optimization, or cleanup-stage splitting.
- Comprehensive needle cleaning station performs both external rinse (outer needle surface) and internal flush (lumen clearance), with user-definable cleaning cycles and solvent combinations to suppress cross-contamination.
Sample Compatibility & Compliance
The EXTRATM accommodates standard 1 mL, 3 mL, 6 mL, and 12 mL SPE cartridges, as well as custom-format columns (e.g., disk-based or micro-SPE formats) via interchangeable adapter plates. It supports a broad range of sorbent chemistries—including C18, C8, silica, Florisil®, SAX, SCX, and mixed-mode phases—for applications spanning environmental analysis (PAHs, pesticides), food safety (veterinary drug residues, mycotoxins), clinical toxicology (drugs of abuse), and forensic chemistry (controlled substance quantification). All fluid-contact components—316L stainless steel manifolds, PTFE tubing, and borosilicate glass reservoirs—are compatible with USP Class VI and FDA-compliant materials standards. System validation documentation packages (IQ/OQ/PQ templates) are available upon request for laboratory qualification under ISO 17025 or pharmaceutical quality systems.
Software & Data Management
Controlled via an intuitive Android-based tablet interface with wireless connectivity (Wi-Fi 5 GHz), the system features drag-and-drop method builder functionality, allowing users to define step sequences, solvent selections, flow profiles, and fraction triggers without scripting. A built-in HD monitoring camera provides real-time visual feedback of cartridge status, liquid levels, and robotic motion. All method parameters, run logs, error events, and timestamped execution records are stored locally with optional cloud synchronization. Audit trails include operator ID, parameter changes, and system alerts—fully compliant with ALCOA+ data integrity principles. Custom solvent libraries and reusable method templates streamline SOP deployment across multiple instruments and operators.
Applications
The EXTRATM is routinely deployed in accredited testing laboratories for residue analysis in complex matrices: pesticide screening in fruits and vegetables (per EN 15662); veterinary drug quantification in meat and milk (EU Commission Decision 2002/657/EC); preservative and colorant profiling in beverages (ISO 13879); and illicit drug confirmation in biological fluids (SWGTOX guidelines). Its high-capacity carousel (up to 5 racks × 21 positions = 108 samples) supports unattended overnight runs for large-scale surveillance programs, while its modular design permits configuration-specific upgrades—including autosampler integration or online LC coupling—without hardware replacement.
FAQ
What types of SPE cartridges are supported?
Standard 1 mL, 3 mL, 6 mL, and 12 mL polypropylene or glass cartridges with fritted beds; custom adapters available for disk-based and micro-cartridge formats.
Can the system be integrated into a LIMS environment?
Yes—via CSV log export and optional API-enabled middleware for bidirectional data exchange with major LIMS platforms (e.g., LabWare, Thermo Fisher SampleManager).
Is method validation support provided?
PreeKem supplies IQ/OQ/PQ protocol templates, performance verification checklists, and calibration certificates traceable to NIST standards.
How is carryover minimized between runs?
Through programmable dual-stage needle cleaning (external + internal), post-run column purging, and automated solvent line flushing with user-defined dwell times.
Does the system meet regulatory requirements for electronic records?
When operated with enabled audit trail logging and user authentication, it satisfies core elements of 21 CFR Part 11 for electronic signatures and record retention in GxP environments.