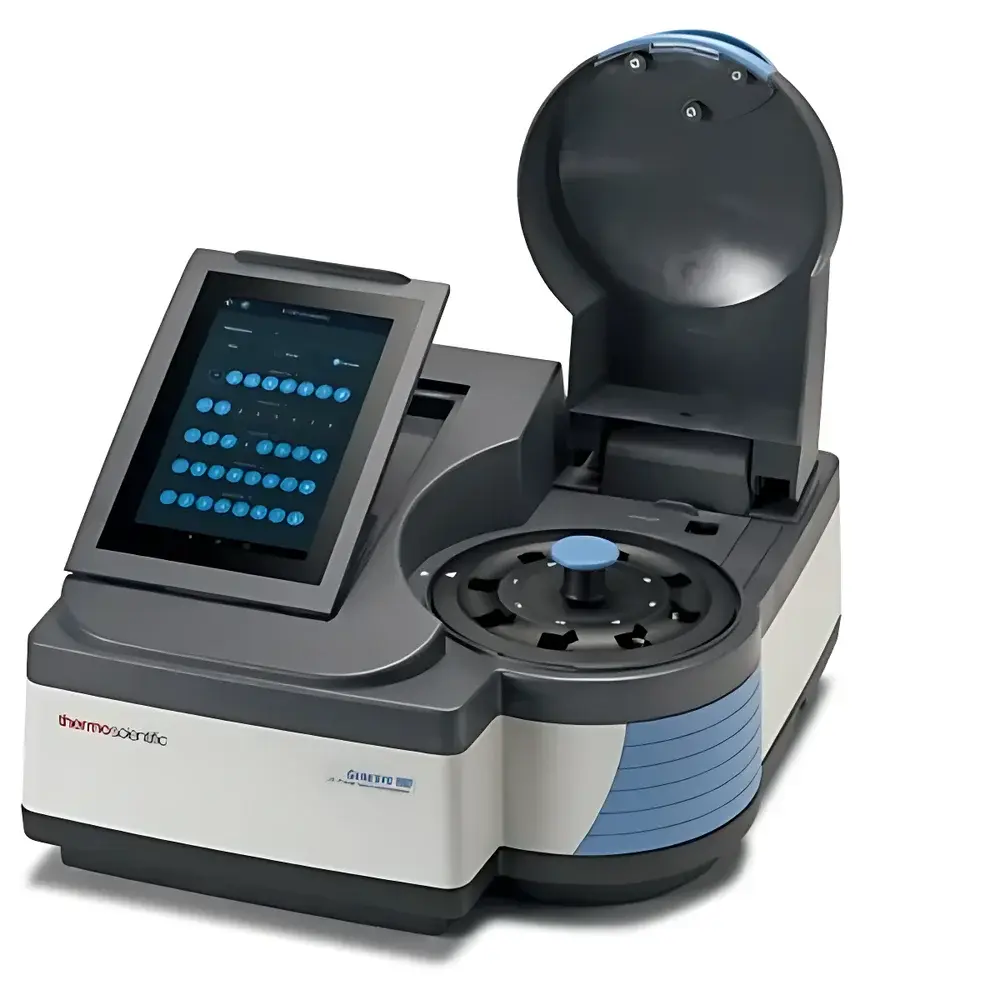
Thermo Fisher GENESYS™ 180 UV-Vis Spectrophotometer (Domestically Manufactured in Shanghai)
| Brand | Thermo Fisher |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Domestic |
| Model | GENESYS™ 180 UV-Vis Spectrophotometer (Domestically Manufactured) |
| Quotation | Upon Request |
| Wavelength Range | 190–1100 nm |
| Spectral Bandwidth | 2 nm |
| Optical Design | Double-Beam |
| Photometric Accuracy | ±0.002 A at 0.5 A, ±0.004 A at 1.0 A, ±0.008 A at 2.0 A |
| Photometric Range | –2 A to +3.5 A |
| Photometric Display Range | –3 A to +5 A |
| Wavelength Accuracy | ±0.5 nm |
| Wavelength Repeatability | ±0.2 nm |
| Wavelength Data Interval | 0.1, 0.2, 0.5, 1, 2, or 5 nm |
| Baseline Flatness | ±0.002 A |
| Stray Light | <1.0%T @ 198 nm (KCl), <0.05%T @ 220 nm (NaI), <0.03%T @ 340 nm (NaNO₂) |
| Noise (RMS, 60 readings @ 500 nm) | ≤0.00020 A at 0 A, ≤0.00030 A at 1 A, ≤0.00040 A at 2 A |
| Detector Type | Dual Silicon Photodiodes |
| Display | 7-inch HD color touchscreen (800 × 1280 px), tilt-adjustable |
| Sample Compartment | Standard 8-cell changer with swappable sample tower |
| Optional Accessories | 4-cell long-path changer (up to 100 mm pathlength), Peltier-controlled single-cell holder (20–60 °C), syringe autosampler, fiber optic probe coupler |
| Connectivity | Dual USB-A ports, Ethernet, Wi-Fi adapter support (optional), USB printing support |
| Dimensions (L × W × H) | 35.5 × 38.5 × 19.5 cm |
| Weight | 7.5 kg |
| Lamp Lifetime | >5 years (3-year limited warranty) |
| Power Supply | Auto-ranging AC/DC adapter (100–240 V, 50/60 Hz) |
| Scan Speed | Slow / Medium / Fast — up to 1600 nm/min |
| Compliance | Designed and validated per ISO 9001 manufacturing standards |
Overview
The Thermo Fisher GENESYS™ 180 UV-Vis Spectrophotometer (Domestically Manufactured in Shanghai) is a double-beam optical instrument engineered for high-fidelity absorbance, transmittance, and concentration measurements across the ultraviolet and visible spectrum (190–1100 nm). Its double-beam architecture continuously compares sample and reference beam intensities in real time, compensating for source drift, detector instability, and environmental fluctuations—ensuring exceptional baseline stability (±0.002 A) and photometric reproducibility. With a fixed 2 nm spectral bandwidth and wavelength accuracy of ±0.5 nm, the system meets routine analytical demands in quality control, academic research, and regulatory-compliant environments. The instrument’s robust mechanical design, thermally stabilized optics, and auto-ranging power supply enable reliable operation under variable ambient lighting conditions—including open-compartment measurement workflows—without compromising photometric integrity.
