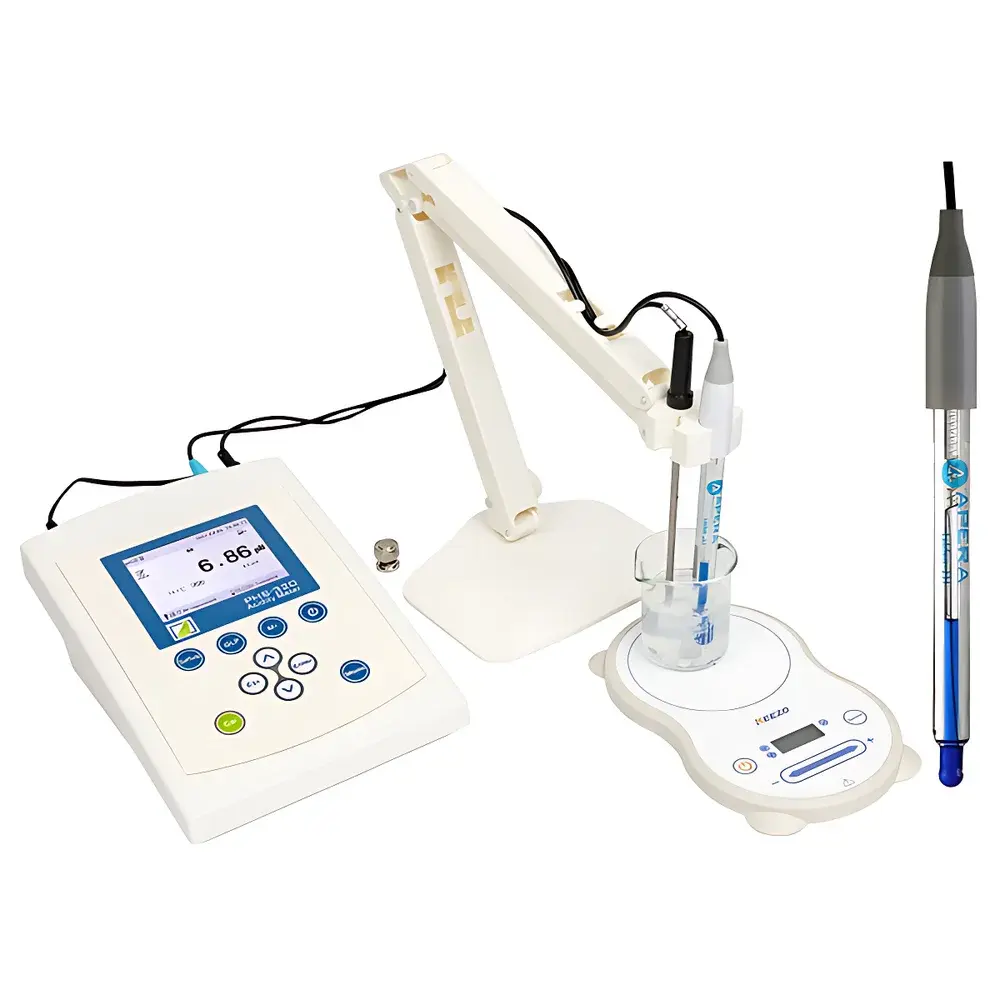
FangZhou PHS-320 Cosmetic pH Meter
| Brand | FangZhou |
|---|---|
| Origin | Sichuan, China |
| Manufacturer Type | Authorized Distributor |
| Product Category | Domestic |
| Model | PHS-320 Cosmetic pH Meter |
| Instrument Type | Multiparameter Benchtop pH Meter |
| Parameter Types | pH, mV, Rel.mV, ORP, ISE, Temperature (°C/°F) |
| Measurement Accuracy | ±0.002 pH |
| pH Range | −5.000 to 20.000 pH |
| Resolution | 0.001 / 0.01 / 0.1 pH (user-selectable) |
| mV Range | −2000.00 to +2000.00 mV |
| mV Resolution | 0.01 / 0.1 / 1 mV (user-selectable) |
| Temperature Range | −20.0 to 135.0 °C |
| Temperature Resolution | 0.1 °C |
| Temp. Accuracy | ±0.2 °C ±1 digit |
| Input Impedance | ≥3 × 10¹² Ω |
| Calibration | Up to 5-point auto-recognized calibration using NIST-traceable buffers (33 standard solutions across 7 buffer groups per JJG 119, DIN 19266/19267, ASTM, JIS Z8802, and USP) |
| ATC | Automatic or Manual Temperature Compensation |
| Iso-potential Adjustment | −20.000 to +20.000 pH |
| Data Storage | ≥100,000 measurements with timestamp (date/time-stamped), GLP-compliant metadata |
| Connectivity | RS232 serial interface (printer, PC export, bidirectional control) |
| Power | DC 9 V, 500 mA (center-positive) |
Overview
The FangZhou PHS-320 Cosmetic pH Meter is a microprocessor-controlled, high-precision benchtop electrochemical analyzer engineered for rigorous pH, redox potential (ORP), ion-selective electrode (ISE), and millivolt (mV) measurements in regulated cosmetic, pharmaceutical, food, and environmental laboratories. Based on the potentiometric principle—measuring the electromotive force (EMF) generated at the interface between a glass pH electrode and reference electrode—the instrument delivers traceable, reproducible results compliant with international metrological standards. Its design emphasizes stability under variable ionic strength, low-conductivity, and viscous sample conditions typical of cosmetic formulations (e.g., emulsions, gels, surfactant-rich solutions), where conventional meters often suffer from slow response, junction clogging, or drift. The PHS-320 integrates an ultra-high-input-impedance amplifier (≥3 × 10¹² Ω) to minimize loading error during low-current signal acquisition, ensuring fidelity in measurements of pure water, microliter-volume samples, and high-resistance media.
