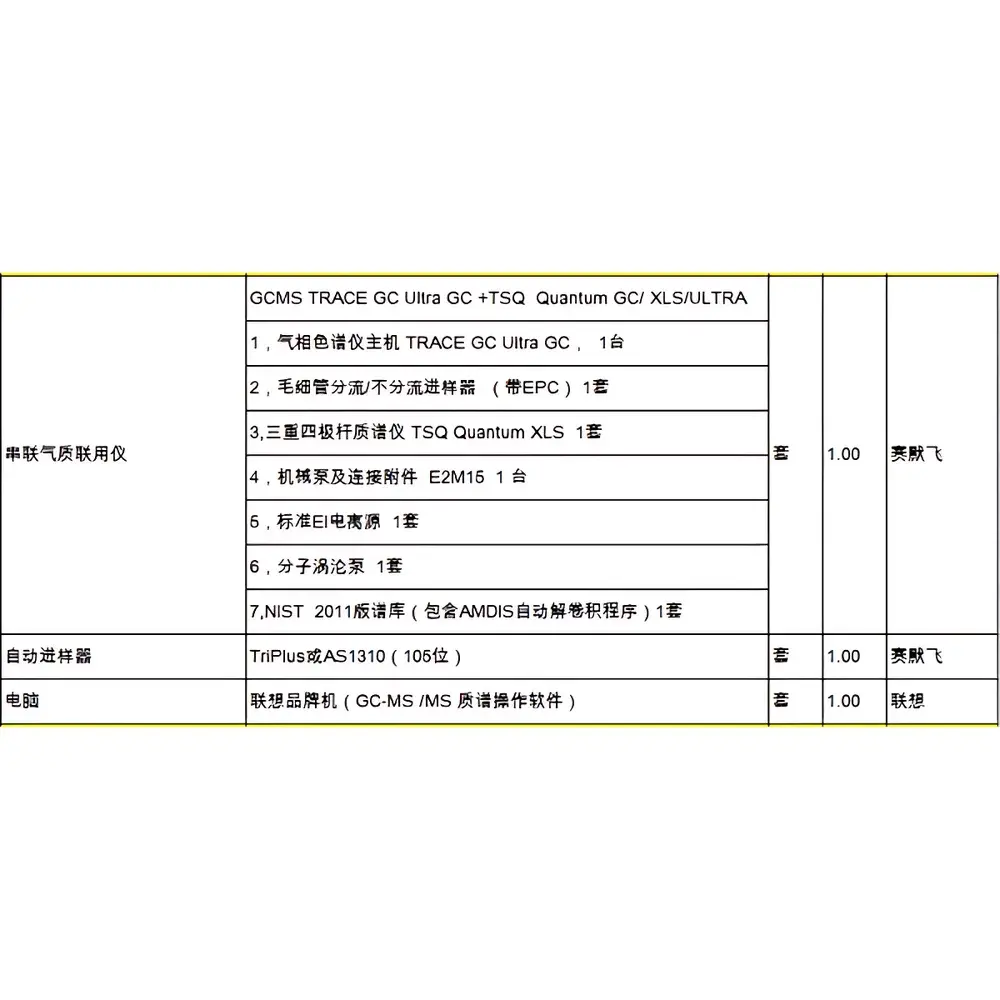
Thermo Fisher TSQ Quantum GC/XLS/ULTRA Used Tandem Quadrupole Gas Chromatography–Mass Spectrometry System
| Brand | Thermo Fisher |
|---|---|
| Origin | USA |
| Model | TRACE GC Ultra + TSQ Quantum GC/XLS/ULTRA |
| Configuration | Used, Fully Functional, Pre-Owned |
| Compliance | Meets Original Factory Specifications (as verified per pre-delivery QC checklist) |
| Service History | Full maintenance log available upon request |
| Calibration Status | Requires user-site requalification per ISO/IEC 17025 or GLP protocols |
Overview
The Thermo Fisher TSQ Quantum GC/XLS/ULTRA is a high-performance triple quadrupole gas chromatography–mass spectrometry (GC-MS/MS) system originally engineered for targeted quantitative analysis in regulated and research-intensive laboratories. This pre-owned instrument integrates the TRACE GC Ultra gas chromatograph — featuring advanced oven temperature control (±0.01 °C stability), programmable pressure/flow control, and low thermal mass inlet technology — with the TSQ Quantum series triple quadrupole mass spectrometer. The latter employs collision-induced dissociation (CID) in a QqQ configuration, enabling selective reaction monitoring (SRM) and multiple reaction monitoring (MRM) with high specificity, low detection limits (sub-pg on-column for many analytes), and robust signal-to-noise performance. Designed for routine operation in environmental testing, food safety screening, clinical toxicology, and pharmaceutical impurity profiling, this system delivers reproducible quantitation across complex matrices when operated within validated method parameters.
