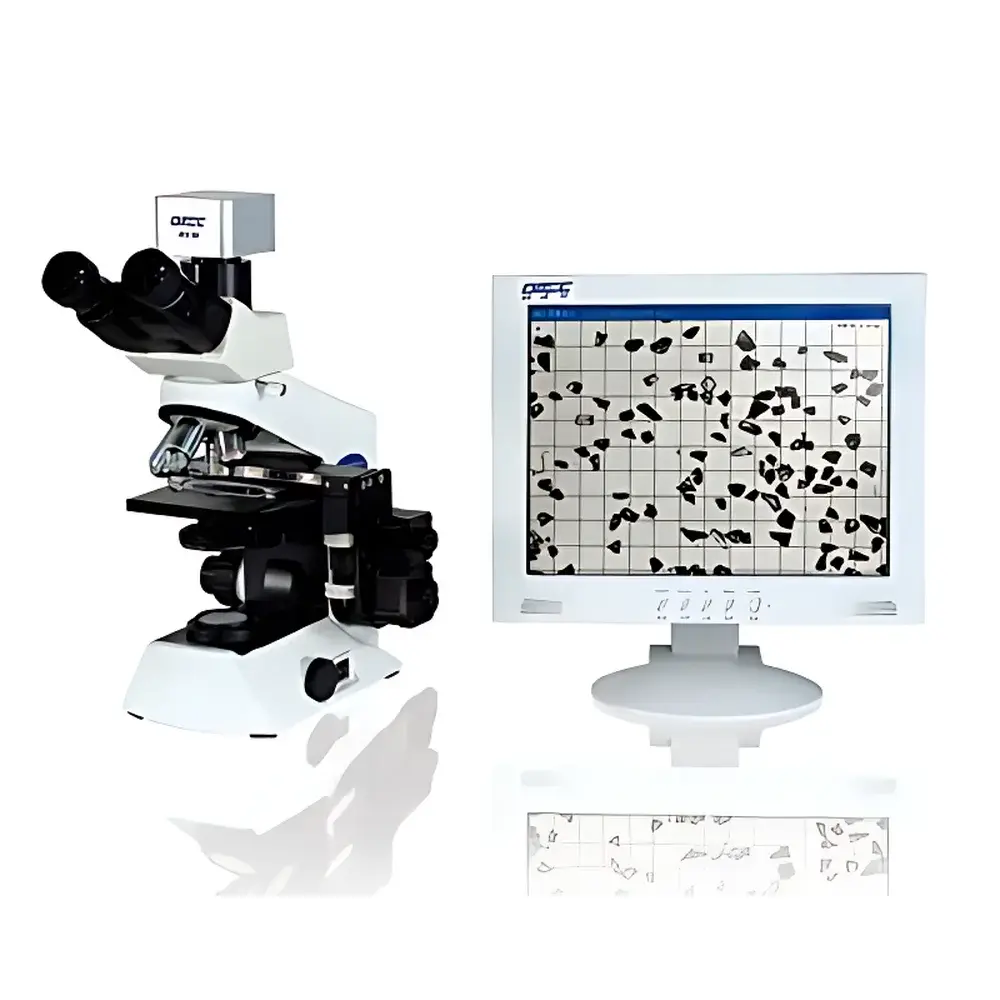
OMEC PIP9.1 Particle Image Analyzer
| Brand | OMEC |
|---|---|
| Origin | Guangdong, China |
| Manufacturer Type | Direct Manufacturer |
| Region Category | Domestic (China) |
| Model | PIP9.1 |
| Pricing | Available Upon Request |
| Measurement Principle | Digital Microscopy-Based Image Analysis |
| Test Range | 0.5–3000 µm |
| Repeatability Error | <3% |
| Output Data | Particle Size Distribution Table & Curve (by Equivalent Area Circle, Equivalent Short Diameter, etc.), Roundness Distribution Table & Curve, D10/D50/D90, Specific Surface Area (calculated), Mean Roundness, Roundness Range, Segment-Averaged Roundness, Representative Single-Particle Images |
| Core Hardware | Olympus Biological Microscope (Japan), 1.3 MP CMOS Sensor, USB 2.0 Interface |
| Key Automation | Auto-Focus, Auto-Field Scanning, Auto-Illumination Adjustment |
| Compliance Context | Aligns with GB/T 6408–2008 (Diamond Micropowder Standard), Supports GLP Documentation Workflow |
Overview
The OMEC PIP9.1 Particle Image Analyzer is a high-precision digital microscopy system engineered for direct morphological and dimensional characterization of dry or dispersed particulate samples. Unlike ensemble-averaging techniques such as laser diffraction or dynamic light scattering, the PIP9.1 employs static image acquisition under magnified optical observation—leveraging an Olympus biological microscope platform coupled with a 1.3-megapixel CMOS sensor—to capture high-fidelity two-dimensional projections of individual particles. Each acquired image undergoes algorithmic segmentation and geometric parameterization to compute multiple equivalent diameters (e.g., area-equivalent circle diameter, Feret short diameter) and shape descriptors—including roundness, aspect ratio, convexity, and solidity—based on ISO 9276-6:2008 (Representation of Results of Particle Size Analysis – Part 6: Descriptions and Quantities in Particle Shape Analysis). The instrument operates across a validated dynamic range of 0.5 µm to 3000 µm, making it suitable for coarse abrasives, submicron pigments, and intermediate-grade mineral fillers alike. Its primary role is not as a standalone primary sizing tool but as a complementary verification and root-cause analysis platform—particularly where particle shape, agglomeration state, or outlier detection (e.g., trace oversized fragments in polishing slurries) is critical to process performance or regulatory compliance.