Key Features
- True-color 4.3-inch TFT LCD display with graphical curve rendering, intuitive menu navigation, and real-time data visualization
- User-configurable resolution modes: 0.001 pH / 0.01 mV (high-precision mode) or coarser steps for rapid screening
- GLP-compliant data management: 10 sample IDs, 10 operator IDs, full audit trail including calibration history, electrode status, and timestamped measurement records
- Multi-point calibration support (1–5 points) with automatic buffer recognition across 33 NIST-traceable and internationally standardized solutions (JJG 119, DIN 19266/19267, ASTM D1293, JIS Z8802)
- Adaptive temperature compensation: ATC via integrated Pt1000 sensor or manual entry; compensation range −20.0 to 135.0 °C
- Five selectable measurement modes: Real-time, Timed (0–6000 s interval), Auto-Fast, Auto-Medium, Auto-Slow—optimized for dynamic equilibration in viscous or heterogeneous matrices
- Iso-potential adjustment (−20.000 to +20.000 pH) enabling compatibility with zero-reference electrodes and non-standard reference systems
- Comprehensive self-diagnostic suite: Electrode impedance check, junction potential assessment, amplifier integrity verification, and battery/PSU health monitoring
Sample Compatibility & Compliance
The PHS-320 is validated for use with diverse sample types critical to cosmetic R&D and QC: low-ionic-strength aqueous phases (e.g., toners, micellar waters), high-viscosity gels and creams (via optional FZ-600T or specialized semi-micro electrodes), protein-laden emulsions (e.g., sunscreens, moisturizers), and organic solvent-containing formulations. Its modular electrode interface supports application-specific probes—including planar surface sensors for skin or film testing, HF-resistant electrodes for acidic actives, and double-junction reference systems for sulfide- or protein-fouling environments. The instrument conforms to ISO/IEC 17025 requirements for testing laboratories and supports full 21 CFR Part 11 compliance when paired with validated software and electronic signature protocols. All calibrations are traceable to NIST SRMs, and documentation meets GLP/GMP record-keeping mandates for regulatory submissions (e.g., FDA OTC Monograph, EU CPNP, ASEAN Cosmetics Directive).
Software & Data Management
Data integrity is enforced through embedded GLP firmware: each stored measurement includes pH/mV value, temperature, electrode ID, calibration ID, operator ID, sample ID, and UTC timestamp. Internal memory retains ≥100,000 records with cyclic overwrite protection. Via RS232, users may:
- Print reports directly to thermal printers in GLP-compliant format (including calibration certificates and uncertainty statements)
- Export raw datasets to Microsoft Excel, Word, or CSV via ASCII protocol—enabling statistical process control (SPC), trend analysis, and LIMS integration
- Execute remote instrument control (e.g., initiate calibration, trigger measurement, recall stored data) using custom scripts or LabVIEW-compatible drivers
No proprietary software installation is required; native ASCII transmission ensures long-term data accessibility and archival stability.
Applications
The PHS-320 serves as a primary pH measurement platform in:
- Cosmetic formulation labs: monitoring pH stability of preservative systems (e.g., parabens, phenoxyethanol), chelating agents (EDTA), and active ingredients (vitamin C, AHAs, peptides)
- Pharmaceutical manufacturing: verifying pH of topical gels, ointments, and injectables per USP and EP 2.2.3
- Food safety testing: measuring acidity in dairy products, fermented cosmetics (e.g., kombucha-based serums), and acidified cleansers
- Environmental QA/QC: analyzing wastewater from cosmetic production facilities per EPA Method 9040C
- Academic research: kinetic studies of hydrolysis, enzymatic activity, and colloidal stability under controlled pH
FAQ
Is the PHS-320 compliant with FDA 21 CFR Part 11 for electronic records?
Yes—when operated with validated RS232 communication software, user authentication, and audit-trail-enabled data export, it satisfies predicate rule requirements for closed-system electronic records.
Can it measure pH in non-aqueous solvents like ethanol or propylene glycol?
It can provide comparative readings, but accuracy requires solvent-specific calibration and electrode selection (e.g., non-aqueous reference electrolyte); NIST-traceable validation is limited to aqueous matrices.
What is the minimum sample volume required for accurate measurement?
With the optional FZ-600T semi-micro electrode, reliable measurements are achievable with ≥200 µL; the micro-electrode variant supports ≥15 µL volumes.
Does the instrument support automatic buffer identification for all 33 standard solutions?
Yes—buffer recognition is implemented per manufacturer-defined voltage signatures and temperature-dependent EMF tables aligned with JJG 119 and DIN 19266.
How frequently must the electrode be recalibrated during continuous operation?
Per GLP best practice, recalibration is recommended before each analytical batch and after every 10–15 measurements—or immediately following exposure to extreme pH, high viscosity, or proteinaceous samples.