Key Features
- Triple quadrupole mass analyzer with independently controlled RF/DC voltages on Q1, q2 (collision cell), and Q3 for optimized ion transmission and fragmentation efficiency
- Electron ionization (EI) and optional chemical ionization (CI) sources, both capable of rapid source switching without venting
- TRACE GC Ultra with dual-channel capability, backflush functionality, and integrated autosampler support (e.g., AS1310 or TriPlus RSH)
- Advanced vacuum system utilizing dual-stage turbomolecular pumps (≥300 L/s for MS, ≥200 L/s for GC interface) ensuring stable operating pressure during extended runs
- Robust front-end electronics supporting real-time peak tracking, dwell time optimization, and dynamic MRM scheduling
- Factory-aligned ion optics and calibrated mass scale traceable to NIST SRM standards
Sample Compatibility & Compliance
This system accommodates standard 0.25–0.53 mm ID fused-silica capillary columns and supports split/splitless, PTV, and direct injection modes. It is routinely deployed for volatile and semi-volatile organic compounds (VOCs/SVOCs), pesticides, PAHs, PCBs, mycotoxins, and small-molecule drugs. From a regulatory standpoint, the hardware architecture complies with design principles aligned with ISO/IEC 17025:2017 (clause 6.4.3 on equipment verification), USP Analytical Instrument Qualification, and FDA 21 CFR Part 11 requirements when paired with compliant data systems (e.g., Thermo Xcalibur v2.3 or later with audit trail enabled). All pre-delivery inspections include leak testing, tuning verification (using perfluorotributylamine, PFTBA), and baseline noise assessment per manufacturer-recommended procedures.
Software & Data Management
Operation is managed via Thermo Scientific Xcalibur software (versions 2.3 through 3.1.65 supported), which provides full method development, acquisition control, quantitative processing (with internal standard calibration, matrix-matched curves, and isotopic dilution options), and reporting templates compliant with LIMS integration standards (ASTM E1467, ASTM E2500). Raw data files (.RAW) are stored in vendor-neutral formats compatible with third-party review tools (e.g., Compound Discoverer, Chromeleon). Audit trail functionality, electronic signatures, and user-access-level configuration are fully functional when deployed in secure network environments meeting IT validation criteria per GAMP 5 guidelines.
Applications
- Environmental analysis: Quantification of organochlorine pesticides in soil extracts per EPA Method 8081B and 8270D
- Food safety: Multi-residue screening of >300 pesticides in fruits and vegetables using QuEChERS extraction coupled with MRM transitions
- Clinical toxicology: Confirmation and quantification of benzodiazepines, opioids, and stimulants in urine and blood at sub-ng/mL levels
- Pharmaceutical quality control: Residual solvent analysis (ICH Q3C) and genotoxic impurity detection (ICH M7) in drug substances
- Forensic chemistry: Differentiation of isomeric compounds (e.g., THC vs. CBD) via diagnostic fragment ion ratios
FAQ
Is this instrument supplied with original documentation and service history?
Yes — full factory shipping records, installation certificates, and service logs (where available from prior ownership) accompany each unit. A pre-shipment technical dossier is provided prior to purchase.
What is the recommended qualification process before first use?
Users must perform Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ) per their internal SOPs, referencing Thermo’s Application Note 092292 and USP . We supply a baseline IQ/OQ checklist as part of the handover package.
Can this system be upgraded to support newer software versions or accessories?
Hardware compatibility with Xcalibur v3.x and current Thermo accessories (e.g., TriPlus RSH autosampler, TurboFlow online SPE) is confirmed; upgrade paths depend on controller firmware version and are assessed during pre-sale technical review.
Does the system include a warranty or post-installation support?
All units are covered by a standard 6-month limited hardware warranty against defects in materials and workmanship. Extended service plans and remote diagnostics support are available under separate agreement.