Key Features
- Optical core based on an Olympus biological microscope (Japan-sourced), ensuring long-term mechanical stability, chromatic fidelity, and parfocal consistency across magnification objectives.
- 1.3 MP global-shutter CMOS imaging sensor with optimized signal-to-noise ratio for reliable edge detection at low contrast boundaries—essential for irregular, translucent, or low-refractive-index particles (e.g., silica, calcium carbonate).
- Fully automated field scanning: motorized XY stage with programmable tiling ensures statistically representative sampling without operator-induced bias; configurable scan density and exclusion zones support ASTM E2455-22 (Standard Guide for Sampling Powder Metallurgy Materials) principles.
- Real-time auto-focus algorithm utilizing contrast gradient maximization—eliminates manual focus drift during extended acquisition sessions and maintains sub-micron axial precision across heterogeneous sample topographies.
- Dynamic illumination control: LED-based Köhler illumination system with closed-loop feedback adjusts intensity per field to maintain optimal gray-level distribution, minimizing saturation or underexposure artifacts in mixed-size populations.
- Native USB 2.0 interface ensures plug-and-play compatibility with Windows 10/11 systems without proprietary drivers; supports time-stamped metadata embedding (acquisition date, objective lens ID, calibration timestamp) per image file.
Sample Compatibility & Compliance
The PIP9.1 accommodates dry powders deposited on conductive stubs (for SEM-style mounting), liquid suspensions on standard glass slides (with coverslip sealing), and air-dispersed aerosols captured on membrane filters. It is routinely deployed in quality control laboratories adhering to GB/T 6408–2008—the national standard specifying image analysis methodology for diamond micropowder grading—and serves as a technical reference for internal SOPs aligned with ISO 13322-1:2014 (Particle Size Analysis – Image Analysis Methods – Part 1: Static Image Analysis). While not intrinsically 21 CFR Part 11 compliant, its software architecture supports audit-trail-enabling configurations (user login logs, parameter change history, raw image archiving) when integrated into validated LIMS environments operating under GLP or GMP frameworks.
Software & Data Management
OMECSight v4.x software provides a modular workflow: image acquisition → background correction → threshold optimization → particle separation (including watershed de-agglomeration) → feature extraction → statistical aggregation. All raw TIFF images are stored losslessly with embedded EXIF tags containing magnification, pixel-to-µm calibration factor, and environmental metadata. Export options include CSV (for Excel or Python post-processing), PDF reports with embedded histograms and scatter plots (roundness vs. size), and annotated PNG thumbnails of user-selected “representative” or “outlier” particles. Batch processing supports up to 500 fields per run, with memory-mapped I/O preventing system lockup during large-session analysis.
Applications
- Quality assurance of superabrasives: detecting fractured diamond grains or silicon carbide splinters that compromise grinding wheel integrity.
- Coating formulation development: correlating pigment particle circularity with dispersion stability and film opacity uniformity.
- Pharmaceutical excipient release: verifying absence of micronized lactose agglomerates (>100 µm) that impair inhaler dose uniformity (per USP <601>).
- Mineral processing: quantifying flake graphite aspect ratio shifts after milling—directly linked to conductivity performance in battery anodes.
- Regulatory documentation: generating visual evidence packages for FDA pre-submission meetings where morphology deviations require justification beyond D50 values.
FAQ
Does the PIP9.1 require certified reference standards for calibration?
Yes—NIST-traceable spherical latex standards (e.g., Thermo Scientific’s NIST SRM 1963) are recommended for pixel-to-length calibration at each magnification; shape descriptor validation uses ISO-defined test patterns.
Can the system analyze particles in suspension without drying?
Yes, provided the suspension medium has sufficient refractive index contrast and settling is minimized via surfactant stabilization or rapid acquisition; optional heated stage prevents solvent crystallization artifacts.
Is roundness calculated using the Wadell definition or alternative metrics?
Roundness is computed per Wadell (perimeter² / (4π × area)), with supplementary convexity and aspect ratio derived from minimum bounding rectangle geometry.
How does the auto-scan ensure statistical representativeness?
Scan path follows a stratified random grid pattern; minimum field count is determined by ANSI/ASQ Z1.4 sampling plans based on batch size and AQL thresholds.
Can raw images be reprocessed with updated algorithms post-acquisition?
Yes—OMECSight retains full parametric history; users may reload original TIFF stacks and apply revised segmentation thresholds or shape filters without rescanning.