Key Features
- 7-inch high-resolution color touchscreen (800 × 1280 px) with ergonomic tilt adjustment and intuitive icon-driven navigation—no external PC required for method execution or data review.
- Standard 8-position automated cell changer with modular, field-swappable sample towers—enabling rapid reconfiguration between cuvette-based and macro-cell configurations.
- Dual silicon photodiode detection system optimized for low-noise performance: RMS noise ≤0.00020 A at 0 A (260/500 nm), supporting high-sensitivity quantitation of dilute biomolecules and trace analytes.
- Comprehensive connectivity suite: dual USB-A ports for peripheral attachment (e.g., flash drives, barcode scanners), Ethernet port for LAN integration, and optional Wi-Fi adapter for secure wireless data transfer compliant with IEEE 802.11ac standards.
- Expandable platform architecture: supports optional accessories including a 4-cell long-path changer (compatible with 10–100 mm pathlength cuvettes), Peltier-controlled single-cell holder (20–60 °C, ±0.2 °C stability), syringe autosampler, and fiber optic probe coupler for remote or flow-through applications.
- Pre-installed onboard software with embedded method templates (e.g., DNA/RNA A260/A280, protein Bradford/Lowry, kinetic assays), customizable report generation (PDF/CSV), and timestamped audit logs aligned with FDA 21 CFR Part 11 principles for electronic records.
Sample Compatibility & Compliance
The GENESYS™ 180 accommodates standard 10 mm square cuvettes as well as extended-path cells up to 100 mm in length—facilitating low-concentration analysis without signal amplification artifacts. Its open-compartment design permits direct insertion of non-standard vessels (e.g., microvolume stands, flow cells, Petri dishes) under controlled ambient light—a capability validated per ICH Q2(R2) guidelines for method robustness assessment. All firmware and software modules undergo periodic verification against NIST-traceable wavelength and photometric standards. Manufacturing adheres to ISO 9001:2015 quality management systems, and instrument documentation supports GLP/GMP laboratory audits through configurable user access levels, electronic signatures, and immutable audit trails for all critical operations (e.g., calibration, method modification, data export).
Software & Data Management
Onboard GENESYS™ Software v3.x provides full method development, spectral acquisition, kinetics monitoring, and multi-point quantitative analysis without dependency on external computing resources. Data files are stored in encrypted internal memory with automatic backup to USB media. Export formats include CSV (for LIMS integration), PDF (with embedded metadata and digital signature), and XML (compatible with Thermo Fisher’s Chromeleon CDS for cross-platform chromatography-spectroscopy correlation). Audit trail functionality records operator ID, timestamp, action type, and pre-/post-value changes for every parameter edit—satisfying ALCOA+ (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available) data integrity criteria. Remote software updates are delivered via secure HTTPS channels with SHA-256 signature validation.
Applications
- Quantitative nucleic acid and protein analysis (A260, A280, A230) using built-in purity ratio calculators and extinction coefficient libraries.
- Colorimetric assay validation (e.g., ELISA, enzymatic kinetics, dye-binding methods) with programmable multi-wavelength endpoints and time-resolved scanning.
- Pharmaceutical excipient and active ingredient verification per USP and EP 2.2.25 monographs, including limit tests for iron, copper, and heavy metals via complexometric spectrophotometry.
- Environmental water quality testing (e.g., nitrate, phosphate, COD) following APHA Standard Methods 4500 series protocols.
- Materials science characterization of thin-film transmittance/absorbance profiles and bandgap estimation via Tauc plot analysis.
FAQ
Is the GENESYS™ 180 compliant with FDA 21 CFR Part 11 for electronic records and signatures?
Yes—the instrument’s onboard software supports role-based user authentication, electronic signatures with biometric or token-based options, and tamper-evident audit trails meeting Part 11 Subpart B requirements.
Can the system be integrated into an existing LIMS environment?
Yes—CSV and XML export formats, combined with Ethernet/Wi-Fi connectivity, enable seamless bidirectional data exchange with major LIMS platforms via HL7 or ASTM E1384-compliant interfaces.
What is the expected lifetime of the deuterium and tungsten-halogen lamps?
Both lamps are rated for >5,000 hours of operational life; Thermo Fisher provides a 3-year limited warranty covering lamp replacement and photometric recalibration.
Does the 8-cell changer support temperature-controlled operation?
The standard 8-cell changer operates at ambient temperature; for thermal regulation, the optional Peltier-controlled single-cell holder (20–60 °C) must be installed separately.
How is wavelength calibration verified and maintained?
The system includes factory-installed holmium oxide and didymium filters for periodic verification; users may perform automated wavelength calibration using the built-in filter wheel or external NIST-traceable standards.